Advanced Palladium-Catalyzed Synthesis of Azacyclolactone Compounds for Commercial Scale-Up
Advanced Palladium-Catalyzed Synthesis of Azacyclolactone Compounds for Commercial Scale-Up
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies to construct complex heterocyclic scaffolds that serve as critical intermediates for bioactive molecules. A significant breakthrough in this domain is detailed in patent CN112624998A, which discloses a novel and efficient preparation method for azacyclolactone compounds. These nitrogen-containing mesocyclic lactones represent a privileged structure in medicinal chemistry, exhibiting potential biological activities such as anti-inflammatory, antifungal, antitumor, and antiviral properties. The patent introduces a groundbreaking [5+6] cycloaddition reaction between vinyl ethylene carbonate and isatoic anhydride derivatives, catalyzed by a palladium complex. This approach not only simplifies the synthetic route but also addresses long-standing challenges in constructing medium-sized rings, offering a streamlined pathway for the production of high-purity pharmaceutical intermediates.
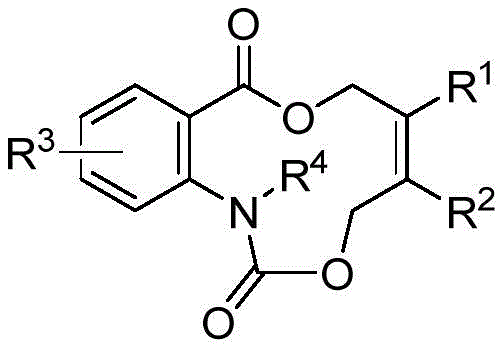
For R&D directors and process chemists, the ability to access these unique skeletons efficiently is paramount. The disclosed method utilizes vinyl ethylene carbonate as a versatile synthon, which undergoes decarboxylation to form a high-activity pi-allyl palladium zwitterion intermediate. This intermediate then engages in a cycloaddition with isatoic anhydride, effectively building the complex ring system in a single operational step. The versatility of this chemistry is underscored by the broad tolerance for substituents, allowing for the introduction of halogens, alkyl, and alkoxy groups, thereby facilitating the rapid generation of diverse compound libraries for drug discovery programs.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of azalide and azacyclolactone compounds has been fraught with significant chemical challenges. The construction of medium-sized rings often suffers from unfavorable entropy effects, where the probability of the reacting ends of a chain meeting is low, leading to poor cyclization efficiency. Furthermore, trans-ring interactions can create steric hindrance that destabilizes the transition state, resulting in low yields and the formation of unwanted byproducts. Traditional methods frequently require harsh reaction conditions, expensive reagents, or multi-step sequences that involve protecting group manipulations, which drastically increase the cost of goods and extend the production timeline. These inefficiencies pose a substantial bottleneck for procurement managers looking to secure cost-effective supplies of complex heterocyclic intermediates for large-scale API manufacturing.
The Novel Approach
In stark contrast, the methodology described in patent CN112624998A offers a paradigm shift by leveraging a palladium-catalyzed [5+6] cycloaddition strategy. This novel approach utilizes readily available building blocks—vinyl ethylene carbonate and isatoic anhydride—to construct the target azacyclolactone skeleton directly and concisely. The reaction proceeds under remarkably mild heating conditions, typically between 50-70°C, often optimized at 60°C, which minimizes thermal degradation of sensitive functional groups. By avoiding the entropic penalties associated with linear cyclization precursors, this method achieves high product yields, with specific examples demonstrating efficiencies as high as 98%. The simplicity of the post-processing, often requiring only standard column chromatography, further enhances its appeal for industrial application, significantly reducing the operational burden on manufacturing teams.
![General reaction scheme showing the Pd-catalyzed [5+6] cycloaddition between vinyl ethylene carbonate and isatoic anhydride](/insights/img/azacyclolactone-synthesis-palladium-pharma-supplier-20260306102706-04.webp)
Mechanistic Insights into Pd-Catalyzed [5+6] Cycloaddition
The core of this technological advancement lies in the sophisticated organometallic mechanism driven by the palladium catalyst. Upon initiation, the palladium(0) species coordinates with the vinyl ethylene carbonate, triggering a decarboxylation event that generates a highly reactive pi-allyl palladium zwitterion intermediate. This species acts as a 5-atom synthon, possessing significant nucleophilic and electrophilic character depending on the resonance structures involved. Simultaneously, the isatoic anhydride derivative serves as the complementary coupling partner. The interaction between these two components is facilitated by the phosphorus-containing ligands, such as triphenylphosphine, which stabilize the palladium center and modulate its electronic properties to favor the cycloaddition pathway over competing side reactions. This precise control over the catalytic cycle ensures high regioselectivity and chemoselectivity, which is critical for maintaining the purity profile required in pharmaceutical synthesis.
From an impurity control perspective, the mechanism inherently limits the formation of oligomeric byproducts often seen in uncatalyzed thermal cyclizations. The use of specific ligands like PPh3 in conjunction with catalysts such as tetrakis(triphenylphosphine)palladium creates a well-defined coordination sphere that directs the reaction towards the desired [5+6] adduct. The mild reaction temperature of 60°C further suppresses thermal decomposition pathways. For quality assurance teams, this means a cleaner crude reaction profile, which simplifies downstream purification and ensures that the final active pharmaceutical ingredient (API) precursor meets stringent regulatory specifications regarding residual solvents and metal impurities. The ability to tune the electronic nature of the substrates through R-group modifications without compromising the catalytic efficiency adds another layer of robustness to the process.
