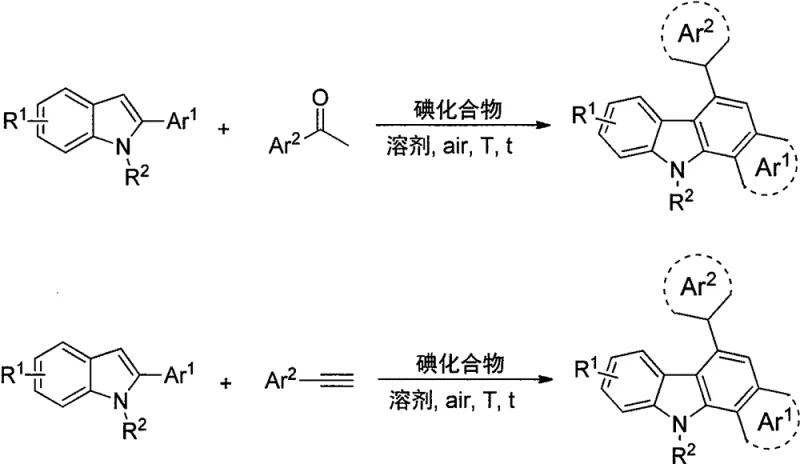
Advanced One-Pot Synthesis of Polysubstituted 6-Arylbenzo[a]Carbazole Derivatives for High-Performance Optoelectronics
The pharmaceutical and electronic materials industries are constantly seeking robust, scalable pathways to access complex nitrogen-containing fused ring systems, particularly those with extended conjugation suitable for optoelectronic applications. Patent CN109400518B introduces a groundbreaking methodology for the synthesis of polysubstituted 6-arylbenzo[a]carbazole derivatives, addressing critical bottlenecks in traditional heterocyclic chemistry. This invention details a highly efficient, one-pot protocol that converts 2-arylindole compounds and aromatic alkynes or ketones directly into stable, high-performance carbazole scaffolds using inexpensive iodine compounds as promoters. Unlike legacy methods that rely on sensitive transition metal catalysts, this approach operates under mild conditions in an air atmosphere, offering a transformative solution for the production of high-purity OLED material intermediates and bioactive scaffolds. The versatility of this chemistry allows for the generation of a vast library of derivatives, as illustrated by the diverse structural motifs achievable through this route.
![Diverse library of polysubstituted 6-arylbenzo[a]carbazole derivatives synthesized via the novel iodine-catalyzed method](/insights/img/polysubstituted-6-arylbenzo-carbazole-iodine-catalysis-electronic-supplier-20260304052608-01.webp)
Furthermore, the technical significance of this patent lies in its ability to bypass the rigorous purification steps often required to remove heavy metal residues, a common pain point in the manufacturing of electronic chemicals. By leveraging the unique reactivity of iodine species to activate C-H bonds and facilitate cyclization, the process achieves high atom economy while minimizing environmental impact. For R&D directors and process chemists, this represents a paradigm shift towards greener, more sustainable synthesis strategies that do not compromise on molecular complexity or product stability. The following analysis dissects the mechanistic advantages and commercial implications of adopting this iodine-catalyzed technology for large-scale production.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the benzo[a]carbazole core has been plagued by synthetic inefficiencies that hinder commercial viability and scalability. Traditional routes typically depend on transition metal catalysts such as palladium, ruthenium, rhodium, or copper, which introduce significant cost burdens and supply chain vulnerabilities due to the volatility of precious metal prices. Moreover, these conventional methods often necessitate multi-step sequences involving the pre-functionalization of starting materials, such as halogenation or boronation, which generates substantial chemical waste and lowers the overall process mass intensity. The requirement for strictly inert atmospheres and anhydrous conditions further complicates reactor operations, demanding specialized equipment and increasing the risk of batch failure due to oxygen or moisture sensitivity. Additionally, the removal of trace metal contaminants from the final product is a critical and expensive downstream processing step, especially for applications in organic electronics where metal impurities can drastically degrade device performance and longevity.
