Advanced Synthesis of Chiral (S,S)-Salen Co(II) Catalyst for High-Purity Epoxide Resolution
Introduction to Advanced Chiral Catalyst Synthesis
The landscape of asymmetric catalysis is undergoing a significant transformation driven by the urgent need for sustainable and cost-effective manufacturing processes, particularly in the production of high-value pharmaceutical intermediates. A pivotal advancement in this field is detailed in patent CN102728407B, which discloses a novel synthetic method for (S,S)-salenCo(II) catalysts and their application in the kinetic resolution of terminal epoxide compounds. This technology addresses critical inefficiencies in traditional methods by integrating waste valorization directly into the synthesis workflow, turning what was previously considered hazardous liquid waste into a valuable resource for producing the complementary enantiomer. By leveraging the filtrate from the preparation of (R,R)-cyclohexanediamine salts, this approach not only minimizes environmental impact but also drastically streamlines the supply chain for chiral building blocks. For R&D directors and procurement managers seeking a reliable pharma intermediates supplier, understanding this closed-loop synthesis is essential for evaluating long-term cost structures and supply security.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of chiral salen cobalt complexes, often referred to as Jacobsen catalysts, has been plagued by several operational and economic bottlenecks that hinder large-scale adoption. The conventional route typically involves a multi-step process starting with the resolution of trans-1,2-cyclohexanediamine using chiral tartaric acid, followed by condensation with salicylaldehyde derivatives in mixed solvent systems. A major drawback of this traditional methodology is the reliance on toxic and difficult-to-recycle solvents such as toluene mixed with methanol, which complicates downstream processing and increases the burden on waste treatment facilities. Furthermore, the oxidation step required to convert the inactive Co(II) precursor into the active Co(III) catalyst often suffers from low conversion rates, frequently yielding less than 70%, which directly inflates the cost of goods sold. Perhaps most critically, the initial resolution step generates a filtrate rich in the unwanted enantiomer of cyclohexanediamine, which is traditionally discarded as liquid waste, representing a massive loss of potential raw material value and creating unnecessary environmental liabilities.
The Novel Approach
In stark contrast to these legacy methods, the technology described in patent CN102728407B introduces a paradigm shift by utilizing the waste filtrate from the (R,R)-process to synthesize the (S,S)-cyclohexanediamine mono-salt, effectively closing the material loop. This innovative strategy involves treating the concentrated filtrate with D-tartaric acid in a single organic solvent, such as ethanol or tetrahydrofuran, to precipitate the desired (S,S)-salt with high purity and yield.  . By eliminating the need for fresh resolution of the diamine for the (S,S) series, the process significantly reduces raw material consumption and simplifies the overall workflow. Additionally, the subsequent formation of the salen ligand and coordination with cobalt acetate is conducted in a single solvent system, avoiding the complexities of solvent exchange and azeotropic distillation associated with mixed solvent protocols. This streamlined approach not only enhances the overall yield of the catalyst to over 96% but also ensures that the final product is free from inorganic salt contaminants that often plague catalysts produced via aqueous workups.
. By eliminating the need for fresh resolution of the diamine for the (S,S) series, the process significantly reduces raw material consumption and simplifies the overall workflow. Additionally, the subsequent formation of the salen ligand and coordination with cobalt acetate is conducted in a single solvent system, avoiding the complexities of solvent exchange and azeotropic distillation associated with mixed solvent protocols. This streamlined approach not only enhances the overall yield of the catalyst to over 96% but also ensures that the final product is free from inorganic salt contaminants that often plague catalysts produced via aqueous workups.
Mechanistic Insights into (S,S)-Salen Co(II) Catalyst Formation and Activation
The core of this technological breakthrough lies in the precise control of stereochemistry during the ligand formation and the unique in situ activation mechanism of the cobalt center. The synthesis begins with the reaction of the recovered (S,S)-cyclohexanediamine mono-salt with 3,5-di-tert-butyl salicylaldehyde in the presence of a base, forming the chiral Schiff base ligand which then coordinates with cobalt acetate.  . This coordination occurs efficiently in solvents like ethanol or THF, where the steric bulk of the tert-butyl groups on the salicylaldehyde moiety plays a crucial role in defining the chiral pocket of the catalyst. Unlike traditional methods that require a separate, often inefficient chemical oxidation step to generate the active Co(III) species, this patent leverages the stability of the Co(II) precursor for storage and handling. The actual activation occurs dynamically within the reaction vessel during the hydrolytic kinetic resolution (HKR) of epoxides, where the introduction of air in the presence of acetic acid facilitates the oxidation of Co(II) to Co(III). This mechanistic nuance ensures that the catalyst is generated exactly when and where it is needed, minimizing decomposition and maximizing catalytic turnover.
