Scalable Production of Bioactive 2,4-Pyrrolidine-Diketones via Novel Cyclization Strategies
Scalable Production of Bioactive 2,4-Pyrrolidine-Diketones via Novel Cyclization Strategies
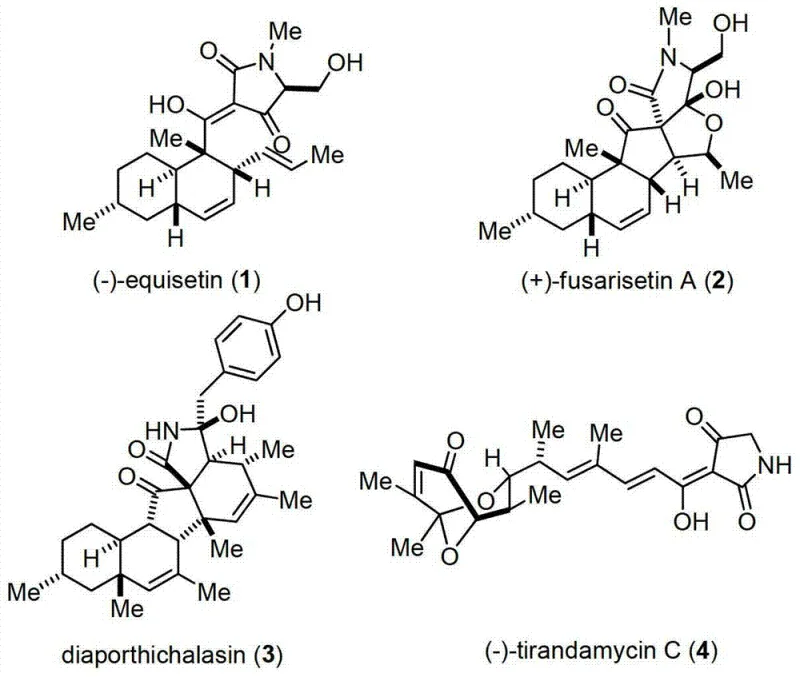
The pharmaceutical industry continuously seeks efficient pathways to access complex natural product scaffolds, particularly those exhibiting potent antitumor activities. Patent CN103483341A introduces a robust synthetic methodology for generating a novel class of 2,4-pyrrolidine-diketone compounds derived from the equisetin structural framework. These tetramic acid derivatives are renowned for their diverse biological profiles, including antibacterial, antiviral, and cytotoxic properties, making them highly valuable candidates for oncology drug discovery programs. The disclosed technology addresses long-standing challenges in the total synthesis of these molecules by offering a concise, eight-step route that begins with readily available chiral pool materials. By leveraging strategic bond disconnections, specifically focusing on the construction of the fused decalin-pyrrolidine core, this innovation provides a reliable pharmaceutical intermediate supplier with the tools necessary to produce high-purity scaffolds efficiently. The significance of this patent lies not only in the novelty of the compounds but also in the practical optimization of reaction conditions that facilitate smoother scale-up potential compared to historical precedents.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the total synthesis of equisetin and related tetramic acids has been plagued by excessive step counts and disappointing overall yields, creating significant bottlenecks for cost reduction in API manufacturing. For instance, the pioneering work by the Danishefsky group in 1989 required fifteen distinct synthetic steps to achieve a mere 3.8% overall yield, rendering the process economically unviable for large-scale production. Subsequent efforts by the Rodríguez and Shishido groups, while improving upon certain aspects, still necessitated thirteen to twenty-four steps, often involving cumbersome protection-deprotection sequences and difficult stereochemical introductions. These traditional routes frequently rely on late-stage functionalization strategies that suffer from poor atom economy and generate substantial chemical waste. Furthermore, the reliance on specialized reagents and harsh reaction conditions in earlier methodologies often complicates purification protocols, leading to impurity profiles that are challenging to manage under stringent GMP standards. Consequently, the supply chain for these high-value intermediates has remained fragile, with limited availability hindering broader preclinical and clinical evaluation of this promising chemical class.
The Novel Approach
In stark contrast to these laborious historical routes, the methodology outlined in CN103483341A employs a convergent strategy centered around a biomimetic intramolecular Diels-Alder reaction to rapidly construct the core bicyclic system. This novel approach streamlines the synthesis into just eight steps, drastically reducing the time and resources required to access the target 2,4-pyrrolidine-diketone skeleton. The process initiates from (-)-citronellal, a cheap and abundant chiral terpene, which is converted into a key polyene aldehyde intermediate through a series of high-yielding transformations including Wittig or Horner-Wadsworth-Emmons olefinations. The pivotal cyclization step utilizes boron trifluoride etherate as a Lewis acid catalyst under controlled low-temperature conditions, ensuring excellent stereocontrol without the need for auxiliary chiral catalysts. Following the ring closure, the pathway proceeds through a mild ammonolysis and a final Dieckmann condensation to install the characteristic tetramic acid moiety. This logical progression not only enhances the overall yield significantly but also simplifies the operational complexity, making it an ideal candidate for commercial scale-up of complex pharmaceutical intermediates.
