Advanced Green Synthesis of Hexahydro-2-cyclopentopyrrolylamine Hydrochloride for Scalable Gliclazide Production
The pharmaceutical landscape for Type II diabetes treatment continues to rely heavily on second-generation sulfonylureas, with Gliclazide standing out as a cornerstone therapy due to its dual action on blood glucose and vascular complications. At the heart of manufacturing this critical active pharmaceutical ingredient lies the efficient production of its key precursor, hexahydro-2-cyclopentopyrrolylamine hydrochloride. Patent CN102924362A introduces a transformative approach to synthesizing this vital intermediate, shifting away from traditional, waste-intensive methodologies toward a sophisticated green chemistry platform. This innovation leverages a specialized Gemini surfactant reaction system to facilitate a one-pot reduction, fundamentally altering the economic and environmental profile of the supply chain. For R&D directors and procurement strategists, this patent represents not merely a procedural update but a strategic opportunity to optimize the cost structure and sustainability metrics of anti-diabetic drug manufacturing. By integrating mild reaction conditions with a recyclable catalytic medium, the technology addresses the growing industry pressure to reduce the carbon footprint of fine chemical production while maintaining rigorous purity standards required for global regulatory compliance.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of cyclic amine intermediates like hexahydro-2-cyclopentopyrrolylamine has been plagued by operational complexities that hinder scalability and inflate production costs. Traditional routes often necessitate the use of stoichiometric amounts of harsh reducing agents in conjunction with strong acid catalysts, which create severe corrosion issues for standard stainless steel reactor vessels and demand expensive lining materials. Furthermore, these conventional processes typically generate substantial quantities of acidic wastewater and salt by-products, imposing a heavy burden on downstream waste treatment facilities and increasing the overall environmental compliance costs for manufacturers. The multi-step nature of older methodologies also introduces multiple isolation points, each representing a potential yield loss and a vector for impurity accumulation, thereby complicating the purification process and jeopardizing the final quality of the Gliclazide API. Additionally, the reliance on volatile organic solvents in large volumes exacerbates safety risks related to flammability and worker exposure, creating a operational environment that is increasingly untenable under modern industrial safety regulations.
The Novel Approach
In stark contrast to these legacy challenges, the methodology disclosed in CN102924362A utilizes a diphenylmethane-based Gemini surfactant to create a highly efficient micro-reactor environment within the reaction vessel. This novel approach eliminates the need for external acid catalysts during the critical reduction phase, relying instead on the unique amphiphilic properties of the surfactant to solubilize the hydrophobic N-amino-1,2-cyclopentanedicarboximide substrate effectively. 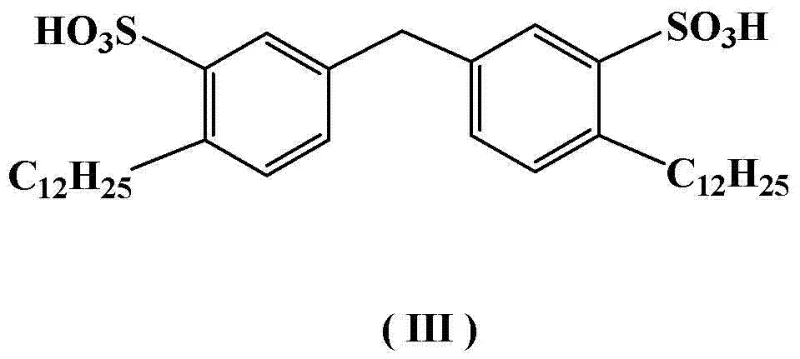 As illustrated by the specific structure of the surfactant, the presence of two hydrophilic sulfonic acid heads and two lipophilic dodecyl tails allows for the formation of stable micelles that concentrate the reactants, thereby accelerating the reaction kinetics at significantly lower temperatures ranging from 50°C to 100°C. This shift to milder thermal conditions not only preserves the integrity of the sensitive cyclic structure but also drastically reduces energy consumption associated with heating and cooling cycles. The ability to operate this system in a true one-pot fashion streamlines the workflow, removing the need for intermediate isolations and significantly shortening the overall cycle time from raw material input to finished intermediate.
As illustrated by the specific structure of the surfactant, the presence of two hydrophilic sulfonic acid heads and two lipophilic dodecyl tails allows for the formation of stable micelles that concentrate the reactants, thereby accelerating the reaction kinetics at significantly lower temperatures ranging from 50°C to 100°C. This shift to milder thermal conditions not only preserves the integrity of the sensitive cyclic structure but also drastically reduces energy consumption associated with heating and cooling cycles. The ability to operate this system in a true one-pot fashion streamlines the workflow, removing the need for intermediate isolations and significantly shortening the overall cycle time from raw material input to finished intermediate.
