Advanced Catalytic Method for N-Arylation of Pyrimidine-2-Amines for Commercial Scale-Up
Advanced Catalytic Method for N-Arylation of Pyrimidine-2-Amines for Commercial Scale-Up
The pharmaceutical industry constantly seeks more efficient and cost-effective pathways for constructing critical nitrogen-containing heterocycles, particularly for antitumor agents like imatinib. Patent CN114315737B introduces a groundbreaking methodology utilizing an N,N-coordinated cuprous complex containing a meta-carborane ligand to catalyze the Buchwald-Hartwig coupling reaction. This innovation allows for the synthesis of N-arylated derivatives of pyrimidin-2-amine under remarkably mild conditions, specifically at room temperature, which represents a significant departure from energy-intensive traditional processes. The structural integrity and unique electronic properties of the meta-carborane ligand enhance the catalytic activity of the copper center, enabling high conversion rates without the need for expensive noble metals. 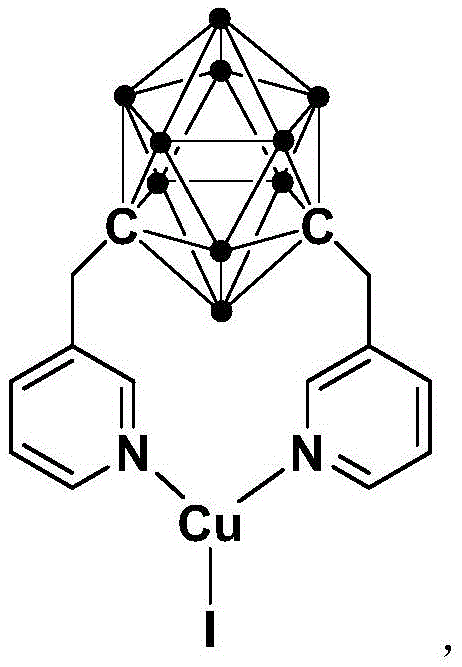
For procurement managers and supply chain directors, this technology offers a compelling value proposition by reducing dependency on volatile precious metal markets while simplifying process safety protocols. The ability to operate at ambient temperatures drastically lowers energy consumption and reduces the thermal stress on reactor equipment, thereby extending asset life and minimizing maintenance downtime. As a reliable pharmaceutical intermediate supplier, understanding these underlying technological shifts is crucial for securing long-term supply continuity and achieving substantial cost savings in API manufacturing. The robustness of this catalytic system ensures consistent quality output, which is paramount for meeting the stringent regulatory standards of global healthcare markets.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of C-N bonds in pyrimidine-2-amine derivatives has relied heavily on palladium-catalyzed Buchwald-Hartwig coupling reactions, which present several inherent logistical and economic challenges. Traditional protocols often necessitate the use of expensive noble metal catalysts and high loadings of specialized phosphine ligands, driving up the overall cost of goods sold significantly. Furthermore, many existing heterogeneous catalyst systems require harsh reaction conditions, such as heating to 110°C under an inert atmosphere for extended periods up to 24 hours, which increases energy expenditure and operational complexity. These severe conditions can also lead to the formation of unwanted byproducts and degradation of sensitive functional groups, complicating downstream purification and reducing overall yield. The low catalyst turnover numbers associated with older technologies mean that larger quantities of catalyst are required, generating more heavy metal waste that requires costly disposal and environmental remediation.
The Novel Approach
In stark contrast, the novel approach detailed in the patent utilizes a specially designed cuprous complex that achieves superior catalytic performance under ambient conditions. By employing an N,N-coordinated ligand system based on meta-carborane, the catalyst maintains high activity at room temperature, eliminating the need for external heating and inert gas protection. 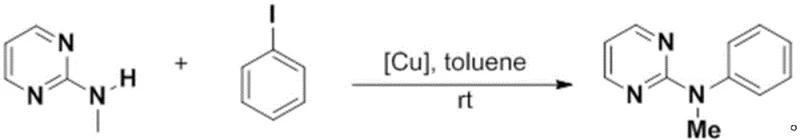
This method demonstrates exceptional versatility, successfully coupling N-methylpyrimidin-2-amine with a wide range of halogenated aromatics including iodobenzene, chlorobenzenes, and bromobenzenes with yields ranging from 85% to 96%. The mild reaction environment not only preserves the integrity of sensitive substrates but also significantly simplifies the workup procedure, allowing for direct concentration and column chromatography. For manufacturers seeking cost reduction in pharmaceutical intermediate manufacturing, this transition from palladium to copper represents a strategic move towards more sustainable and economically viable production models. The reduced reaction time of 6 to 10 hours further enhances throughput capacity, allowing facilities to produce more batches within the same operational window.