Mechanistic Insights into Lewis Acid-Catalyzed Cyclization and Dieckmann Condensation
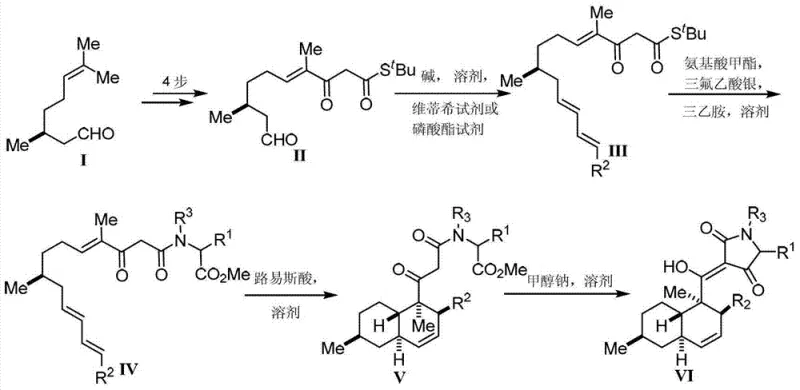
The success of this synthetic route hinges on the precise execution of the intramolecular Diels-Alder (IMDA) reaction, which serves as the cornerstone for establishing the molecular architecture. Mechanistically, the polyene precursor, activated by the coordination of boron trifluoride to the carbonyl oxygen, undergoes a concerted [4+2] cycloaddition. This transition state is highly organized, favoring the formation of the trans-fused decalin system observed in natural equisetin derivatives due to secondary orbital interactions and steric minimization. The use of dichloromethane as the solvent at temperatures ranging from -78°C to 0°C is critical; it suppresses competing polymerization pathways and ensures that the kinetic product is formed with high fidelity. Following the cyclization, the resulting thioester intermediate is subjected to ammonolysis using amino acid methyl esters in the presence of silver trifluoroacetate and triethylamine. This step effectively swaps the thioester leaving group for the nitrogen nucleophile, setting the stage for the final ring closure. The subsequent Dieckmann condensation, promoted by sodium methoxide in methanol, involves the deprotonation of the alpha-carbon adjacent to the amide carbonyl, followed by nucleophilic attack on the distal ketone ester. This intramolecular Claisen condensation closes the five-membered pyrrolidine-2,4-dione ring, completing the tetramic acid core with the retention of stereochemical integrity established in the earlier steps.
From an impurity control perspective, the robustness of this mechanism offers distinct advantages for maintaining high-purity standards required by regulatory bodies. The IMDA reaction is inherently selective, minimizing the formation of regioisomers that often plague intermolecular variants. Furthermore, the choice of silver trifluoroacetate for the aminolysis step is strategic; silver ions coordinate strongly with sulfur, driving the equilibrium towards the desired amide product and preventing the accumulation of unreacted thioester starting material. The final Dieckmann step is quenched carefully with hydrochloric acid to prevent retro-Claisen degradation, ensuring the stability of the sensitive enolized beta-diketone system. By understanding these mechanistic nuances, process chemists can fine-tune reaction parameters such as addition rates and stoichiometry to further suppress trace impurities. This level of control is essential for producing clinical-grade materials where impurity thresholds are exceptionally low, thereby reducing the burden on downstream purification processes like chromatography or recrystallization.
How to Synthesize Equisetin Derivatives Efficiently
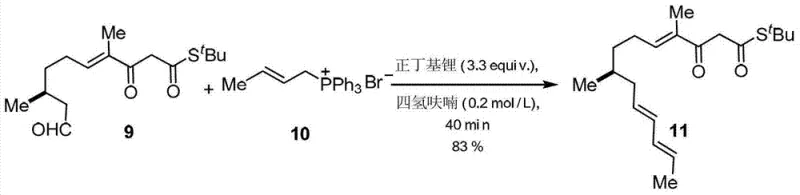
Implementing this synthesis requires careful attention to the preparation of the polyene precursor and the strict control of temperature during the cyclization phase to ensure reproducibility. The detailed standardized synthesis steps see the guide below for specific operational parameters regarding reagent grades and workup procedures.