Mechanistic Insights into Gemini Surfactant-Mediated Reduction
The core of this technological breakthrough lies in the synergistic interaction between the potassium borohydride reducing agent and the organized assembly of the Gemini surfactant molecules. In this system, the surfactant acts as a phase transfer catalyst and a solubilizing agent simultaneously, creating a localized high-concentration zone for the reactants within the micellar pseudophase. 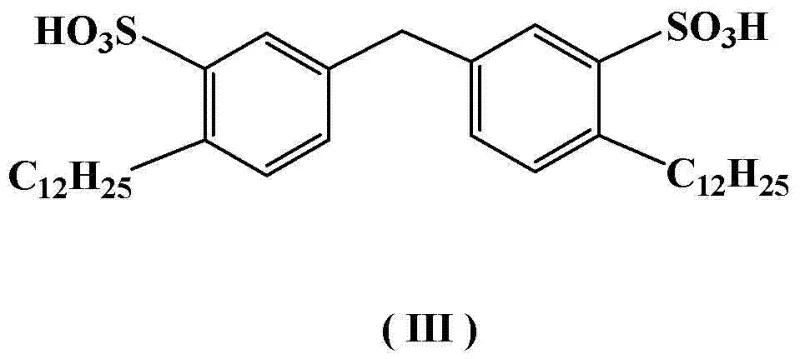 The mechanism proceeds through the nucleophilic attack of the hydride ion on the carbonyl carbons of the imide ring, facilitated by the proximity effects enforced by the micellar structure. Unlike bulk solvent reactions where diffusion limits the rate, the micellar environment ensures that the borohydride anions are readily available at the interface where the organic substrate resides. This precise control over the reaction milieu suppresses competing side reactions, such as the over-reduction of the ring or the hydrolysis of the amine product, which are common pitfalls in aqueous reductions. The result is a highly selective transformation that consistently delivers the target hexahydro-2-cyclopentopyrrolylamine with exceptional chemical fidelity, minimizing the formation of structural analogs that are difficult to separate in later stages.
The mechanism proceeds through the nucleophilic attack of the hydride ion on the carbonyl carbons of the imide ring, facilitated by the proximity effects enforced by the micellar structure. Unlike bulk solvent reactions where diffusion limits the rate, the micellar environment ensures that the borohydride anions are readily available at the interface where the organic substrate resides. This precise control over the reaction milieu suppresses competing side reactions, such as the over-reduction of the ring or the hydrolysis of the amine product, which are common pitfalls in aqueous reductions. The result is a highly selective transformation that consistently delivers the target hexahydro-2-cyclopentopyrrolylamine with exceptional chemical fidelity, minimizing the formation of structural analogs that are difficult to separate in later stages.
From an impurity control perspective, the absence of strong mineral acids during the reduction step is paramount for maintaining product quality. Acidic conditions can often lead to the protonation of the amine product prematurely or induce ring-opening degradation pathways, generating complex impurity profiles that require extensive chromatographic purification. By maintaining a near-neutral to mildly acidic environment governed by the surfactant's own sulfonic groups and the controlled addition of HCl only during the workup phase, the process ensures that the amine salt forms cleanly and precipitates or crystallizes with high purity. The patent data indicates that this mechanistic advantage translates directly into analytical results, with gas chromatography analysis confirming purities exceeding 98% without the need for aggressive recrystallization protocols. This level of intrinsic purity is critical for pharmaceutical intermediates, as it reduces the burden on the API manufacturer to perform additional purification steps, thereby preserving yield and reducing the overall cost of goods for the final Gliclazide drug substance.
How to Synthesize Hexahydro-2-cyclopentopyrrolylamine Hydrochloride Efficiently
Implementing this synthesis route requires precise adherence to the thermal and stoichiometric parameters defined in the patent to maximize the benefits of the Gemini surfactant system. The process begins with the preparation of the reaction medium, where the surfactant is preheated to ensure proper fluidity and micelle formation before the introduction of the reducing agent. Operators must carefully manage the addition sequence, introducing potassium borohydride prior to the substrate to establish the reducing environment, followed by the controlled addition of N-amino-1,2-cyclopentanedicarboximide. The reaction is then maintained at an optimal temperature window of 70°C to 80°C for a duration of 4 to 5 hours, a balance that ensures complete conversion while preventing thermal degradation. Following the reaction, the workup involves a straightforward extraction with dichloromethane and pH adjustment to precipitate the hydrochloride salt, which is finally purified via recrystallization from absolute ethanol.
- Preheat the gemini surfactant (diphenylmethane derivative) to 50°C and add potassium borohydride with stirring.
- Introduce N-amino-1,2-cyclopentanedicarboximide and maintain the reaction temperature between 70°C and 80°C for 4 to 5 hours.
