Advanced Metal-Free Synthesis of 3-Spiro-Tricyclic Indolone Derivatives for Commercial Scale-Up
Introduction to Patent CN112250614B: A Breakthrough in Spiro-Indolone Chemistry
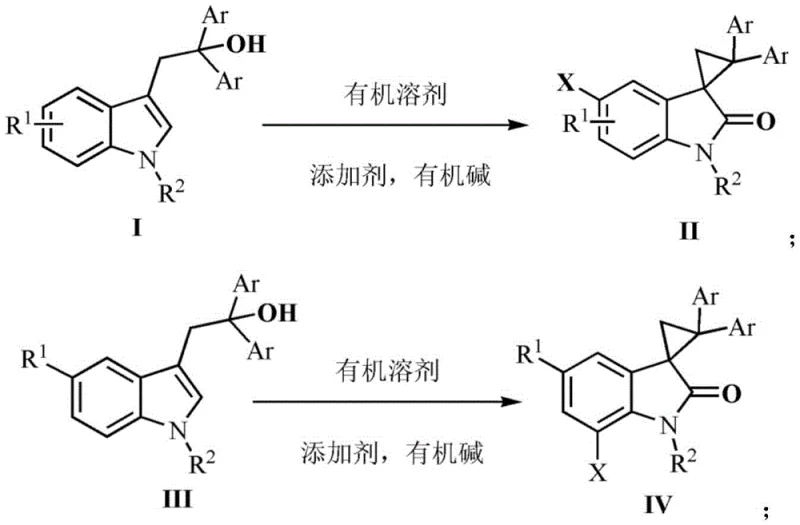
The pharmaceutical and agrochemical industries continuously seek efficient routes to complex heterocyclic scaffolds, particularly those containing spiro-cyclic motifs which are prevalent in bioactive molecules. Patent CN112250614B discloses a highly innovative synthesis method for 3-spiro-tricyclic indolone derivatives, a core structure found in potential anti-HIV agents, obesity inhibitors, and adrenergic agonists. This technology represents a significant leap forward by enabling the construction of the spiro[cyclopropane-1,3'-indol]-2'-one skeleton in a single pot, bypassing the multi-step sequences typically required for such complex architectures. The process utilizes readily available 3-indole ethanol compounds as starting materials and employs N-bromosuccinimide (NBS) or N-chlorosuccinimide (NCS) as the key additive under mild alkaline conditions. By operating at temperatures between 20°C and 60°C, this method offers a safer and more energy-efficient alternative to traditional high-energy cyclizations. For R&D teams and procurement specialists alike, this patent outlines a pathway to high-purity intermediates that avoids the economic and environmental burdens of heavy metal catalysis.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of spiro(3,3'-cyclopropane)-2-indolones has been plagued by significant technical and economic hurdles that hinder their widespread adoption in commercial manufacturing. Traditional routes often necessitate the pre-functionalization of substrates with alkyl chains followed by separate cyclization steps, effectively doubling the unit operations and reducing overall throughput. Furthermore, many established protocols rely on hazardous reagents such as diazo compounds, which pose severe safety risks regarding explosion and toxicity during scale-up. Perhaps most critically for cost-conscious manufacturers, conventional methods frequently depend on noble metal salts like palladium or silver to drive the cyclization. These precious metals not only inflate raw material costs but also introduce complex downstream purification challenges, requiring expensive scavenger resins to meet stringent residual metal specifications for pharmaceutical applications. The cumulative effect of these factors is a synthesis route that is fragile, costly, and difficult to scale reliably.
The Novel Approach
In stark contrast, the methodology described in CN112250614B revolutionizes the production landscape by introducing a direct, metal-free one-pot transformation. This novel approach leverages the dual functionality of N-halosuccinimides (NBS or NCS) to act simultaneously as an oxidant and a halogen source, facilitating a cascade of dearomatization, cyclization, and rearomatization events without external metal catalysts. The reaction conditions are remarkably mild, proceeding efficiently at room temperature or slightly elevated temperatures (up to 60°C) in common organic solvents like acetonitrile. This simplicity translates directly into operational excellence; the elimination of hazardous diazo reagents enhances workplace safety, while the absence of precious metals drastically simplifies the workup procedure. For a reliable pharmaceutical intermediate supplier, this means a robust process capable of delivering consistent quality with significantly reduced input costs and waste generation, aligning perfectly with modern green chemistry principles and supply chain efficiency goals.
