Advanced Manganese Catalyst Technology for Efficient Pharmaceutical Intermediate Synthesis
Advanced Manganese Catalyst Technology for Efficient Pharmaceutical Intermediate Synthesis
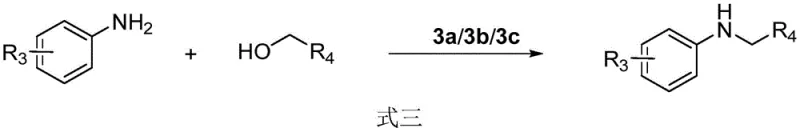
The landscape of organic synthesis is undergoing a significant transformation driven by the urgent need for sustainable and cost-effective manufacturing processes. A pivotal development in this field is detailed in patent CN109046463B, which discloses a novel class of bidentate azacarbene manganese catalysts. This technology represents a paradigm shift away from traditional noble metal catalysis, offering a robust solution for constructing critical carbon-nitrogen and carbon-carbon bonds. For R&D directors and process chemists, this innovation provides a viable pathway to synthesize complex pharmaceutical intermediates under remarkably mild conditions. The catalyst's unique structural design eliminates the need for toxic phosphorus ligands, addressing both environmental compliance and operator safety concerns simultaneously. By leveraging earth-abundant manganese, this methodology not only reduces reliance on volatile supply chains associated with ruthenium and iridium but also enhances the overall atom economy of alkylation reactions.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, alkylation reactions have relied heavily on the use of organohalogen reagents coupled with strong bases, a approach that generates substantial amounts of stoichiometric salt waste and poses significant environmental hazards. Furthermore, when transition metal catalysis is employed to improve selectivity, the industry has predominantly depended on precious metals such as ruthenium and iridium. These noble metal catalysts are not only prohibitively expensive due to their scarcity but often require harsh reaction conditions and specialized ligands that complicate downstream purification. The reliance on aldehyde reactants in traditional hydrogen borrowing strategies also introduces stability issues, as aldehydes are prone to oxidation and polymerization, leading to inconsistent batch quality. Consequently, manufacturers face escalating costs related to raw material procurement, waste disposal, and the rigorous removal of trace heavy metals to meet stringent pharmaceutical purity standards.
The Novel Approach
The technology described in the patent introduces a groundbreaking alternative by utilizing simple, renewable alcohols as direct alkylating agents in a hydrogen borrowing mechanism. This approach fundamentally alters the reaction profile by generating water as the sole byproduct, thereby drastically simplifying waste management protocols. The core of this innovation lies in the specific bidentate azacarbene manganese complex, which exhibits superior activity and stability compared to previous non-noble metal attempts. Unlike earlier manganese catalysts that required phosphorus-based ligands, this new structure is phosphorus-free, enhancing its environmental profile and reducing potential contamination risks in the final API. The reaction proceeds efficiently at moderate temperatures ranging from 30°C to 100°C, allowing for the functionalization of sensitive substrates that would otherwise degrade under harsher thermal conditions. This versatility makes it an ideal candidate for the late-stage functionalization of complex drug molecules.
Mechanistic Insights into Bidentate Azacarbene Manganese Catalysis
The efficacy of this catalytic system stems from the precise electronic and steric environment created by the bidentate azacarbene ligand surrounding the manganese center. As illustrated in the structural formula, the ligand framework stabilizes the manganese atom in a low oxidation state, facilitating the crucial dehydrogenation of the alcohol substrate to form an aldehyde or ketone intermediate in situ. This transient carbonyl species then undergoes condensation with the nucleophile, whether it be an amine or an active methylene compound, to form an imine or enone intermediate. The manganese hydride species generated in the initial dehydrogenation step subsequently reduces this unsaturated intermediate, completing the catalytic cycle and releasing the alkylated product. This seamless integration of dehydrogenation and hydrogenation steps within a single metal center ensures high atom efficiency and minimizes the accumulation of reactive intermediates that could lead to side reactions.
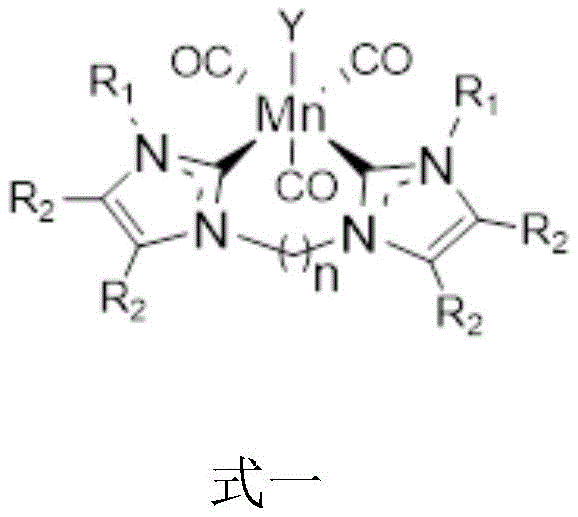
From an impurity control perspective, the robustness of the azacarbene ligand prevents the leaching of manganese ions into the reaction mixture, a common issue with less stable complexes. The absence of phosphorus ligands further simplifies the impurity profile, as there are no phosphine oxides or degraded phosphorus species that require difficult chromatographic removal. The catalyst demonstrates remarkable tolerance to various functional groups, including electron-donating and electron-withdrawing substituents on the aromatic rings of the substrates. This broad substrate scope implies that the electronic properties of the ligand can be finely tuned by modifying the R1 and R2 groups, allowing process chemists to optimize the catalyst for specific steric demands. Such tunability is critical for scaling up reactions where minor changes in substrate electronics can drastically affect yield and selectivity in conventional systems.
