Advanced Manufacturing of 2-Alkyl Amino Pyrimidones for Global Pharmaceutical Intermediates
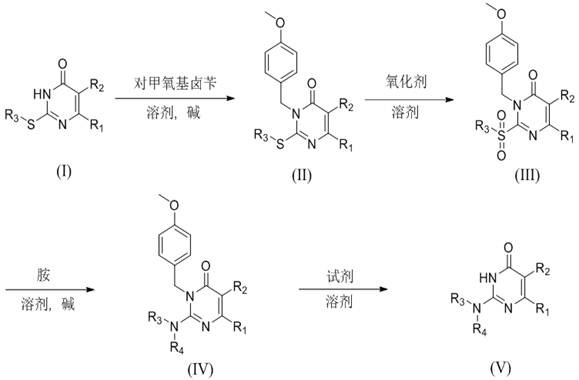
The pharmaceutical and fine chemical industries are constantly seeking robust synthetic routes for heterocyclic scaffolds that serve as critical building blocks for bioactive molecules. Patent CN110078674B discloses a highly efficient preparation method for 2-alkyl amino pyrimidones, a class of compounds renowned for their diverse pharmacological activities including anti-cancer, anti-viral, and anti-inflammatory properties. This innovative methodology transforms easily accessible 2-alkyl mercapto pyrimidones into valuable amino-substituted derivatives through a strategic four-step sequence involving protection, oxidative activation, nucleophilic substitution, and deprotection. By leveraging the superior leaving group ability of the sulfone moiety generated in situ, this route overcomes many kinetic barriers associated with direct amination of pyrimidine rings. The process is characterized by mild reaction conditions, typically operating between 0°C and 100°C, which significantly reduces energy consumption and thermal degradation risks. Furthermore, the use of inexpensive reagents like p-methoxybenzyl chloride and ceric ammonium nitrate ensures that the economic viability of the process remains high even at substantial production volumes.
For R&D directors evaluating new supply chains, the structural fidelity and impurity profile offered by this patent are of paramount importance, as the method allows for the rapid synthesis of a wide library of substituted analogs for biological screening without compromising on purity standards.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic pathways for accessing 2-amino pyrimidones often rely on direct nucleophilic aromatic substitution using halogenated precursors, which frequently necessitate harsh reaction conditions such as high temperatures and strong bases that can degrade sensitive functional groups. In many conventional protocols, the introduction of the amino group at the C2 position is hindered by the moderate electrophilicity of the carbon center, leading to incomplete conversions and the formation of difficult-to-remove byproducts. Additionally, direct alkylation strategies often suffer from poor regioselectivity, resulting in mixtures of N-alkylated and C-alkylated products that require extensive and costly chromatographic purification. The reliance on expensive transition metal catalysts in some modern cross-coupling approaches further exacerbates the cost burden and introduces the risk of heavy metal contamination, which is strictly regulated in pharmaceutical intermediates. Moreover, the lack of effective protecting group strategies in older methods often leads to polymerization or decomposition of the pyrimidone core under vigorous reaction conditions. These cumulative inefficiencies result in lower overall yields and extended production timelines, making conventional methods less attractive for commercial scale-up.
The Novel Approach
The novel approach detailed in the patent data revolutionizes this landscape by employing a thioether-to-sulfone activation strategy that dramatically enhances the reactivity of the pyrimidine ring towards nucleophilic attack. By first protecting the ring nitrogen with a p-methoxybenzyl (PMB) group, the method effectively masks the nucleophilic site, preventing unwanted side reactions and directing the subsequent chemistry exclusively to the C2 position. The oxidation of the thioether to a sulfone creates an exceptional leaving group, facilitating smooth displacement by various amines under remarkably mild conditions, often below 50°C. This strategic modification not only accelerates the reaction kinetics but also broadens the scope of compatible amines, allowing for the incorporation of sterically hindered or electronically diverse amine fragments. The final deprotection step utilizes mild oxidative or acidic conditions to cleanly remove the PMB group, yielding the target molecule with high structural integrity. This comprehensive workflow eliminates the need for extreme temperatures or toxic catalysts, offering a cleaner, safer, and more economically sustainable pathway for manufacturing complex pyrimidinone derivatives.