Mechanistic Insights into NBS-Mediated Electrophilic Cyclization
Understanding the mechanistic underpinnings of this transformation is crucial for R&D directors aiming to optimize impurity profiles and ensure batch-to-batch consistency. The reaction initiates with the generation of a bromonium ion species from NBS, which acts as an electrophile towards the electron-rich indole ring. In the case of unsubstituted substrates (Formula I), the bromonium ion attacks the C2-C3 double bond or facilitates an electrophilic aromatic substitution depending on the specific electronic environment, leading to a key intermediate. Subsequently, the pendant alcoholic hydroxyl group on the side chain performs a nucleophilic attack, triggering a ring-closing event that forms a transient five-membered ether or similar cyclic intermediate. This is followed by a dehydrohalogenation step mediated by the organic base, which restores aromaticity to the benzene ring. The final and most critical step involves an intramolecular oxygen migration and ring contraction, driven by the instability of the intermediate furan-like skeleton in the presence of the diphenyl-substituted cyclopropane precursor. This cascade elegantly constructs the strained spiro-cyclopropane ring fused to the indolone core in a single operation.
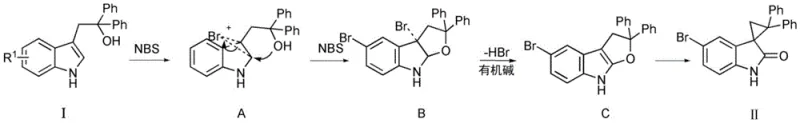
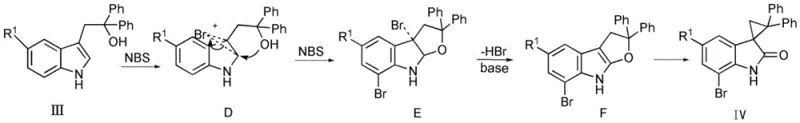
Regioselectivity is a paramount concern when dealing with substituted indoles, and the patent provides clear mechanistic rationale for controlling substitution patterns. When the starting material possesses a substituent at the C5 position (Formula III), the electronic density and steric hindrance dictate that the electrophilic bromination occurs preferentially at the C7 position. This results in the formation of a distinct brominated intermediate (Intermediate E) which follows a similar cyclization and rearrangement pathway to yield the 7-bromo spiro-indolone derivative (Formula IV). This level of control is vital for medicinal chemists who may require specific halogen handles for subsequent cross-coupling reactions. The ability to predictably direct the halogenation site simply by choosing the appropriate starting indole substrate demonstrates the versatility of this chemistry. It allows for the modular synthesis of diverse libraries of spiro-indolones, enabling rapid structure-activity relationship (SAR) studies without the need for protecting group strategies or late-stage functionalization steps that often compromise yield.
How to Synthesize 3-Spiro-Tricyclic Indolone Derivatives Efficiently
The practical execution of this synthesis is designed for ease of implementation in both laboratory and pilot plant settings. The protocol requires standard glassware and does not demand exotic equipment, making it accessible for immediate technology transfer. The process begins by charging the reactor with the 3-indole ethanol substrate and the halogenating agent under an inert atmosphere to prevent oxidative side reactions. Following the addition of the solvent and base, the reaction is allowed to proceed with simple stirring, monitoring progress via standard analytical techniques such as TLC or HPLC. Upon completion, the workup involves straightforward concentration and purification, typically yielding products with purity exceeding 95% as confirmed by NMR spectroscopy. For detailed operational parameters including exact stoichiometric ratios, solvent volumes, and specific chromatography conditions, please refer to the standardized guide below.
- Dissolve the 3-indole ethanol substrate (Formula I or III) and N-bromosuccinimide (NBS) in an organic solvent such as acetonitrile under a protective atmosphere.
- Add an organic base such as triethylamine to the reaction mixture and stir at a temperature between 20°C and 60°C for 6 to 15 hours.
