Advanced Palladium-Catalyzed Dehydrogenation Coupling for Commercial Enamide Production
Advanced Palladium-Catalyzed Dehydrogenation Coupling for Commercial Enamide Production
The landscape of fine chemical synthesis is constantly evolving, driven by the need for more efficient, sustainable, and cost-effective methodologies. A significant breakthrough in this domain is detailed in patent CN114014802A, which introduces a novel method for synthesizing enamide compounds through palladium-catalyzed dehydrogenation coupling. This technology represents a paradigm shift for manufacturers of pharmaceutical intermediates, offering a robust alternative to legacy synthetic routes that have long plagued the industry with low yields and harsh reaction conditions. By leveraging the unique reactivity of 2-pyridone compounds and acrylic acid derivatives under the influence of a palladium catalyst and oxidant, this process achieves high product yields while maintaining mild operational parameters. For R&D directors and procurement managers alike, understanding the nuances of this patent is crucial, as it unlocks new possibilities for producing high-purity OLED materials, agrochemical intermediates, and bioactive molecules with greater economic efficiency.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of enamides from 2-pyridone scaffolds has been fraught with significant technical and economic challenges that hinder large-scale production. The traditional reliance on nucleophilic substitution reactions with halogenated olefins necessitates the use of strongly basic conditions, which often leads to substrate decomposition and narrow applicability across different functional groups. Furthermore, the requirement to pre-synthesize halogenated olefins adds extra steps to the supply chain, increasing both the lead time and the overall cost of goods sold. Another conventional pathway involves addition reactions with propiolic acid derivatives under alkaline conditions; however, these starting materials are often expensive and suffer from limited commercial availability. These legacy methods not only struggle with low atom economy but also generate substantial waste streams, creating environmental compliance burdens for modern chemical facilities seeking to reduce their carbon footprint.
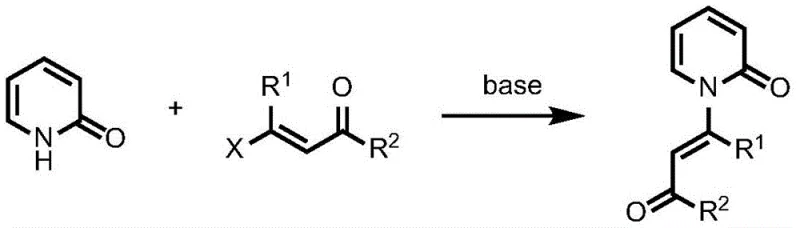
The Novel Approach
In stark contrast to these outdated techniques, the innovative strategy outlined in the patent utilizes a direct dehydrogenation coupling mechanism that bypasses the need for pre-functionalized substrates. By employing palladium acetate as a catalyst in conjunction with a silver-based oxidant, the reaction directly couples 2-pyridones with widely available acrylic acid derivatives. This approach eliminates the necessity for strong bases and allows the reaction to proceed under significantly milder thermal conditions, typically ranging from 40 to 100 degrees Celsius. The versatility of this method is demonstrated by its tolerance for a wide array of substituents on both the pyridone ring and the acrylate moiety, including alkyl, alkoxy, nitro, and halogen groups. This broad substrate scope ensures that manufacturers can produce a diverse library of enamide derivatives without needing to redesign the core process for each new target molecule, thereby streamlining the development pipeline for new drug candidates.
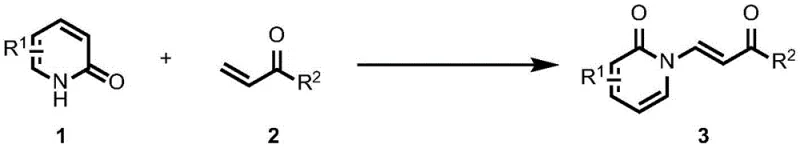
Mechanistic Insights into Pd-Catalyzed Dehydrogenation Coupling
The core of this technological advancement lies in the sophisticated catalytic cycle mediated by the palladium species. The reaction initiates with the coordination of the palladium catalyst to the 2-pyridone substrate, facilitating a C-H activation step that generates a reactive organopalladium intermediate. This step is critical as it avoids the need for pre-installed leaving groups, which is a major bottleneck in classical cross-coupling reactions. Subsequently, the acrylic acid derivative undergoes insertion into the palladium-carbon bond, followed by a beta-hydride elimination that releases the desired enamide product and generates a palladium-hydride species. To sustain the catalytic turnover, the oxidant plays an indispensable role by re-oxidizing the reduced palladium species back to its active state, thus closing the catalytic loop. This efficient regeneration mechanism allows for the use of catalytic amounts of palladium, minimizing the residual metal content in the final product, which is a stringent requirement for pharmaceutical grade intermediates.
From an impurity control perspective, the mild nature of this oxidative coupling significantly reduces the formation of side products commonly associated with harsh alkaline treatments. Traditional base-mediated reactions often promote polymerization of the acrylate or degradation of sensitive functional groups on the pyridone ring, leading to complex impurity profiles that are difficult to purge. In this Pd-catalyzed system, the selectivity is governed by the electronic and steric properties of the ligand environment around the metal center, favoring the formation of the thermodynamic enamide product. The use of silver salts as oxidants further aids in scavenging halide ions that might otherwise poison the catalyst, ensuring consistent reaction performance batch after batch. This high level of chemoselectivity translates directly to simplified downstream processing, as fewer purification steps are required to meet the rigorous purity specifications demanded by global regulatory bodies.
