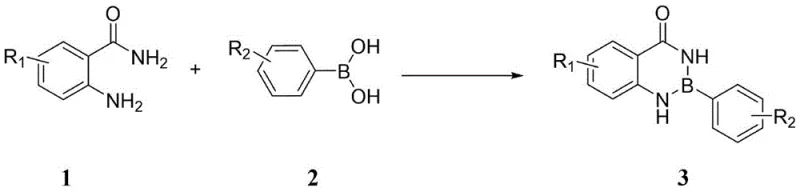
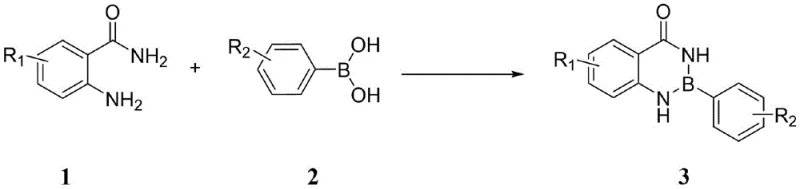
Advanced Catalyst-Free Synthesis of 1,3,2-Benzodiazaborinones for Commercial Scale-Up
The pharmaceutical and fine chemical industries are constantly seeking more efficient pathways to access complex heterocyclic scaffolds, particularly those containing boron-nitrogen bonds which are increasingly vital in modern medicinal chemistry. Patent CN114276375A introduces a groundbreaking methodology for the synthesis of 1,3,2-benzodiazaborinones and their derivatives, addressing critical bottlenecks in current manufacturing protocols. This innovation leverages a direct dehydration condensation strategy between anthranilamide derivatives and phenylboronic acids, eliminating the need for costly transition metal catalysts that have traditionally plagued this chemical space. For R&D directors and procurement specialists alike, this represents a significant shift towards greener, more cost-effective production of high-purity pharmaceutical intermediates. The technology not only simplifies the synthetic route but also enhances the overall sustainability profile of the supply chain by reducing waste and energy consumption associated with high-temperature reflux or complex purification steps required to remove metal residues.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of 1,3,2-benzodiazaborinone cores has relied heavily on methodologies that are both economically and environmentally burdensome. Traditional literature methods often necessitate the use of expensive noble metal catalysts such as nickel or palladium, which not only inflate the raw material costs but also introduce significant downstream processing challenges. The removal of trace heavy metals to meet stringent pharmaceutical purity standards requires additional purification steps, such as specialized scavenging resins or repeated recrystallizations, which inevitably reduce overall process yield and extend production timelines. Furthermore, conventional approaches frequently demand harsh reaction conditions, including high-temperature reflux in toxic solvents like toluene or the use of sensitive reagents like potassium organotrifluoroborates. These factors collectively contribute to a high carbon footprint and increased operational expenditure, making large-scale commercialization difficult for many fine chemical manufacturers who prioritize safety and regulatory compliance.
The Novel Approach
In stark contrast, the novel approach detailed in the patent data utilizes a catalyst-free dehydration condensation mechanism that operates under remarkably mild conditions. By simply mixing anthranilamide derivatives with phenylboronic acids in a suitable solvent, the reaction proceeds efficiently at room temperature, bypassing the need for external heating or inert atmosphere protection in many cases. This method capitalizes on the intrinsic reactivity of the amino and amide groups within the anthranilamide structure to facilitate ring closure with the boronic acid. The elimination of transition metals not only drastically reduces the cost of goods sold (COGS) but also simplifies the workup procedure to a basic filtration and concentration step. This streamlined process allows for the rapid generation of diverse libraries of benzodiazaborinone derivatives, accelerating the drug discovery timeline for medicinal chemists while providing a robust platform for industrial scale-up.
Mechanistic Insights into Catalyst-Free Dehydration Condensation
The core of this technological advancement lies in the unique interaction between the boron center of the phenylboronic acid and the nitrogen atoms of the anthranilamide. Unlike metal-catalyzed cross-couplings that rely on oxidative addition and reductive elimination cycles, this transformation is driven by thermodynamic stability gained through the formation of strong boron-nitrogen coordinate bonds and the release of water. The reaction kinetics are highly sensitive to the electronic nature of the substituents on the aromatic rings. Data indicates that electron-withdrawing groups, such as nitro or halogen substituents at the para or meta positions, generally enhance the electrophilicity of the boron center or stabilize the transition state, leading to excellent isolated yields ranging from 80% to 96%. Conversely, strong steric hindrance, particularly with ortho-substituted boronic acids, can impede the approach of the nucleophilic amine, resulting in lower conversions or complete reaction failure, highlighting the importance of substrate selection in process design.
Solvent engineering plays a pivotal role in optimizing this dehydration equilibrium. While traditional methods might employ tetrahydrofuran (THF) or toluene, extensive screening revealed that ethyl acetate serves as the superior medium for this transformation. In ethyl acetate, the reaction achieves a remarkable 93% yield at room temperature within just 3 hours, outperforming polar aprotic solvents like DMF or DMSO which often complicate product isolation. The choice of ethyl acetate also aligns with green chemistry principles, as it is less toxic and easier to recover than many alternative organic solvents. This mechanistic understanding allows process chemists to fine-tune reaction parameters, such as the molar ratio of reactants (optimized at 1:1.2), to maximize throughput while minimizing waste, ensuring that the final API intermediates meet the rigorous quality specifications required by global regulatory bodies.