Mechanistic Insights into Cu-Catalyzed Buchwald-Hartwig Coupling
The efficacy of this system lies in the unique electronic and steric environment provided by the meta-carborane cage attached to the pyridine arms of the ligand. The carborane cluster is known for its three-dimensional aromaticity and electron-withdrawing characteristics, which stabilize the copper center and facilitate the oxidative addition and reductive elimination steps critical to the catalytic cycle. 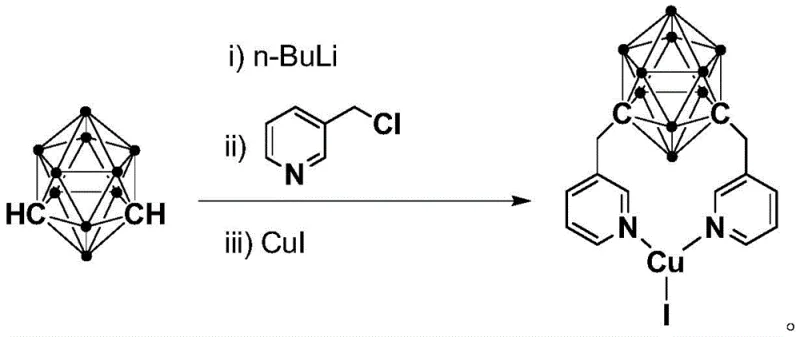
Unlike traditional ligands that may dissociate under thermal stress, this N,N-coordinated framework forms a robust chelate with the cuprous ion, preventing catalyst decomposition and ensuring a long catalytic lifetime. The mechanism likely proceeds through a standard Cu(I)/Cu(III) cycle where the amine coordinates to the metal center, followed by deprotonation and subsequent coupling with the aryl halide. The stability of the complex against air and moisture suggests that the coordination sphere is sufficiently saturated to prevent quenching by atmospheric oxygen or water molecules. This mechanistic robustness translates directly to process reliability, as variations in ambient humidity or minor leaks in the reactor setup are less likely to result in batch failure. For R&D directors, this implies a wider operating window and greater forgiveness in process parameters, reducing the risk of scale-up failures.
Impurity control is another critical aspect where this mechanism excels, as the mild conditions suppress side reactions such as homocoupling of the aryl halide or over-alkylation of the amine. The high selectivity observed across various substrates, including those with electron-withdrawing nitro groups or electron-donating methoxy groups, indicates a balanced electronic influence from the ligand. This balance ensures that the catalyst remains active regardless of the electronic nature of the substrate, providing a universal solution for diverse synthetic needs. The absence of heavy metal residues from noble metals simplifies the purification process, often removing the need for specialized scavenging resins to meet strict ppm limits for residual palladium. Consequently, the final API intermediate possesses a cleaner impurity profile, facilitating faster regulatory approval and reducing the burden on quality control laboratories.
How to Synthesize N-Arylated Pyrimidine-2-Amine Efficiently
The synthesis protocol outlined in the patent is designed for simplicity and reproducibility, making it highly suitable for transfer from laboratory bench to pilot plant operations. The process begins with the preparation of the catalyst itself, which involves a straightforward one-pot reaction of m-carborane with n-BuLi, followed by quenching with 3-chloromethylpyridine and complexation with CuI. Once the catalyst is secured, the coupling reaction is performed by simply dissolving the catalyst, the amine substrate, and the aryl halide in toluene at room temperature. Detailed standardized synthesis steps see the guide below.
- Dissolve the N,N-coordinated cuprous complex catalyst, N-methylpyrimidin-2-amine, and the selected halogenated aromatic compound in toluene solvent.
- Maintain the reaction mixture at room temperature and stir continuously for a duration of 6 to 10 hours to ensure complete coupling.
- Concentrate the reaction liquid and perform separation and purification via silica gel column chromatography to obtain the final N-arylation derivative.
Commercial Advantages for Procurement and Supply Chain Teams
Adopting this catalytic technology offers profound benefits for procurement strategies, primarily driven by the substitution of expensive palladium with abundant and inexpensive copper. The elimination of noble metals removes exposure to the volatile pricing fluctuations often seen in the precious metals market, allowing for more accurate long-term budget forecasting and cost stabilization. Additionally, the simplified reaction conditions reduce the demand for high-grade inert gases and energy-intensive heating systems, leading to lower utility costs per kilogram of product produced. These factors combine to create a significantly reduced cost base for the manufacturing of these critical pharmaceutical intermediates, enhancing overall margin potential for downstream drug producers. The operational simplicity also means that training requirements for plant operators are minimized, further contributing to indirect cost savings.