Mechanistic Insights into Oxidative Activation and Nucleophilic Substitution
The core mechanistic breakthrough of this synthesis lies in the oxidative conversion of the thioether intermediate to the corresponding sulfone, which serves as the pivotal activation step for the entire sequence. As illustrated in the transformation of compound II to compound III, the sulfur atom acts as a redox switch; initially, the thioether is relatively stable, but upon treatment with oxidants like m-chloroperoxybenzoic acid (m-CPBA), it is converted into a highly electron-withdrawing sulfonyl group. 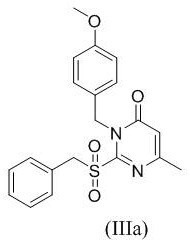
This electronic modification profoundly increases the electrophilicity of the adjacent C2 carbon atom by pulling electron density away from the ring system through strong inductive effects. Consequently, the energy barrier for the nucleophilic attack by amines is significantly lowered, allowing the substitution reaction to proceed efficiently even with weaker nucleophiles or at lower temperatures. The stability of the sulfone intermediate also permits isolation and purification if necessary, providing a crucial quality control checkpoint before the introduction of the amine fragment. This level of control is essential for maintaining batch-to-batch consistency in a GMP environment.
Impurity control is meticulously managed through the use of the p-methoxybenzyl (PMB) protecting group, which plays a dual role in stabilizing the molecule and directing regioselectivity. During the amination step, the PMB group on the nitrogen atom prevents the amine reactant from attacking the ring nitrogen, a common side reaction in unprotected systems that leads to quaternary salts or polymeric impurities. The steric bulk and electronic properties of the PMB group ensure that the incoming amine attacks solely at the activated C2-sulfone position, resulting in a clean substitution profile. Furthermore, the final deprotection using ceric ammonium nitrate proceeds via a single-electron transfer mechanism that cleaves the benzylic C-N bond without affecting the newly formed amino group or the pyrimidone core. This orthogonal reactivity ensures that the final product, such as compound Va shown in the examples, is obtained with high HPLC purity, often exceeding 97%, minimizing the need for resource-intensive recrystallization or column chromatography steps.
How to Synthesize 2-Alkyl Amino Pyrimidone Efficiently
The synthesis protocol outlined in the patent provides a standardized framework for producing these valuable intermediates with high reproducibility and yield. The process begins with the protection of the starting mercapto pyrimidone, followed by the critical oxidation step that activates the ring for substitution. Detailed operational parameters, including specific solvent choices like DMF or dichloromethane and precise molar ratios of reagents, are optimized to maximize efficiency. The subsequent amination and deprotection steps are designed to be telescoped where possible, reducing solvent swaps and handling time. For a complete breakdown of the standardized operating procedures and safety guidelines, please refer to the technical guide below.
- Protect the amino group of 2-alkyl mercapto pyrimidone using p-methoxybenzyl chloride in the presence of a base and solvent.
- Oxidize the protected thioether intermediate to the corresponding 2-sulfonyl pyrimidone using an oxidant like m-CPBA.
- Perform nucleophilic substitution with various amines or amine salts to replace the sulfone group, forming the protected amino derivative.