. This coordination occurs efficiently in solvents like ethanol or THF, where the steric bulk of the tert-butyl groups on the salicylaldehyde moiety plays a crucial role in defining the chiral pocket of the catalyst. Unlike traditional methods that require a separate, often inefficient chemical oxidation step to generate the active Co(III) species, this patent leverages the stability of the Co(II) precursor for storage and handling. The actual activation occurs dynamically within the reaction vessel during the hydrolytic kinetic resolution (HKR) of epoxides, where the introduction of air in the presence of acetic acid facilitates the oxidation of Co(II) to Co(III). This mechanistic nuance ensures that the catalyst is generated exactly when and where it is needed, minimizing decomposition and maximizing catalytic turnover.
Furthermore, the application of this catalyst in the splitting of terminal epoxides demonstrates exceptional enantioselectivity, driven by the cooperative bimetallic mechanism inherent to salen cobalt complexes. 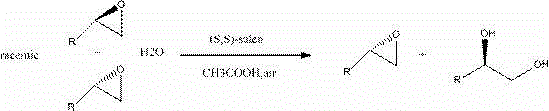 . In this process, one molecule of the activated Co(III)-salen complex acts as a Lewis acid to coordinate the epoxide, while another acts as a nucleophile to deliver a hydroxide ion, leading to the ring-opening of the epoxide. The patent data indicates that this method can achieve enantiomeric excess (ee) values of up to 99% for products like R-epichlorohydrin and S-propylene oxide, which are critical precursors for beta-blockers and other pharmaceutical agents. The ability to control impurity profiles is further enhanced by the single-solvent recrystallization steps, which effectively remove inorganic salts and unreacted aldehydes that could otherwise poison downstream reactions or complicate purification. For R&D teams, this level of mechanistic clarity and impurity control translates to more robust process validation and easier regulatory filing.
. In this process, one molecule of the activated Co(III)-salen complex acts as a Lewis acid to coordinate the epoxide, while another acts as a nucleophile to deliver a hydroxide ion, leading to the ring-opening of the epoxide. The patent data indicates that this method can achieve enantiomeric excess (ee) values of up to 99% for products like R-epichlorohydrin and S-propylene oxide, which are critical precursors for beta-blockers and other pharmaceutical agents. The ability to control impurity profiles is further enhanced by the single-solvent recrystallization steps, which effectively remove inorganic salts and unreacted aldehydes that could otherwise poison downstream reactions or complicate purification. For R&D teams, this level of mechanistic clarity and impurity control translates to more robust process validation and easier regulatory filing.
How to Synthesize (S,S)-Salen Co(II) Catalyst Efficiently
Implementing this synthesis route requires careful attention to solvent selection and temperature control to maximize the recovery of the diamine salt and the subsequent yield of the cobalt complex. The process is designed to be scalable, utilizing standard unit operations such as reflux, centrifugation, and drying that are common in fine chemical manufacturing facilities. Operators must ensure that the dehydration of the initial filtrate is thorough to prevent hydrolysis of the imine bonds during the ligand formation stage. The following guide outlines the standardized procedure derived from the patent examples, ensuring reproducibility and high quality.
- Recover (S,S)-cyclohexanediamine mono-salt from the filtrate of the (R,R)-process by reacting with D-tartaric acid in ethanol or THF under reflux.
- React the purified (S,S)-cyclohexanediamine salt with 3,5-di-tert-butyl salicylaldehyde and cobalt acetate in a single organic solvent to form the Co(II) complex.
- Activate the catalyst in situ during the epoxide splitting reaction by blowing air to oxidize Co(II) to the active Co(III) species.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented synthesis method offers profound strategic advantages that extend far beyond simple technical metrics. By transforming a waste stream into a primary feedstock, the process fundamentally alters the cost structure of chiral catalyst production, decoupling it from the volatility of fresh diamine markets. This waste-to-value proposition means that manufacturers can offer significantly reduced pricing for high-purity catalysts without compromising on quality margins. Moreover, the elimination of toxic solvents like toluene simplifies the regulatory compliance landscape, reducing the administrative burden and costs associated with environmental health and safety (EHS) reporting. The use of common solvents like ethanol or THF also enhances supply chain resilience, as these materials are widely available globally and less prone to logistical disruptions compared to specialized solvent blends.