- Cost Reduction in Manufacturing: The shift from palladium to copper catalysts fundamentally alters the cost structure of the synthesis, removing the need for expensive metal recovery processes often required to meet regulatory limits. Since the catalyst loading is low (0.01 to 0.03 molar ratio relative to substrate), the absolute amount of metal used is minimal, yet the turnover number is high enough to drive the reaction to completion efficiently. The mild conditions also extend the lifespan of reactor vessels and seals, which would otherwise degrade under the high temperatures and pressures of traditional methods. Furthermore, the ease of product separation via simple column chromatography reduces solvent consumption and processing time, streamlining the entire production workflow. These cumulative effects result in substantial cost savings without compromising the quality or purity of the final active pharmaceutical ingredient.
- Enhanced Supply Chain Reliability: The insensitivity of the catalyst to air and water greatly simplifies logistics and storage requirements, as special handling procedures for moisture-sensitive reagents are no longer necessary. This robustness ensures that raw materials can be sourced from a broader range of suppliers without risking quality degradation during transit or storage. The shorter reaction times of 6 to 10 hours compared to 24 hours for conventional methods increase the throughput of existing manufacturing assets, allowing for faster fulfillment of customer orders. This increased agility enables supply chain managers to respond more quickly to market demands and reduces the need for large safety stocks of finished goods. Ultimately, this leads to a more resilient supply chain capable of withstanding disruptions and maintaining continuous delivery schedules.
- Scalability and Environmental Compliance: Scaling this reaction from gram to ton scale is facilitated by the exothermic nature being manageable at room temperature, removing the need for complex cooling systems to control runaway reactions. The reduction in waste generation, particularly heavy metal waste, aligns with increasingly stringent environmental regulations and corporate sustainability goals. The use of toluene as a solvent is well-established in industrial settings, and the ability to recover and recycle it further minimizes the environmental footprint of the process. The high atom economy and selectivity mean that less raw material is wasted as byproducts, maximizing the efficiency of resource utilization. These environmental advantages not only reduce disposal costs but also enhance the corporate social responsibility profile of the manufacturing entity.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this catalytic system in industrial settings. These answers are derived directly from the experimental data and technical specifications provided in the patent documentation to ensure accuracy and reliability. Understanding these details is essential for technical teams evaluating the feasibility of integrating this method into their existing production lines. The information covers catalyst stability, substrate scope, and operational parameters to provide a comprehensive overview.
Q: What are the primary advantages of this cuprous complex catalyst over traditional palladium systems?
A: The novel N,N-coordinated cuprous complex containing a meta-carborane ligand operates effectively at room temperature, eliminating the need for harsh heating conditions (often 110°C) required by conventional heterogeneous catalysts. Furthermore, it utilizes inexpensive copper instead of noble metals like palladium, significantly lowering raw material costs while maintaining high catalytic efficiency and yields between 85% and 96%.
Q: Is the catalyst sensitive to air or moisture during the reaction process?
A: No, the catalyst exhibits stable physical and chemical properties, including thermal stability, and is notably insensitive to both air and water. This robustness simplifies the operational requirements for industrial scale-up, as it removes the necessity for stringent inert atmosphere controls often mandated by sensitive organometallic catalysts.
Q: What types of halogenated aromatic substrates are compatible with this method?
A: The method demonstrates excellent universality and can catalyze substrates including iodobenzene, various chlorobenzenes (such as 4-methylchlorobenzene and 2-methylchlorobenzene), bromobenzenes (like 4-methoxybromobenzene and 4-nitrobromobenzene), and even bulky substrates like 2-bromonaphthalene, providing a versatile tool for drug synthesis.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Pyrimidine-2-Amine Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this copper-catalyzed technology for the production of high-value pharmaceutical intermediates. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab-scale discovery to full-scale manufacturing is seamless and efficient. We are committed to maintaining stringent purity specifications through our rigorous QC labs, guaranteeing that every batch meets the exacting standards required by global regulatory bodies. Our infrastructure is designed to handle complex chemistries safely and effectively, leveraging the latest advancements in process engineering to maximize yield and minimize waste. By partnering with us, you gain access to a supply chain that is both robust and adaptable to your specific project needs.
We invite you to contact our technical procurement team to discuss how this innovative catalytic method can be applied to your specific product portfolio. We are prepared to provide a Customized Cost-Saving Analysis that quantifies the potential economic benefits of switching to this copper-based system for your operations. Please reach out to request specific COA data for our reference standards and route feasibility assessments tailored to your target molecules. Our experts are ready to collaborate with your R&D and supply chain teams to optimize your manufacturing strategy and secure a competitive advantage in the marketplace.
