Advanced Pd-Catalyzed Synthesis of Chiral Lignan Intermediates for Commercial Scale-Up
The pharmaceutical and fine chemical industries are constantly seeking more efficient pathways to access bioactive natural products, particularly lignans, which exhibit potent antitumor, antioxidant, and anti-inflammatory properties. Patent CN112110933A introduces a groundbreaking methodology for the asymmetric total synthesis of lignan natural products and their critical intermediates. This technology addresses the longstanding challenges associated with constructing complex chiral tetrahydrofuran rings containing three stereocenters. By leveraging a sophisticated palladium-catalyzed allylic cycloaddition strategy, the invention enables the conversion of readily available commercial raw materials into high-value chiral intermediates with exceptional stereochemical fidelity. For R&D Directors and Procurement Managers, this represents a pivotal shift from laborious chiral pool strategies to a more convergent and catalytic approach. The ability to generate optical purity through catalysis rather than resolution or stoichiometric chiral auxiliaries fundamentally alters the economic and technical feasibility of producing these complex molecules at scale.
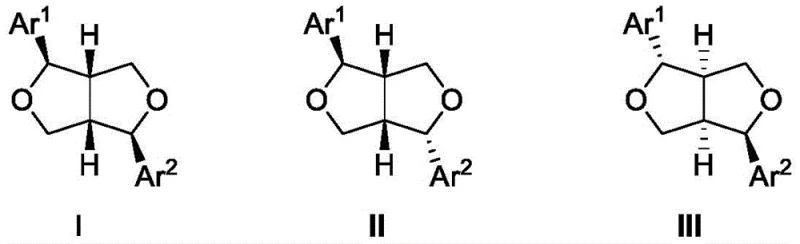
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the asymmetric total synthesis of lignan natural products has been hindered by reliance on chiral pool starting materials or the use of stoichiometric chiral auxiliaries, which inherently limit scalability and increase waste. Conventional routes often necessitate lengthy synthetic sequences, sometimes exceeding nine steps, to establish the requisite stereochemistry, leading to cumulative yield losses and increased operational costs. Furthermore, methods dependent on specific natural chiral sources face supply chain vulnerabilities and batch-to-batch variability issues that complicate regulatory compliance for pharmaceutical intermediates. The use of harsh reaction conditions in older methodologies can also degrade sensitive functional groups, requiring additional protection and deprotection steps that further elongate the process timeline. These inefficiencies create significant bottlenecks for supply chain heads who require consistent, high-volume delivery of complex intermediates without the risk of prolonged lead times or unpredictable purification challenges associated with multi-step linear syntheses.
The Novel Approach
The innovative method disclosed in the patent overcomes these barriers by employing a palladium-catalyzed asymmetric allylic cycloaddition as the key stereochemistry-setting step. This approach utilizes vinyl ethylene carbonate and substituted 2-nitroacrylates to construct the chiral tetrahydrofuran core in a highly convergent manner. By generating a palladium complex in situ with a chiral ligand, the reaction proceeds under mild conditions, typically between 0°C and 60°C, avoiding the thermal stress that compromises product integrity in traditional methods. The strategic use of commercially accessible reactants eliminates the dependency on scarce chiral pool materials, thereby stabilizing the supply chain and reducing raw material procurement risks. This streamlined pathway not only shortens the overall number of reaction steps but also enhances the atom economy of the process, aligning with modern green chemistry principles that are increasingly demanded by global regulatory bodies and corporate sustainability goals.

Mechanistic Insights into Pd-Catalyzed Allylic Cycloaddition
The core of this technological advancement lies in the precise mechanistic orchestration of the palladium-catalyzed cycle, which dictates the stereochemical outcome of the intermediate formation. The reaction initiates with the coordination of a palladium source, such as Pd2(dba)3·CHCl3, with a specialized chiral phosphine ligand to form an active catalytic species. This complex facilitates the ionization of vinyl ethylene carbonate to generate a π-allyl palladium intermediate, which is then subjected to nucleophilic attack by the 2-nitroacrylate derivative. The chirality of the ligand creates a sterically defined environment around the metal center, ensuring that the nucleophile approaches from a specific face, thus establishing the three stereocenters with high diastereoselectivity and enantioselectivity. The choice of ligand is critical; for instance, the use of (S)-SEGPHOS derivatives has been shown to optimize both yield and optical purity, demonstrating the tunability of the system for different substrate variations. This level of mechanistic control allows chemists to predictably access specific enantiomers required for biological activity, reducing the need for downstream chiral separation processes.

Impurity control is intrinsically built into this catalytic system through the high specificity of the transition state. Unlike non-catalytic methods that may produce racemic mixtures requiring difficult resolution, this pathway favors the formation of the desired stereoisomer directly, minimizing the generation of hard-to-remove diastereomeric impurities. The mild reaction temperatures, preferably maintained between 20°C and 25°C for optimal selectivity, prevent side reactions such as polymerization or decomposition of the sensitive nitro and ester functionalities. Furthermore, the compatibility of the catalyst system with a wide range of solvents, including tetrahydrofuran and dioxane, provides flexibility in process optimization to ensure maximum solubility and reaction kinetics. For quality assurance teams, this means a cleaner crude reaction profile, which simplifies purification workflows and ensures that the final intermediate meets stringent purity specifications required for subsequent pharmaceutical transformations without extensive recrystallization cycles.