- Cost Reduction in Manufacturing: The most immediate financial benefit stems from the complete utilization of the cyclohexanediamine resolution by-product, which effectively halves the raw material cost for the diamine component of the catalyst. By avoiding the separate purchase and resolution of the (S,S)-diamine, manufacturers can achieve substantial cost savings that can be passed down to the customer. Additionally, the high yield of the catalyst synthesis step, reported at approximately 96%, minimizes material loss and reduces the frequency of batch failures. The simplified single-solvent system further lowers utility costs by reducing the energy required for solvent recovery and distillation, contributing to a leaner and more competitive manufacturing model.
- Enhanced Supply Chain Reliability: The robustness of this synthetic route ensures a consistent and reliable supply of critical chiral catalysts, which are often bottleneck items in the production of active pharmaceutical ingredients. Because the process relies on stable intermediates and avoids sensitive oxidation steps prior to use, the shelf-life and transport stability of the Co(II) precursor are improved. This reliability allows for better inventory planning and reduces the risk of production stoppages due to catalyst shortages. Furthermore, the scalability of the process, demonstrated from kilogram to multi-kilogram scales in the patent examples, assures buyers that supply can be ramped up quickly to meet surging demand for key intermediates like epichlorohydrin derivatives.
- Scalability and Environmental Compliance: From an operational perspective, the process is inherently designed for scale-up, utilizing equipment and conditions that are standard in the fine chemical industry. The absence of mixed solvents and the efficient solid-liquid separation via centrifugation make the transition from pilot plant to commercial production seamless. Environmentally, the conversion of waste filtrate into product aligns perfectly with green chemistry principles, significantly reducing the volume of hazardous waste requiring disposal. This not only lowers waste treatment costs but also enhances the sustainability profile of the final pharmaceutical products, a factor that is increasingly important for end-users and regulatory bodies alike.
Frequently Asked Questions (FAQ)
To assist technical decision-makers in evaluating the feasibility of integrating this catalyst technology into their existing workflows, we have compiled answers to common inquiries based on the patent specifications. These questions address critical aspects regarding catalyst activity, process safety, and product purity, providing a clear picture of what to expect during technology transfer. Understanding these details is vital for assessing the compatibility of this method with current Good Manufacturing Practice (cGMP) standards.
Q: How does this patent improve upon the conventional Jacobsen catalyst synthesis?
A: The patented method eliminates the need for mixed solvents like toluene and methanol, replacing them with a single solvent system such as ethanol or tetrahydrofuran. Furthermore, it valorizes the waste filtrate containing the unwanted enantiomer from the (R,R)-process, converting it into valuable (S,S)-cyclohexanediamine salt, thereby significantly reducing raw material costs and environmental waste.
Q: What is the catalytic activity of the synthesized (S,S)-Salen Co(II) complex?
A: While the synthesized complex is initially in the Co(II) state, which typically lacks catalytic activity for hydrolytic kinetic resolution, the patent describes an in situ activation method. By blowing air through the reaction mixture in the presence of acetic acid, the Co(II) species is oxidized to the active Co(III) state, achieving enantiomeric excess (ee) values as high as 99% for terminal epoxides.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process is specifically designed for industrial scalability. It simplifies the workflow by reducing reaction steps, utilizes common and easily recyclable solvents like ethanol, and achieves high yields (up to 96% for the catalyst). The elimination of toxic toluene and the efficient recovery of by-products make it highly compliant with modern environmental and safety standards for large-scale manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (S,S)-Salen Co(II) Catalyst Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of complex chiral intermediates relies on a partnership grounded in technical expertise and manufacturing excellence. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is smooth and efficient. We are committed to delivering (S,S)-Salen Co(II) Catalyst and related chiral intermediates with stringent purity specifications, supported by our rigorous QC labs that employ advanced analytical techniques to verify enantiomeric excess and metal content. Our facility is equipped to handle the specific solvent systems and reaction conditions required by this patented process, guaranteeing a supply that meets the highest international standards.
We invite you to engage with our technical procurement team to discuss how this advanced synthesis method can optimize your specific production requirements. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the potential economic benefits of switching to this waste-utilizing catalyst technology. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your project needs, ensuring that your supply chain is both cost-effective and resilient for the long term.