- Upon completion, concentrate the reaction mixture under vacuum and purify the crude product via column chromatography to isolate the target spiro-indolone derivative.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this NBS-mediated synthesis strategy offers compelling economic and logistical benefits that extend far beyond the laboratory bench. The primary value driver is the complete elimination of noble metal catalysts, which removes a major variable cost component and eradicates the need for costly metal scavenging processes. This simplification of the downstream processing workflow significantly reduces the consumption of auxiliary materials and shortens the overall production cycle time. Furthermore, the use of commodity chemicals like NBS and triethylamine ensures a stable and resilient supply chain, mitigating the risk of raw material shortages that often plague specialty catalyst markets. The mild reaction conditions also translate to lower energy consumption, as there is no requirement for cryogenic cooling or high-temperature heating, contributing to a reduced carbon footprint and lower utility costs per kilogram of product manufactured.
- Cost Reduction in Manufacturing: The transition to a metal-free protocol fundamentally alters the cost structure of producing these complex intermediates. By removing palladium or silver salts, manufacturers avoid the volatility of precious metal prices and the capital expenditure associated with metal recovery systems. Additionally, the one-pot nature of the reaction consolidates multiple synthetic steps into a single vessel, reducing labor hours, solvent usage, and waste disposal fees. This streamlined approach allows for substantial cost savings in API manufacturing, making the final drug substance more competitive in the global market while maintaining healthy margins for the intermediate supplier.
- Enhanced Supply Chain Reliability: Reliability is the cornerstone of a robust pharmaceutical supply chain, and this synthesis method enhances security of supply through the use of widely available reagents. Unlike specialized ligands or air-sensitive catalysts that may have long lead times or single-source dependencies, NBS and organic bases are produced at massive scales globally. This abundance ensures that production schedules are not disrupted by raw material logistics. Moreover, the operational simplicity and safety profile of the process reduce the likelihood of unplanned shutdowns due to safety incidents, ensuring consistent delivery performance to downstream customers who rely on just-in-time inventory models for their own production lines.
- Scalability and Environmental Compliance: Scaling chemical processes often introduces new challenges, but the benign nature of this reaction facilitates smooth scale-up from grams to tons. The absence of explosive diazo compounds removes a significant safety barrier to increasing batch sizes, allowing for larger production runs that improve economies of scale. From an environmental perspective, the atom economy of the reaction is superior to traditional methods, generating less hazardous waste. The use of acetonitrile, a solvent with well-established recovery and recycling protocols, further supports sustainability goals. This alignment with green chemistry principles helps manufacturers meet increasingly stringent regulatory requirements regarding waste discharge and solvent emissions, future-proofing the production facility against evolving environmental legislation.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and technical disclosures within the patent documentation to provide accurate guidance for potential partners. Understanding these nuances is essential for evaluating the feasibility of integrating this route into existing manufacturing portfolios.
Q: What are the primary advantages of this NBS-mediated synthesis over traditional methods?
A: This method eliminates the need for hazardous diazo compounds and expensive noble metal catalysts like palladium or silver. It operates under mild conditions (20-60°C) in a one-pot process, significantly improving atom economy and operational safety while reducing purification costs associated with metal removal.
Q: How does the regioselectivity work when the indole C5 position is substituted?
A: When the C5 position is substituted (Formula III), the electrophilic bromination occurs selectively at the C7 position due to steric and electronic factors. This ensures the formation of the 7-bromo spiro-indolone derivative (Formula IV) rather than the 5-bromo isomer, allowing for precise structural control in downstream derivatization.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the patent demonstrates successful gram-scale synthesis with high yields and purity (>95%). The use of common solvents like acetonitrile and inexpensive reagents like NBS and triethylamine makes the process highly scalable and economically viable for industrial manufacturing without requiring specialized high-pressure or cryogenic equipment.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Spiro-Tricyclic Indolone Supplier
The technological advancements detailed in CN112250614B represent a significant opportunity for the pharmaceutical industry to access high-quality spiro-indolone intermediates more efficiently. At NINGBO INNO PHARMCHEM, we possess the technical expertise and infrastructure to leverage this metal-free synthesis route for your specific project needs. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from clinical trials to market launch is seamless. We operate stringent purity specifications and maintain rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee that every batch meets the highest standards of quality and consistency required for GMP manufacturing environments.
We invite you to collaborate with us to explore how this innovative synthesis method can optimize your supply chain and reduce your overall cost of goods. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements and quality targets. Please contact us today to request specific COA data for our catalog compounds or to discuss route feasibility assessments for your proprietary molecules. Let us be your partner in turning complex chemical challenges into commercial successes.