- Extract the reaction mixture with dichloromethane, adjust pH to 2-3 with hydrochloric acid, and recrystallize from absolute ethanol.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this Gemini surfactant-mediated synthesis offers compelling economic arguments that extend beyond simple yield improvements. The most significant value driver is the drastic simplification of the waste management profile; by eliminating the massive salt loads associated with traditional acid-base neutralization steps, the facility can achieve substantial cost savings in effluent treatment and hazardous waste disposal. Furthermore, the patent explicitly highlights the reusability of the Gemini surfactant, a feature that transforms a consumable reagent into a recoverable asset, effectively decoupling production volume from raw material consumption for this critical component. This circular economy aspect within the manufacturing process provides a hedge against volatility in surfactant pricing and ensures a more predictable cost structure for long-term supply contracts. The simplified one-pot operation also reduces the requirement for complex multi-vessel setups, allowing existing manufacturing lines to be repurposed or debottlenecked with minimal capital expenditure, thereby enhancing overall plant utilization rates.
- Cost Reduction in Manufacturing: The elimination of expensive acid catalysts and the reduction in solvent usage directly lower the variable costs per kilogram of the intermediate produced. By avoiding the need for extensive washing steps to remove inorganic salts, the process saves both water and energy, contributing to a leaner manufacturing budget. The high conversion rate ensures that raw material utilization is maximized, minimizing the financial loss associated with unreacted starting materials. Additionally, the mild reaction conditions reduce the wear and tear on reactor equipment, extending the lifecycle of capital assets and lowering maintenance overheads. These cumulative efficiencies result in a significantly more competitive cost position for the intermediate in the global marketplace.
- Enhanced Supply Chain Reliability: The robustness of the one-pot method reduces the risk of batch failures caused by complex multi-step transfers or sensitive intermediate handling. Since the surfactant can be recovered and reused, the supply chain is less vulnerable to disruptions in the sourcing of specialty chemicals, as the initial inventory can sustain multiple production cycles. The simplified process flow also shortens the manufacturing lead time, enabling faster response to fluctuations in demand for Gliclazide APIs. This agility is crucial for maintaining service levels to downstream pharmaceutical clients who operate on just-in-time inventory models. Consequently, suppliers adopting this technology can offer more reliable delivery schedules and greater flexibility in order fulfillment.
- Scalability and Environmental Compliance: The green chemistry credentials of this process align perfectly with the increasingly stringent environmental regulations governing pharmaceutical manufacturing globally. The reduction in hazardous waste generation simplifies the permitting process for capacity expansions and reduces the risk of regulatory fines or shutdowns. The process is inherently scalable because the micellar kinetics do not suffer from the same heat and mass transfer limitations often seen in heterogeneous multiphase reactions at large scales. This means that technology transfer from pilot plant to commercial production is smoother and carries lower technical risk. Companies investing in this technology future-proof their operations against tightening carbon emission targets and sustainability mandates from corporate stakeholders.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis route for hexahydro-2-cyclopentopyrrolylamine hydrochloride. These answers are derived directly from the experimental data and claims presented in patent CN102924362A, providing a factual basis for evaluating the technology's fit within your supply chain. Understanding these details is essential for R&D teams assessing process feasibility and procurement officers calculating total cost of ownership.
Q: What are the primary advantages of using a Gemini surfactant system for this reduction?
A: The Gemini surfactant system provides a unique micro-emulsion environment that enhances the solubility of organic substrates in the aqueous phase, leading to higher conversion rates and allowing the surfactant to be recovered and reused, which significantly lowers material costs.
Q: How does this method improve impurity control compared to traditional acid-catalyzed routes?
A: By eliminating the need for strong acid catalysts during the main reduction step and operating under mild thermal conditions (50-100°C), the process minimizes side reactions such as ring-opening or polymerization, resulting in a crude purity exceeding 98%.
Q: Is this process suitable for large-scale commercial manufacturing of Gliclazide intermediates?
A: Yes, the one-pot nature of the reaction simplifies equipment requirements, reduces solvent consumption, and generates less hazardous waste, making it highly scalable and compliant with modern environmental regulations for pharmaceutical production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Hexahydro-2-cyclopentopyrrolylamine Hydrochloride Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to greener, more efficient synthetic routes is not just a regulatory necessity but a strategic imperative for maintaining competitiveness in the pharmaceutical intermediate market. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the innovative Gemini surfactant methodology described in CN102924362A can be seamlessly integrated into our manufacturing infrastructure. We are committed to delivering high-purity intermediates that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. Our capability to handle complex chemistries involving specialized surfactants and sensitive reduction reactions positions us as a preferred partner for global pharmaceutical companies seeking to optimize their Gliclazide supply chain.
We invite you to engage with our technical procurement team to discuss how this advanced synthesis route can benefit your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the potential economic advantages of switching to this green manufacturing protocol. We encourage potential partners to contact us to obtain specific COA data from our recent pilot batches and to request detailed route feasibility assessments tailored to your volume needs. Let us collaborate to build a more sustainable and cost-effective supply chain for next-generation diabetes medications.