How to Synthesize Enamides Efficiently
Implementing this synthesis route in a laboratory or pilot plant setting requires careful attention to reagent stoichiometry and reaction monitoring to maximize yield. The general protocol involves dissolving the 2-pyridone compound and the acrylic acid derivative in a suitable organic solvent such as 1,2-dichloroethane, toluene, or chlorobenzene. Once the substrates are in solution, the palladium acetate catalyst and the silver oxidant are introduced, and the mixture is heated with stirring. The reaction progress is typically monitored via TLC or HPLC until the starting material is consumed, usually within a 24-hour window at temperatures around 60 degrees Celsius. Following the reaction, the mixture is filtered through kieselguhr to remove metal residues, concentrated, and the crude product is purified by column chromatography to isolate the pure enamide.
- Dissolve the 2-pyridone compound and acrylic acid derivative in an organic solvent such as 1,2-dichloroethane or toluene.
- Add palladium acetate catalyst and a silver-based oxidant (e.g., silver trifluoroacetate) to the reaction mixture.
- Heat the mixture to 40-100°C with stirring for approximately 24 hours, then filter and purify via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this palladium-catalyzed methodology offers tangible benefits that extend far beyond simple yield improvements. The primary driver for cost reduction is the elimination of expensive and specialized starting materials; acrylic acid derivatives are commodity chemicals produced on a massive scale, ensuring a stable and competitive pricing structure compared to proprietary halogenated olefins. Additionally, the removal of strong base requirements simplifies the reactor infrastructure needed for production, as there is no need for specialized corrosion-resistant lining or extensive neutralization waste treatment systems. This simplification of the process workflow drastically reduces the operational expenditure associated with utility consumption and waste disposal, allowing for a more lean manufacturing model. The ability to source raw materials from multiple global suppliers further mitigates supply chain risks, ensuring continuity of supply even during market fluctuations.
- Cost Reduction in Manufacturing: The economic impact of switching to this dehydrogenation coupling route is profound, primarily due to the streamlined material inputs and reduced processing steps. By avoiding the synthesis of halogenated precursors, manufacturers save on the reagents, solvents, and labor hours previously dedicated to those upstream processes. The catalytic nature of the palladium system means that expensive metal is used in minimal quantities, and the efficient oxidant usage ensures high atom economy. Furthermore, the mild reaction conditions lower energy consumption for heating and cooling, contributing to a smaller overall carbon footprint and reduced utility bills. These cumulative savings allow companies to offer more competitive pricing for high-purity pharmaceutical intermediates while maintaining healthy profit margins.
- Enhanced Supply Chain Reliability: Supply chain resilience is significantly bolstered by the use of universally available feedstocks like 2-pyridones and acrylates, which are not subject to the same geopolitical or logistical bottlenecks as specialized fine chemicals. The robustness of the reaction conditions means that production can be scaled up or down rapidly in response to market demand without requiring extensive re-validation of the process. Since the method tolerates a wide range of substituents, a single production line can be adapted to manufacture various derivatives with minimal changeover time. This flexibility is invaluable for contract manufacturing organizations that need to pivot quickly between different client projects, ensuring that lead times for high-purity intermediates remain short and predictable.
- Scalability and Environmental Compliance: Scaling this process from gram to ton scale is facilitated by the absence of hazardous reagents and the stability of the catalytic system under standard operating pressures. The reduction in waste generation, particularly the avoidance of halogenated byproducts and strong alkaline effluents, simplifies environmental compliance and lowers the cost of waste treatment. The process aligns well with green chemistry principles by maximizing resource efficiency and minimizing the use of auxiliary substances. For facilities aiming to achieve ISO 14001 certification or meet stricter local environmental regulations, this technology provides a clear pathway to sustainable operations. The ease of purification also reduces the volume of organic solvents required for chromatography or crystallization, further enhancing the environmental profile of the manufacturing site.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis method. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing a reliable foundation for decision-making. Understanding these details helps stakeholders evaluate the feasibility of integrating this technology into their existing production portfolios.
Q: What are the primary advantages of this Pd-catalyzed method over traditional nucleophilic substitution?
A: Unlike traditional methods requiring strong alkaline conditions and pre-functionalized halogenated olefins, this Pd-catalyzed dehydrogenation coupling utilizes readily available acrylic acid derivatives under mild conditions, significantly improving atom economy and substrate applicability.
Q: Which oxidants are compatible with this palladium catalytic system?
A: The patent specifies several effective silver-based oxidants including silver nitrate, silver trifluoroacetate, silver acetate, silver carbonate, and silver phosphate, which facilitate the catalytic cycle regeneration.
Q: Is this synthesis method scalable for industrial production?
A: Yes, the method employs cheap and easily obtained raw materials and operates under mild heating conditions (40-100°C), making it highly suitable for commercial scale-up of complex pharmaceutical intermediates without requiring extreme pressure or temperature.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Enamide Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced catalytic technologies like the Pd-catalyzed dehydrogenation coupling described in CN114014802A. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive consistent quality regardless of order volume. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of enamide intermediate performs reliably in downstream synthesis. We understand that in the fast-paced world of drug development, time is of the essence, and our team is dedicated to accelerating your timeline from bench to market through optimized process engineering.
We invite you to collaborate with us to leverage this cutting-edge synthesis route for your next project. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and target specifications. By partnering with us, you gain access to specific COA data and route feasibility assessments that demonstrate the viability of this method for your unique application. Contact us today to discuss how we can support your supply chain with high-quality, cost-effective enamide solutions that drive innovation in the pharmaceutical and fine chemical industries.