How to Synthesize Chiral Tetrahydrofuran Intermediates Efficiently
The practical implementation of this synthesis route involves a carefully sequenced series of operations designed to maximize catalyst turnover and product isolation efficiency. The process begins with the preparation of the catalytic system under inert atmosphere to prevent oxidation of the palladium center, followed by the controlled addition of substrates to manage exothermicity. Detailed standard operating procedures dictate the precise molar ratios of palladium source, chiral ligand, and reactants to maintain the delicate balance required for high stereoselectivity. Following the cycloaddition, the workflow transitions into downstream processing steps involving reduction and oxidative cleavage to unlock the final lignan scaffold. The detailed standardized synthesis steps see the guide below for specific parameters regarding reagent grades and workup protocols.
- Perform palladium-catalyzed allylic cycloaddition between vinyl ethylene carbonate and 2-nitroacrylate using a chiral ligand to form the key tetrahydrofuran intermediate.
- Execute reduction and oxidative cleavage sequences on the intermediate to generate the necessary dialdehyde or diol precursors.
- Complete the total synthesis through intramolecular cyclization and Grignard addition to yield the final optically pure lignan natural products.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this patented technology offers substantial strategic advantages beyond mere technical novelty. The shift towards a catalytic asymmetric synthesis fundamentally reduces the dependency on expensive, stoichiometric chiral reagents that traditionally drive up the cost of goods sold for complex intermediates. By utilizing commercially available starting materials like vinyl ethylene carbonate, the process mitigates supply risks associated with proprietary or scarce chiral building blocks, ensuring a more resilient and continuous supply chain. The simplification of the synthetic route directly translates to reduced manufacturing lead times, allowing for faster response to market demands and more agile inventory management. Additionally, the mild reaction conditions lower energy consumption and reduce the burden on waste treatment facilities, contributing to a lower environmental footprint and potentially reduced regulatory compliance costs associated with hazardous waste disposal.
- Cost Reduction in Manufacturing: The elimination of stoichiometric chiral auxiliaries and the reduction in total synthetic steps significantly lower the raw material and operational expenses per kilogram of product. This efficiency gain allows for a more competitive pricing structure without compromising on the high purity standards required for pharmaceutical applications. The high catalytic activity ensures that expensive palladium metals are used in minimal quantities, further optimizing the cost basis of the manufacturing process. Consequently, this creates a sustainable economic model for producing high-value lignan intermediates that can withstand market price fluctuations.
- Enhanced Supply Chain Reliability: Sourcing strategies are greatly improved as the key reactants are commodity chemicals with established global supply networks, reducing the risk of single-source bottlenecks. The robustness of the catalytic system against minor variations in reaction parameters ensures consistent batch-to-batch quality, which is critical for maintaining long-term contracts with multinational pharmaceutical clients. This reliability minimizes the need for safety stock and allows for leaner inventory models, freeing up working capital for other strategic investments within the organization. The predictable nature of the synthesis also facilitates better production planning and capacity utilization across manufacturing sites.
- Scalability and Environmental Compliance: The process is inherently designed for scale-up, with reaction conditions that are easily manageable in large-scale reactors without requiring exotic equipment or extreme pressures. The use of common organic solvents and the avoidance of highly toxic reagents simplify the waste stream management, making it easier to meet increasingly strict environmental regulations in major manufacturing hubs. This environmental compatibility not only reduces disposal costs but also enhances the corporate social responsibility profile of the supply chain. The ability to scale from gram to ton quantities while maintaining stereochemical integrity ensures that the technology remains viable throughout the entire product lifecycle from clinical trials to commercial launch.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this asymmetric synthesis technology. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation to provide clarity on process capabilities. Understanding these aspects is crucial for technical teams evaluating the feasibility of integrating this route into existing manufacturing portfolios. The responses highlight the balance between high-performance chemistry and practical industrial application.
Q: How does this Pd-catalyzed method improve upon traditional lignan synthesis?
A: Traditional methods often rely on chiral pools or auxiliaries requiring lengthy synthetic steps. This novel approach utilizes asymmetric palladium catalysis to construct three stereocenters in a single key step, significantly shortening the reaction sequence and improving overall efficiency.
Q: What are the stereoselectivity capabilities of this process?
A: The process demonstrates exceptional stereocontrol, achieving high enantiomeric excess (ee) and diastereoselectivity (dr) through the precise selection of chiral phosphine ligands, such as SEGPHOS derivatives, ensuring the production of high-purity intermediates.
Q: Are the starting materials commercially viable for large-scale production?
A: Yes, the method utilizes commercially available vinyl ethylene carbonate and substituted 2-nitroacrylates as starting materials. This accessibility, combined with mild reaction conditions, supports robust commercial scale-up and supply chain stability.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Tetrahydrofuran Lignan Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of translating advanced academic research into robust commercial reality for our global partners. As a premier CDMO expert, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the intricate details of this palladium-catalyzed process are mastered at an industrial level. Our commitment to quality is underscored by our stringent purity specifications and rigorous QC labs, which guarantee that every batch of chiral tetrahydrofuran intermediate meets the exacting standards required for downstream pharmaceutical synthesis. We understand that consistency is key in the fine chemical industry, and our infrastructure is designed to deliver that reliability consistently.
We invite you to collaborate with us to leverage this cutting-edge technology for your specific project needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements and timeline constraints. Please contact us to request specific COA data and route feasibility assessments that demonstrate how we can optimize your supply chain for high-purity lignan intermediates. Together, we can accelerate the development of next-generation therapeutics by ensuring a secure and efficient supply of these vital building blocks.