- Remove the p-methoxybenzyl protecting group using ceric ammonium nitrate or similar reagents to obtain the final 2-alkyl amino pyrimidone.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, this synthetic route offers substantial advantages by leveraging commodity chemicals and eliminating process bottlenecks associated with traditional methods. The reliance on widely available starting materials such as 2-mercapto pyrimidones and p-methoxybenzyl chloride ensures a stable and resilient supply chain that is less susceptible to market volatility. The avoidance of precious metal catalysts removes the complexity and cost associated with metal scavenging and residual metal testing, which are significant hidden costs in pharmaceutical manufacturing. Additionally, the mild reaction temperatures reduce the energy load on production facilities, contributing to lower utility costs and a smaller carbon footprint. The simplicity of the workup procedures, which often involve basic filtration or liquid-liquid extraction, translates to faster cycle times and higher throughput in multipurpose reactors.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts and the use of cost-effective oxidants like hydrogen peroxide or m-CPBA drastically lowers the raw material bill of goods. By avoiding high-temperature and high-pressure equipment requirements, the process reduces capital expenditure on specialized reactor infrastructure and maintenance. The high yields reported in the examples, often exceeding 90% for key steps, minimize waste generation and maximize the output per batch, leading to significant overall cost savings. Furthermore, the ability to use crude intermediates in subsequent steps without rigorous purification reduces solvent consumption and labor costs associated with isolation.
- Enhanced Supply Chain Reliability: The reagents required for this synthesis, including common bases like sodium carbonate and solvents like ethanol or acetonitrile, are globally sourced commodities with robust supply networks. This diversification of raw material sources mitigates the risk of supply disruptions that can occur with specialized or proprietary reagents. The operational flexibility of the process, which tolerates a range of solvents and bases, allows manufacturing sites to adapt quickly to local availability without compromising product quality. This resilience ensures consistent delivery schedules for downstream customers, fostering stronger long-term partnerships.
- Scalability and Environmental Compliance: The process is inherently scalable due to its exothermic nature being manageable under mild conditions, allowing for safe expansion from pilot plant to commercial tonnage. The waste streams generated are primarily organic salts and aqueous washes that are easier to treat compared to heavy metal-containing effluents from catalytic processes. The high atom economy of the substitution step and the efficient recovery of solvents contribute to a greener manufacturing profile that aligns with increasingly stringent environmental regulations. This compliance reduces the regulatory burden and potential fines, securing the long-term viability of the production line.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation to provide clarity on feasibility and performance. Understanding these details helps stakeholders make informed decisions about integrating this route into their existing manufacturing portfolios.
Q: What are the key advantages of the sulfone activation strategy in this synthesis?
A: The oxidation of the thioether to a sulfone group significantly increases the electrophilicity of the C2 position, allowing for mild nucleophilic substitution with diverse amines under relatively low temperatures, thereby minimizing side reactions and improving overall yield.
Q: How does this method ensure high purity for pharmaceutical applications?
A: By utilizing a p-methoxybenzyl (PMB) protecting group on the ring nitrogen, the method effectively prevents unwanted N-alkylation during the amine substitution step, ensuring that the reaction occurs selectively at the C2 position and simplifying downstream purification.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the process utilizes cheap and readily available reagents such as p-methoxybenzyl chloride and common oxidants, operates under mild temperature conditions ranging from 0°C to 100°C, and involves straightforward workup procedures like filtration and extraction, making it highly scalable.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Alkyl Amino Pyrimidone Supplier
At NINGBO INNO PHARMCHEM, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from laboratory concept to industrial reality. Our state-of-the-art facilities are equipped to handle the specific solvent systems and reaction conditions required for this pyrimidone synthesis, maintaining stringent purity specifications throughout the manufacturing process. With our rigorous QC labs and advanced analytical capabilities, we guarantee that every batch meets the highest international standards for pharmaceutical intermediates. We understand the critical nature of supply continuity and have established robust inventory management systems to support your long-term production needs.
We invite you to contact our technical procurement team to discuss how this innovative synthesis route can optimize your supply chain and reduce your overall manufacturing costs. Request a Customized Cost-Saving Analysis today to see the specific financial benefits tailored to your volume requirements. Our experts are ready to provide specific COA data and route feasibility assessments to demonstrate our commitment to quality and partnership. Let us be your trusted partner in delivering high-performance chemical solutions for the global market.
