Advancing Pharmaceutical Synthesis with Stable Copper Trifluoromethylselenide Reagents and Commercial Scale-Up
The landscape of organoselenium chemistry has long been dominated by the challenge of safely and efficiently introducing the trifluoromethylselenyl (-SeCF3) group into organic frameworks, a transformation of paramount importance in modern medicinal chemistry. Patent CN103193803A presents a groundbreaking advancement in this field by disclosing a novel method for synthesizing air-stable copper(I) trifluoromethylselenide reagents. This technology represents a significant departure from conventional methodologies that rely on hazardous gaseous precursors, offering a robust pathway for the production of high-purity fine chemical intermediates. By utilizing industrially inexpensive raw materials such as cuprous iodide and selenium powder, this invention not only mitigates severe safety risks associated with toxic gases but also establishes a definitive molecular structure for the resulting copper complexes, resolving long-standing ambiguities in the field. For R&D directors and procurement specialists alike, this development signals a shift towards more sustainable, cost-effective, and reliable sourcing strategies for critical fluorinated building blocks used in the development of next-generation agrochemicals and pharmaceuticals.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of metal trifluoromethylselenide reagents has been fraught with significant technical and safety hurdles that hindered their widespread industrial adoption. Traditional protocols frequently necessitated the use of bis(trifluoromethyl) diselenide (CF3SeSeCF3), a reagent that is not only highly toxic but also exists as a gas at room temperature, demanding complex and expensive containment systems to prevent environmental contamination and operator exposure. Furthermore, prior art often relied on precious metals such as gold, silver, or platinum to stabilize the reactive selenium species, driving up the raw material costs to prohibitive levels for large-scale manufacturing. Even when copper was employed, the resulting species were often ill-defined, lacking single-crystal structural characterization and relying solely on spectroscopic data like 19F NMR for identification. This lack of structural certainty introduced variability in reaction outcomes, complicating impurity profiling and regulatory filing processes for pharmaceutical clients who require absolute confidence in the identity and purity of their starting materials.
The Novel Approach
The methodology outlined in the patent data revolutionizes this process by replacing hazardous gaseous inputs with safe, solid, and liquid reagents that are readily available on the global chemical market. The core innovation lies in the direct reaction of cuprous iodide, red selenium powder, anhydrous potassium fluoride, and trifluoromethyltrimethylsilane in acetonitrile, facilitated by nitrogen or phosphine ligands. This approach eliminates the need for handling toxic CF3SeSeCF3 gas entirely, thereby simplifying the engineering controls required for production and significantly lowering the barrier to entry for commercial scale-up. Moreover, the process yields well-defined, crystalline copper complexes that are stable in air, a property that dramatically enhances their shelf-life and ease of transport compared to their unstable predecessors. This structural clarity, confirmed by single-crystal X-ray diffraction, provides the rigorous analytical validation necessary for high-stakes applications in drug substance manufacturing, ensuring that every batch meets stringent quality specifications.
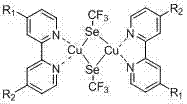
Mechanistic Insights into Copper-Catalyzed Trifluoromethylselenylation
The mechanistic foundation of this technology rests on the efficient formation of a Cu-Se bond through the activation of elemental selenium by fluoride ions in the presence of a trifluoromethyl source. The fluoride anion acts as a crucial activator, facilitating the nucleophilic attack on the silicon atom of trifluoromethyltrimethylsilane to generate a reactive trifluoromethyl anion equivalent in situ. This species subsequently reacts with the selenium powder and copper iodide to form the central [CuSeCF3] motif. The coordination environment is stabilized by bidentate ligands such as 2,2'-bipyridine or 1,10-phenanthroline, which chelate the copper center and prevent decomposition or disproportionation reactions that typically plague low-valent copper species. The resulting complex features a distorted geometry where the selenium atom bridges the copper center and the trifluoromethyl group, creating a stable reservoir of the -SeCF3 moiety that can be transferred to organic electrophiles with high fidelity.
From an impurity control perspective, the crystallization step described in the patent serves as a powerful purification mechanism that ensures the removal of unreacted starting materials and side products. By dissolving the crude residue in acetonitrile and slowly diffusing a ligand-containing ether solution at low temperatures (-25°C), the process selectively precipitates the desired copper complex while leaving soluble impurities in the mother liquor. This physical separation technique is far superior to chromatographic methods for bulk manufacturing, as it minimizes solvent waste and maximizes throughput. The definitive structural characterization provided by the single-crystal data allows quality control teams to establish precise acceptance criteria for bond lengths and angles, ensuring that the reagent supplied to downstream users possesses the exact electronic properties required for consistent catalytic performance in the trifluoromethylselenylation of aryl halides and heterocycles.
How to Synthesize Copper Trifluoromethylselenide Reagent Efficiently
The synthesis protocol detailed in the patent offers a straightforward yet highly effective route to producing these valuable reagents, balancing operational simplicity with high yield and purity. The process begins with the careful weighing and mixing of stoichiometric amounts of cuprous iodide, selenium powder, potassium fluoride, and the trifluoromethyl source in a sealed reactor equipped with a magnetic stirrer. Maintaining an inert nitrogen atmosphere is critical during the initial mixing phase to prevent oxidation of the sensitive copper(I) species before the complex is fully formed. Following the overnight stirring period at room temperature, the reaction mixture undergoes a series of workup steps including filtration through diatomaceous earth and vacuum solvent removal, which isolates the crude solid product. The final purification involves a recrystallization technique using a layered solvent system of acetonitrile and ether containing the specific ligand, which promotes the growth of high-quality crystals suitable for both immediate use and long-term storage.