How to Synthesize 1,3,2-Benzodiazaborinone Efficiently
To implement this synthesis effectively in a laboratory or pilot plant setting, operators must adhere to precise stoichiometric and environmental controls to ensure reproducibility and high purity. The process begins with the careful weighing of high-quality anthranilamide and phenylboronic acid starting materials, ensuring that moisture content is minimized to prevent premature hydrolysis of the boronic acid prior to reaction. The mixture is then suspended in ethyl acetate, chosen for its ability to solubilize reactants while facilitating the removal of water generated during condensation. Continuous stirring at ambient temperature is critical to maintain homogeneity and heat dissipation, although the reaction is exothermic enough that active cooling is rarely required. Monitoring the reaction progress via thin-layer chromatography (TLC) allows for precise determination of the endpoint, typically reached within 3 to 4 hours, after which the product can be isolated through simple filtration followed by vacuum drying.
- Combine anthranilamide derivatives and phenylboronic acid derivatives in ethyl acetate solvent with a molar ratio of approximately 1: 1.2.
- Stir the reaction mixture at room temperature for 3 to 4 hours to allow complete dehydration condensation.
- Filter the reaction mixture, concentrate the filtrate under reduced pressure, and purify the resulting solid to obtain the target product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this catalyst-free synthesis route offers transformative benefits that extend far beyond simple yield improvements. The most immediate impact is seen in the drastic reduction of raw material costs, as the elimination of palladium or nickel catalysts removes one of the most expensive line items from the bill of materials. Furthermore, the absence of heavy metals negates the need for costly metal scavenging resins and the associated validation testing required to prove residual metal levels are below ppm thresholds, thereby shortening the quality control cycle time significantly. This simplification of the downstream processing workflow translates directly into faster batch turnover rates and improved capacity utilization within existing manufacturing facilities, allowing suppliers to respond more agilely to fluctuating market demands without requiring capital-intensive equipment upgrades.
- Cost Reduction in Manufacturing: The economic advantage of this process is primarily derived from the complete removal of precious metal catalysts and the associated ligands, which traditionally account for a substantial portion of synthesis costs. By utilizing commodity chemicals like ethyl acetate and readily available boronic acids, the variable cost per kilogram of the final intermediate is significantly lowered. Additionally, the simplified workup procedure reduces solvent consumption and waste disposal fees, contributing to a leaner and more profitable manufacturing model that enhances competitiveness in the global marketplace for fine chemical intermediates.
- Enhanced Supply Chain Reliability: Reliance on complex catalytic systems often introduces supply chain vulnerabilities due to the geopolitical concentration of precious metal mining and refining. By shifting to a catalyst-free protocol, manufacturers can source all key starting materials from a broader base of global suppliers, mitigating the risk of shortages or price volatility. The robustness of the reaction conditions, which tolerate ambient temperatures and standard atmospheric pressure, further ensures that production can continue uninterrupted even during periods of energy constraint or infrastructure instability, guaranteeing consistent delivery schedules for downstream pharmaceutical clients.
- Scalability and Environmental Compliance: Scaling this reaction from gram to tonnage quantities presents minimal technical barriers, as the exotherm is manageable and the solvent system is well-understood in large-scale chemical engineering. The use of ethyl acetate, a solvent with a favorable environmental profile compared to chlorinated or aromatic hydrocarbons, facilitates easier compliance with increasingly strict environmental regulations regarding volatile organic compound (VOC) emissions. This alignment with green chemistry principles not only reduces the regulatory burden on the manufacturer but also appeals to end-users who are prioritizing sustainability metrics in their supplier selection criteria, fostering long-term strategic partnerships.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology, drawing directly from the experimental data and beneficial effects outlined in the patent documentation. Understanding these nuances is essential for technical teams evaluating the feasibility of integrating this route into their existing production portfolios. The answers provided reflect the specific capabilities of the catalyst-free dehydration method, highlighting its versatility across different substitution patterns while acknowledging specific limitations regarding steric hindrance.
Q: Does this synthesis require expensive transition metal catalysts?
A: No, the method described in patent CN114276375A operates without nickel or palladium catalysts, utilizing a direct dehydration condensation mechanism that significantly reduces raw material costs and eliminates heavy metal contamination risks.
Q: What is the optimal solvent for maximizing yield in this reaction?
A: Ethyl acetate has been identified as the superior solvent, providing yields up to 93% at room temperature, outperforming traditional solvents like THF, toluene, or DMF which often require harsher conditions or offer lower conversion rates.
Q: Are there steric limitations regarding the substituents on the boronic acid?
A: Yes, while para and meta substitutions generally proceed with high efficiency (80-96% yield), ortho-substituted phenylboronic acids, particularly those with halogens or multiple groups, show significantly reduced reactivity due to steric hindrance affecting the B-N bond formation.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1,3,2-Benzodiazaborinone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting innovative synthetic technologies to maintain a competitive edge in the fast-evolving landscape of pharmaceutical intermediates. Our team of expert process chemists has thoroughly analyzed the potential of the catalyst-free dehydration condensation route described in CN114276375A and is fully prepared to translate this laboratory-scale success into commercial reality. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from pilot batches to full-scale manufacturing is seamless and compliant with cGMP standards. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of 1,3,2-benzodiazaborinone delivered meets the exacting requirements of global drug developers.
We invite you to collaborate with us to leverage this cost-effective and environmentally friendly synthesis method for your next project. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume needs, demonstrating exactly how this new route can optimize your budget. We encourage you to contact us today to discuss your requirements,索取 specific COA data for our reference standards, and receive comprehensive route feasibility assessments that will accelerate your path to market. Let us be your trusted partner in delivering high-quality boron-containing intermediates that drive innovation in modern medicine.