- Preparation of polyene precursor III via Wittig or Horner-Wadsworth-Emmons reaction from aldehyde II.
- Intramolecular Diels-Alder cyclization using boron trifluoride etherate to form the decalin core structure.
- Ammonolysis with amino acid esters followed by Dieckmann condensation using sodium methoxide to close the pyrrolidine-2,4-dione ring.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented methodology represents a strategic opportunity to optimize the sourcing of complex anticancer intermediates. The shift from lengthy, low-yielding linear syntheses to this concise eight-step route fundamentally alters the cost structure of production. By eliminating nearly half the number of unit operations compared to legacy methods, the process inherently reduces labor costs, solvent consumption, and energy usage associated with heating and cooling cycles over extended periods. The reliance on (-)-citronellal as a starting material is particularly advantageous; as a bulk commodity chemical derived from citrus oils, it offers a stable and scalable supply base that is immune to the volatility often seen with specialized synthetic building blocks. This stability translates directly into enhanced supply chain reliability, allowing manufacturers to commit to longer-term delivery schedules with greater confidence. Moreover, the avoidance of exotic transition metal catalysts, which often require expensive removal steps to meet residual metal specifications, simplifies the purification train and reduces the environmental footprint of the manufacturing process.
- Cost Reduction in Manufacturing: The streamlined nature of this synthesis drives down the cost of goods sold (COGS) by maximizing material throughput and minimizing waste generation. The high yields reported in the key cyclization and condensation steps mean that less raw material is required to produce a kilogram of final product, directly impacting the bottom line. Additionally, the use of common laboratory reagents like boron trifluoride etherate and sodium methoxide eliminates the need for custom-synthesized catalysts, further lowering input costs. The simplified workup procedures, which often involve straightforward extractions and crystallizations rather than complex chromatographic separations, reduce the consumption of silica gel and eluents, contributing to substantial cost savings in consumables.
- Enhanced Supply Chain Reliability: Sourcing risk is significantly mitigated by the use of commercially available reagents that are produced by multiple global vendors. Unlike proprietary catalysts or unstable intermediates that might have single-source suppliers, the chemicals used in this route are standard inventory items for most fine chemical distributors. This redundancy ensures that production is not halted due to a shortage of a single critical component. Furthermore, the robustness of the reaction conditions allows for flexibility in manufacturing locations, enabling companies to diversify their production sites geographically to mitigate regional disruptions. The ability to produce these intermediates reliably supports continuous development pipelines, ensuring that clinical trials are not delayed due to material shortages.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing reaction types that are well-understood in kilo-lab and pilot plant environments. The exothermic nature of the cyclization is manageable with standard cooling infrastructure, and the solvent systems employed are compatible with large-scale recovery and recycling protocols. From an environmental standpoint, the improved atom economy and reduced step count lead to a lower E-factor (mass of waste per mass of product), aligning with modern green chemistry principles. This compliance with environmental standards facilitates easier permitting and reduces the costs associated with waste disposal, making the process sustainable for long-term commercial operation.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology, based on the specific data and embodiments provided in the patent documentation.
Q: What are the key advantages of this synthesis route over traditional methods?
A: Unlike previous 15-24 step routes with low overall yields (3.8%-10%), this patent describes an 8-step sequence with significantly improved operational simplicity and higher yields, utilizing commercially available starting materials like (-)-citronellal.
Q: How is stereochemical control achieved in the cyclization step?
A: The process utilizes a Lewis acid-catalyzed intramolecular Diels-Alder reaction at low temperatures (-78°C to 0°C), which ensures high stereoselectivity in forming the fused ring system critical for biological activity.
Q: Is this process suitable for large-scale manufacturing?
A: Yes, the route avoids exotic reagents and uses standard organic transformations like Wittig reactions and base-catalyzed condensations, making it highly amenable to scale-up for commercial API intermediate production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Equisetin Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this patented synthetic route for accelerating the development of next-generation anticancer therapeutics. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project can transition seamlessly from benchtop discovery to full-scale manufacturing. Our facilities are equipped with state-of-the-art reactors capable of handling the low-temperature conditions required for the Diels-Alder step, alongside rigorous QC labs that enforce stringent purity specifications for every batch released. We understand that consistency is key in pharmaceutical development, and our quality management systems are designed to deliver intermediates that meet the highest international regulatory standards.
We invite you to collaborate with us to leverage this efficient technology for your specific drug discovery programs. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating how this route can optimize your budget without compromising quality. Please contact our technical procurement team today to request specific COA data for our available tetramic acid intermediates and to discuss route feasibility assessments for your custom synthesis needs. Together, we can bring these promising bioactive compounds to the market faster and more economically.