- Combine cuprous iodide, red selenium powder, anhydrous potassium fluoride, and trifluoromethyltrimethylsilane in acetonitrile within a sealed reactor under nitrogen protection.
- Stir the mixture vigorously at room temperature overnight to facilitate the formation of the copper-selenium bond and subsequent complexation.
- Filter the reaction mixture, remove solvents under vacuum, wash residues with n-hexane, and recrystallize the product using a ligand ether solution at -25°C.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition to this novel synthesis route offers profound strategic benefits that extend far beyond simple unit cost savings. The elimination of toxic gaseous reagents fundamentally alters the risk profile of the supply chain, removing the need for specialized gas cylinders, leak detection systems, and emergency scrubbing infrastructure that typically inflate capital expenditure and operational overhead. By shifting to solid and liquid raw materials like selenium powder and trifluoromethyltrimethylsilane, manufacturers can leverage existing standard chemical handling protocols, thereby reducing lead times for raw material acquisition and minimizing the risk of supply disruptions caused by hazardous material transport regulations. This simplification of the input logistics directly translates to enhanced supply chain reliability, ensuring that critical intermediates are available exactly when needed for downstream drug synthesis campaigns without the delays associated with managing controlled substances.
- Cost Reduction in Manufacturing: The economic impact of substituting precious metals like gold or silver with abundant copper cannot be overstated, as it drastically lowers the raw material cost base for the reagent. Furthermore, the use of inexpensive industrial-grade potassium fluoride and selenium powder, combined with a high-yield crystallization process that avoids costly chromatographic purification, results in substantial cost savings per kilogram of finished product. The avoidance of expensive gas handling equipment and the reduction in waste disposal costs associated with toxic byproducts further contribute to a leaner, more competitive cost structure that allows for better pricing flexibility in the global market for fine chemical intermediates.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals that are widely produced and stocked by multiple global suppliers mitigates the risk of single-source dependency, a critical factor for maintaining business continuity in pharmaceutical manufacturing. Since the reagents involved are solids or liquids with high boiling points, they are significantly easier and safer to ship internationally compared to pressurized toxic gases, reducing the likelihood of customs delays or shipping refusals. This logistical robustness ensures a steady flow of materials into the production facility, allowing supply chain planners to optimize inventory levels and reduce the safety stock required to buffer against potential disruptions, ultimately improving working capital efficiency.
- Scalability and Environmental Compliance: The ambient temperature conditions and simple stirring requirements of this reaction make it inherently scalable from gram-scale laboratory synthesis to multi-ton commercial production without the need for complex thermal management systems. The process generates minimal hazardous waste, as the byproducts are primarily inorganic salts that can be treated using standard effluent protocols, aligning perfectly with increasingly stringent environmental regulations and corporate sustainability goals. This green chemistry profile not only reduces the environmental footprint of the manufacturing site but also simplifies the permitting process for capacity expansion, enabling rapid response to surging market demand for fluorinated and selenated building blocks.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and application of this copper trifluoromethylselenide technology. These insights are derived directly from the experimental data and structural analysis provided in the patent documentation, offering clarity on performance metrics and operational parameters. Understanding these details is essential for technical teams evaluating the feasibility of integrating this reagent into their existing synthetic routes for API intermediates or advanced material precursors.
Q: What are the safety advantages of this new CuSeCF3 synthesis method compared to traditional routes?
A: Traditional methods often require the handling of highly toxic and gaseous bis(trifluoromethyl) diselenide (CF3SeSeCF3), which poses significant operational hazards and requires specialized gas handling infrastructure. The patented method utilizes solid selenium powder and liquid trifluoromethyltrimethylsilane, drastically reducing toxicity risks and simplifying the operational requirements for industrial scale-up.
Q: How does the stability of this reagent impact its application in drug discovery?
A: Unlike previous copper-selenium species that were often unstable or only characterized by NMR without definite structures, this reagent forms air-stable crystalline complexes. This stability allows for easier storage, transportation, and handling during the trifluoromethylselenylation of aryl halides, ensuring consistent reaction performance in the synthesis of bioactive pharmaceutical intermediates.
Q: Can this reagent be used for substrates other than simple aryl iodides?
A: Yes, the patent demonstrates high efficiency across a broad range of substrates including aryl bromides, heterocyclic compounds, and functionalized aromatics containing groups like cyano, nitro, and ester moieties. The isolated yields for these diverse substrates consistently range from high 70s to nearly 99%, indicating robust versatility for complex molecule synthesis.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Copper Trifluoromethylselenide Reagent Supplier
As the demand for fluorinated and selenated motifs continues to surge in the development of high-performance pharmaceuticals and agrochemicals, securing a partner with deep technical expertise and robust manufacturing capabilities is essential. NINGBO INNO PHARMCHEM stands at the forefront of this sector, leveraging extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production to deliver consistent quality at any volume. Our state-of-the-art facilities are equipped with rigorous QC labs and advanced analytical instrumentation to ensure that every batch of reagent meets stringent purity specifications, providing the reliability that R&D and production teams demand for critical synthesis steps. We understand that the introduction of new reagents requires validation, and our team is prepared to support your technical due diligence with comprehensive data packages and sample availability.
We invite you to engage with our technical procurement team to discuss how this innovative copper-catalyzed technology can be integrated into your supply chain to drive efficiency and reduce costs. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic benefits specific to your production volume and application needs. We encourage potential partners to contact us directly to obtain specific COA data and route feasibility assessments, ensuring that you have all the necessary information to make informed decisions about adopting this next-generation trifluoromethylselenylation platform for your upcoming projects.