How to Synthesize Azacyclolactone Efficiently
The practical implementation of this synthesis route is designed for ease of execution in both laboratory and pilot plant settings. The protocol involves charging a reaction vessel with the vinyl ethylene carbonate derivative and the isatoic anhydride derivative, typically in a molar ratio of 2:1 to drive the equilibrium towards product formation. A palladium catalyst, such as Pd(PPh3)4, is added at a loading of 2.5-10 mol%, along with a phosphorus ligand if not pre-complexed. The reaction is conducted in a green and industrially friendly solvent like ethyl acetate, heated to 60°C, and monitored via TLC until completion. The detailed standardized synthesis steps, including specific workup procedures and purification parameters, are outlined below to ensure reproducibility and safety during scale-up operations.
- Combine vinyl ethylene carbonate and isatoic anhydride derivatives in a reaction vessel with a molar ratio of approximately 2: 1.
- Add a palladium catalyst (e.g., Pd(PPh3)4) and a phosphorus-containing ligand (e.g., PPh3) in an organic solvent such as ethyl acetate.
- Heat the mixture to 50-70°C and stir for 1-10 hours until reaction completion, followed by purification via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented synthesis route translates into tangible strategic benefits. The primary advantage lies in the drastic simplification of the supply chain for raw materials. Vinyl ethylene carbonate and isatoic anhydrides are commodity chemicals that are commercially available in bulk quantities, reducing the risk of supply disruptions associated with exotic or custom-synthesized starting materials. Furthermore, the high atom economy of the [5+6] cycloaddition minimizes waste generation, aligning with modern sustainability goals and reducing disposal costs. The operational simplicity allows for faster batch turnover times, enhancing overall manufacturing throughput without the need for specialized high-pressure or cryogenic equipment.
- Cost Reduction in Manufacturing: The economic impact of this process is significant due to the elimination of complex multi-step sequences. By consolidating the ring construction into a single catalytic step, manufacturers save on labor, energy, and solvent consumption. The use of low catalyst loadings (starting from 2.5 mol%) reduces the expense associated with precious metals, while the mild conditions lower energy utility costs. Additionally, the high yields observed across a wide substrate scope mean less raw material is wasted on failed batches or low-conversion runs, directly improving the cost per kilogram of the final intermediate.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions ensures consistent output quality, which is vital for maintaining continuous supply to downstream API producers. The tolerance for various functional groups means that a single platform technology can be used to produce a family of related intermediates, providing flexibility to respond to changing market demands. The use of common solvents like ethyl acetate simplifies logistics and storage requirements, as there is no need for hazardous or hard-to-source reagents that could delay production schedules.
- Scalability and Environmental Compliance: Scaling this process from gram to ton scale is straightforward due to the absence of exothermic hazards or unstable intermediates. The reaction can be safely performed in standard stainless steel reactors. From an environmental standpoint, the reduced waste stream and the potential for solvent recovery contribute to a lower environmental footprint. This compliance with green chemistry principles facilitates easier regulatory approval and supports corporate social responsibility initiatives focused on sustainable manufacturing practices.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this azacyclolactone synthesis technology. These insights are derived directly from the experimental data and technical specifications provided in the patent documentation, ensuring that stakeholders have accurate information for decision-making. Understanding these nuances helps in evaluating the feasibility of integrating this route into existing production portfolios.
Q: What are the key advantages of this [5+6] cycloaddition method over traditional synthesis routes?
A: This novel method overcomes adverse entropy effects and trans-ring interactions common in azalide construction. It operates under mild conditions (60°C) with high yields (up to 98%) and uses readily available building blocks like vinyl ethylene carbonate.
Q: Is this process scalable for industrial production of pharmaceutical intermediates?
A: Yes, the process utilizes simple operation steps, common solvents like ethyl acetate, and low catalyst loading (2.5-10 mol%), making it highly suitable for commercial scale-up and reducing downstream processing complexity.
Q: What is the substrate scope for this azacyclolactone synthesis?
A: The method demonstrates a wide substrate application range, tolerating various substituents on the aryl rings including halogens (F, Cl, Br), alkyl groups (methyl), and alkoxy groups (methoxy), allowing for diverse structural modifications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Azacyclolactone Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this palladium-catalyzed [5+6] cycloaddition technology for the development of next-generation therapeutics. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from benchtop discovery to full-scale manufacturing. Our state-of-the-art facilities are equipped to handle palladium-catalyzed reactions with rigorous safety protocols, and our stringent purity specifications guarantee that every batch of azacyclolactone intermediate meets the highest industry standards. Our rigorous QC labs utilize advanced analytical techniques to verify structural integrity and impurity profiles, providing you with the confidence needed for regulatory filings.
We invite you to collaborate with us to leverage this efficient synthesis route for your specific drug development programs. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating how this novel method can optimize your budget. Please contact our technical procurement team today to request specific COA data for our catalog compounds or to discuss route feasibility assessments for your custom targets. Let us be your partner in turning complex chemical challenges into commercial successes.