The Novel Approach
In stark contrast, the methodology disclosed in CN109400518B revolutionizes the synthesis landscape by employing cheap, earth-abundant iodine compounds to drive the cyclization under ambient air conditions. This novel approach eliminates the need for expensive transition metals and their associated ligands, thereby drastically simplifying the reaction setup and reducing the raw material costs. The process tolerates a wide range of functional groups on both the indole and the ketone or alkyne partners, allowing for direct coupling without the need for tedious pre-activation steps. Operating in an air atmosphere not only enhances operational safety by removing the hazards associated with pressurized inert gases but also streamlines the workflow, making it highly amenable to continuous flow processing or large-batch manufacturing. The one-pot nature of the reaction ensures that the target polysubstituted 6-arylbenzo[a]carbazole is formed selectively with high efficiency, overcoming the yield losses inherent in multi-step telescoping processes.
Mechanistic Insights into Iodine-Catalyzed Oxidative Cyclization
The core of this technological breakthrough relies on the unique ability of iodine species to act as soft electrophiles that activate the carbonyl group of aromatic ketones or the triple bond of aromatic alkynes towards nucleophilic attack by the electron-rich 2-arylindole system. As depicted in the reaction scheme, the iodine catalyst facilitates the formation of a branched olefin intermediate at the 3-position of the indole ring, which subsequently undergoes an intramolecular oxidative cyclization to close the new six-membered ring of the benzo[a]carbazole skeleton. This mechanism avoids the high-energy barriers typically associated with C-H activation, allowing the reaction to proceed at moderate temperatures between 120°C and 150°C. The use of mixed iodine sources, such as combinations of elemental iodine with iodides or hypoiodites, appears to synergistically enhance the catalytic turnover, likely by regenerating the active iodine species in situ through redox cycles involving atmospheric oxygen.
From an impurity control perspective, this mechanism is exceptionally clean because it avoids the formation of homocoupling byproducts often seen with palladium catalysis. The selectivity is driven by the specific electronic matching between the iodine-activated electrophile and the nucleophilic indole, ensuring that the cyclization occurs regioselectively at the desired position. Furthermore, the absence of strong bases or harsh oxidants minimizes side reactions such as polymerization or decomposition of sensitive functional groups, resulting in a cleaner crude reaction profile. This inherent selectivity reduces the burden on downstream purification units, allowing for simpler crystallization or chromatography protocols to achieve the stringent purity specifications required for high-end electronic materials. Understanding this mechanistic pathway allows process engineers to fine-tune reaction parameters, such as solvent polarity and iodine loading, to maximize conversion rates for specific substrate combinations.
How to Synthesize Polysubstituted 6-Arylbenzo[a]Carbazole Efficiently
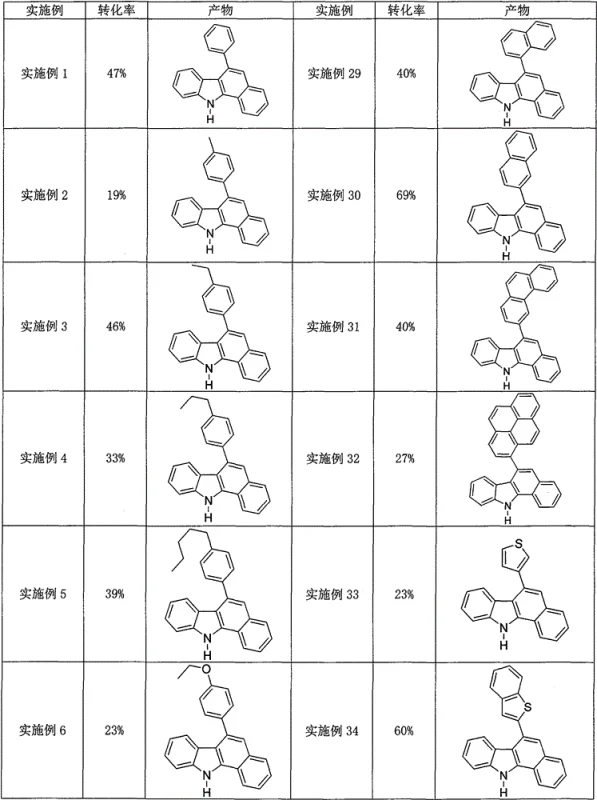
The practical implementation of this synthesis route is designed for ease of execution, requiring standard laboratory or plant equipment without the need for specialized gloveboxes or high-pressure reactors. The protocol involves simply mixing the 2-arylindole substrate with the chosen aromatic ketone or alkyne partner in a suitable organic solvent such as chlorobenzene or o-dichlorobenzene, followed by the addition of the iodine catalyst system. The detailed standardized synthesis steps, including specific molar ratios, temperature profiles, and workup procedures validated across dozens of examples, are outlined in the guide below to ensure reproducible results for your process development team.