How to Synthesize Bidentate Azacarbene Manganese Catalyst Efficiently
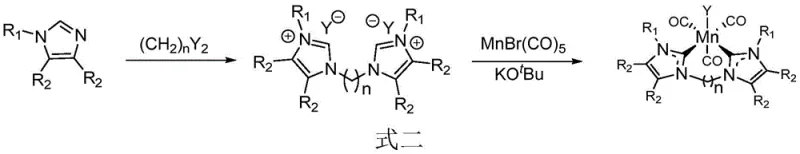
The preparation of this high-performance catalyst is designed for operational simplicity, utilizing readily available starting materials and standard laboratory techniques. The synthesis begins with the formation of the bidentate azaimidazolium salt ligand, followed by metallation with manganese pentacarbonyl bromide in the presence of a base. This two-step sequence avoids the need for exotic reagents or extreme pressure conditions, making it highly accessible for pilot plant production. The resulting catalyst is a stable solid that can be handled in air for short periods, although storage under inert atmosphere is recommended to maintain maximum activity over long durations. For detailed procedural specifics regarding stoichiometry and workup, please refer to the standardized synthesis guide below.
- Prepare the bidentate azaimidazolium salt ligand by reacting imidazole derivatives with alkyl halides in tetrahydrofuran at elevated temperatures.
- Combine the ligand with manganese pentacarbonyl bromide and potassium tert-butoxide in an inert solvent under nitrogen protection.
- Stir the mixture at moderate temperatures (25-100°C) for 10-24 hours, then isolate the solid catalyst via washing and drying.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this manganese-based technology offers compelling strategic advantages that extend beyond mere technical performance. The shift from noble metals to manganese represents a fundamental decoupling from the volatile pricing and geopolitical supply risks associated with platinum group metals. By utilizing alcohols instead of halides, manufacturers can source raw materials from the bulk chemical sector, where prices are stable and supply is continuous, rather than relying on specialized fine chemical suppliers for activated alkylating agents. This transition significantly lowers the barrier to entry for producing high-value intermediates, enabling more competitive pricing structures for downstream API manufacturers. Furthermore, the simplified waste stream, consisting primarily of water and benign salts, reduces the burden on environmental health and safety departments, leading to lower operational expenditures related to waste treatment and regulatory compliance.
- Cost Reduction in Manufacturing: The elimination of expensive ruthenium or iridium catalysts results in a drastic reduction in direct material costs, as manganese is orders of magnitude cheaper and more abundant. Additionally, the use of alcohols as alkylating agents removes the need for costly activation steps and the disposal of halogenated waste, further driving down the total cost of goods sold. The mild reaction conditions also translate to lower energy consumption, as there is no need for high-temperature reflux or cryogenic cooling, optimizing the utility usage profile of the manufacturing facility.
- Enhanced Supply Chain Reliability: Sourcing manganese salts and simple imidazole derivatives is far more reliable than securing high-purity noble metal complexes, which often have long lead times and limited supplier bases. The robustness of the catalyst allows for longer shelf life and easier logistics, reducing the risk of production delays caused by reagent degradation. This stability ensures consistent batch-to-batch quality, which is paramount for maintaining validation status in regulated pharmaceutical manufacturing environments.
- Scalability and Environmental Compliance: The reaction's tolerance to air and moisture during setup, combined with the absence of toxic phosphorus ligands, simplifies the scale-up process from gram to ton scale. The green chemistry metrics of this process, particularly the high atom economy and water byproduct, align perfectly with modern sustainability goals and corporate ESG mandates. This alignment facilitates smoother regulatory approvals and enhances the marketability of the final pharmaceutical products as being manufactured via eco-friendly processes.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this manganese catalytic system in industrial settings. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing a factual basis for feasibility assessments. Understanding these nuances is essential for project managers evaluating the integration of this technology into existing production lines.
Q: What are the primary advantages of this manganese catalyst over traditional ruthenium or iridium catalysts?
A: The primary advantages include significantly lower cost due to the abundance of manganese compared to noble metals, reduced toxicity, and the elimination of phosphorus ligands which are environmentally hazardous. Additionally, it operates under milder conditions.
Q: Can this catalyst be used for both C-N and C-C bond formation?
A: Yes, the patent explicitly demonstrates the catalyst's versatility in catalyzing the alkylation of amines to form C-N bonds and the alkylation of ketones to form C-C bonds using alcohols as the alkylating agents.
Q: What represents the main cost-saving driver in this synthetic methodology?
A: The main cost drivers are the replacement of expensive organohalogen reagents with cheap, renewable alcohols, and the substitution of precious metal catalysts with earth-abundant manganese, which simplifies waste treatment and raw material procurement.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Manganese Catalyst Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this bidentate azacarbene manganese technology for the next generation of pharmaceutical synthesis. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from lab bench to manufacturing plant is seamless and efficient. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of catalyst or intermediate meets the highest international standards. We are committed to supporting your R&D efforts by providing custom synthesis services that leverage this cost-effective and environmentally friendly chemistry.
We invite you to engage with our technical procurement team to discuss how this innovative manganese catalyst can optimize your specific synthetic routes. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic benefits tailored to your volume requirements. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that drive value and sustainability in your supply chain.