- Combine 2-arylindole compounds with aromatic alkyne or ketone compounds and an iodine-containing catalyst in an organic solvent.
- Heat the reaction mixture to 120-150°C under an air atmosphere for 3 to 12 hours to facilitate cyclization.
- Purify the resulting crude mixture to isolate the high-purity polysubstituted 6-arylbenzo[a]carbazole derivative.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this iodine-catalyzed technology presents a compelling value proposition centered on cost stability and operational resilience. By shifting away from precious metal catalysts, manufacturers can insulate their production costs from the fluctuating market prices of palladium and rhodium, leading to significant long-term savings in raw material expenditures. The ability to run reactions in air rather than under inert gas significantly reduces utility costs and simplifies facility requirements, allowing for higher throughput in existing infrastructure without major capital investment. Moreover, the use of commodity chemicals like acetophenones and indoles as starting materials ensures a robust and diversified supply base, mitigating the risk of single-source dependency that often plagues specialized reagent procurement.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts and the associated ligand systems removes a major cost driver from the bill of materials, while the simplified one-pot process reduces labor and energy consumption per kilogram of product. The high atom economy of the reaction means less waste disposal cost, and the avoidance of pre-functionalization steps cuts down on the number of unit operations required, further driving down the overall cost of goods sold. This economic efficiency makes the commercial scale-up of complex electronic chemical intermediates far more viable, enabling competitive pricing strategies in the global market.
- Enhanced Supply Chain Reliability: Sourcing iodine compounds and simple aromatic ketones is significantly more reliable than securing high-purity organometallic catalysts, which often have long lead times and limited supplier options. The robustness of the reaction conditions, specifically the tolerance to air and moisture, reduces the risk of batch failures due to environmental excursions, ensuring consistent delivery schedules to downstream customers. This reliability is crucial for maintaining the continuity of supply for critical applications in the display and lighting industries, where production downtime can have cascading effects on the entire value chain.
- Scalability and Environmental Compliance: The mild reaction conditions and the use of less hazardous reagents align perfectly with modern green chemistry principles, facilitating easier regulatory approval and environmental compliance. The process generates minimal heavy metal waste, simplifying effluent treatment and reducing the environmental footprint of the manufacturing site. This sustainability profile is increasingly important for meeting corporate ESG goals and satisfying the stringent vendor audits conducted by major multinational corporations in the electronics and pharmaceutical sectors.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and scope of this patented synthesis method, providing clarity for technical teams evaluating its adoption. These answers are derived directly from the experimental data and embodiments described in the patent documentation, ensuring accuracy and relevance for process optimization discussions.
Q: What catalysts are used in this novel carbazole synthesis?
A: The process utilizes inexpensive iodine-containing compounds such as elemental iodine, iodine chloride, iodine bromide, or mixtures thereof, eliminating the need for precious transition metals like palladium or ruthenium.
Q: Does the reaction require an inert atmosphere?
A: No, a key advantage of this method is that it proceeds efficiently under a standard air atmosphere, removing the operational complexity and cost associated with inert gas protection.
Q: What are the typical starting materials for this transformation?
A: The synthesis couples readily available 2-arylindole derivatives with aromatic alkynes or aromatic ketones, allowing for broad structural diversity in the final carbazole products.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Polysubstituted 6-Arylbenzo[a]Carbazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this iodine-catalyzed technology for producing next-generation optoelectronic materials and pharmaceutical intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from lab-scale discovery to industrial manufacturing is seamless and efficient. Our state-of-the-art facilities are equipped to handle the specific solvent systems and thermal profiles required for this chemistry, while our rigorous QC labs enforce stringent purity specifications to guarantee that every batch meets the exacting standards of the global electronics industry.
We invite you to collaborate with our technical procurement team to explore how this cost-effective synthesis route can be tailored to your specific product portfolio. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic benefits of switching to this metal-free protocol. Contact us today to discuss your project requirements, obtain specific COA data for relevant derivatives, and receive comprehensive route feasibility assessments that will accelerate your time to market.
