Advanced Synthetic Route for Alpha-Acetyl-Gamma-Butyrolactone Enhancing Commercial Scalability
The chemical industry is constantly evolving towards safer and more efficient manufacturing protocols, a trend vividly exemplified by the technological breakthroughs detailed in patent CN103387556A. This specific intellectual property outlines a revolutionary synthetic method for alpha-acetyl-gamma-butyrolactone, a critical intermediate in the production of various high-value pharmaceutical and agrochemical compounds. The core innovation lies in the strategic replacement of volatile and dangerous reagents with stable, manageable alternatives, thereby addressing long-standing safety concerns that have plagued the sector for decades. By shifting away from the traditional reliance on ethylene oxide and metallic sodium, this process not only mitigates severe operational hazards but also streamlines the purification workflow. For R&D directors and process engineers, understanding this shift is paramount as it represents a move towards inherently safer design principles that align with modern regulatory standards. The patent describes a robust two-step sequence that leverages the reactivity of ketene dimer with halo-alcohols, creating a pathway that is both chemically elegant and industrially viable. This report analyzes the technical nuances of this method, highlighting its potential to redefine supply chain reliability for high-purity pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of alpha-acetyl-gamma-butyrolactone has been dominated by two primary methodologies, both of which carry significant drawbacks that impact operational continuity and worker safety. The first method, often referred to as the epoxy method, relies heavily on the reaction between methyl acetoacetate and ethylene oxide in an aqueous system. This approach is fraught with peril due to the extremely low flash point and high vapor pressure of ethylene oxide, making transport and storage logistically difficult and inherently dangerous. Furthermore, this legacy process necessitates the use of benzene as an extraction solvent, a known carcinogen that imposes strict environmental disposal costs and health monitoring requirements. The overall yield of this method is notoriously poor, often hovering around 60%, which translates to substantial raw material waste and increased cost per kilogram of finished product. The second conventional route utilizes metallic sodium as a strong base in a toluene system, which presents an even more acute safety profile due to the pyrophoric nature of sodium metal. The generation of hydrogen gas during the reaction creates a high risk of explosion, and the violent nature of the reaction requires specialized equipment and rigorous temperature control to prevent runaway scenarios. These traditional pathways represent a significant liability for any modern chemical manufacturer aiming for sustainable and safe operations.
The Novel Approach
In stark contrast to the hazardous legacy methods, the novel approach disclosed in the patent utilizes a much safer and more controlled reaction environment centered around the use of 2-chloroethanol or 2-bromoethanol. This substitution effectively removes the need for gaseous ethylene oxide and solid metallic sodium, replacing them with stable liquid reagents that are easier to handle and dose with precision. The process initiates with an aldol reaction between ketene dimer and the halo-alcohol, catalyzed by sodium alkoxide, which proceeds smoothly at a moderate temperature of 75°C. This first step generates an intermediate ester, specifically acetyl chloroethanol acetate, which serves as the precursor for the subsequent cyclization. The second stage involves an intramolecular Claisen-Schmidt condensation triggered by an alkali catalyst such as sodium carbonate or liquid caustic soda. This cyclization occurs under mild conditions, typically between 15°C and 30°C, allowing for excellent thermal control and minimizing the formation of thermal degradation byproducts. The result is a direct generation of alpha-acetyl-gamma-butyrolactone with an associating yield exceeding 85%, demonstrating a clear superiority in both safety and efficiency over the conventional epoxy and sodium metal routes.
Mechanistic Insights into Ketene Dimer Ring-Opening and Cyclization
The chemical elegance of this synthesis lies in the specific reactivity of ketene dimer, which acts as a versatile building block for constructing the gamma-lactone ring system. In the initial step, the nucleophilic oxygen of the 2-chloroethanol attacks the electrophilic carbonyl carbon of the ketene dimer, facilitated by the basic environment provided by the sodium alkoxide catalyst. This ring-opening reaction is highly selective, ensuring that the acetyl group remains intact while forming the necessary ester linkage with the chloroethyl chain. The stability of the intermediate formed in this step is crucial, as it allows for isolation or direct progression to the next stage without significant decomposition. The subsequent cyclization is driven by the deprotonation of the alpha-carbon adjacent to the ketone group by the alkali catalyst, generating a reactive enolate species. This enolate then undergoes an intramolecular nucleophilic substitution, displacing the chloride ion and closing the five-membered lactone ring. This mechanism avoids the harsh conditions required for direct lactonization of hydroxy acids and bypasses the need for high-energy intermediates associated with ethylene oxide polymerization.
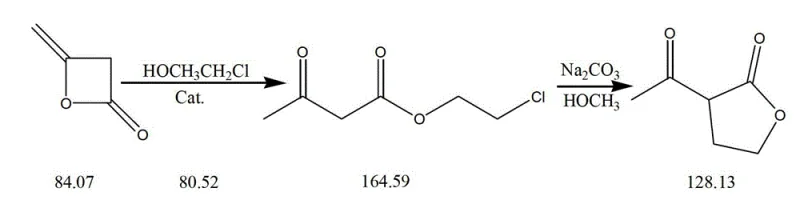
From an impurity control perspective, this mechanistic pathway offers distinct advantages by minimizing side reactions that typically plague high-temperature processes. The mild temperature range of 15°C to 30°C during the critical cyclization step suppresses the formation of polymeric byproducts and prevents the degradation of the sensitive acetyl moiety. Furthermore, the use of sodium carbonate or controlled amounts of liquid caustic soda allows for precise pH management, ensuring that the reaction stops at the desired lactone stage without over-hydrolysis of the ester bond. The patent data indicates that the crude product obtained after concentration possesses a purity content of approximately 90% as determined by HPLC, which significantly reduces the burden on downstream purification units. For R&D teams focused on impurity profiling, this route presents a cleaner reaction profile with fewer unknown peaks, simplifying the validation process for regulatory filings. The ability to tune the catalyst strength and reaction temperature provides a robust handle for optimizing the ratio of the desired alpha-acetyl isomer against potential beta-isomers or open-chain acid impurities.
How to Synthesize Alpha-Acetyl-Gamma-Butyrolactone Efficiently
Implementing this synthesis route requires careful attention to the stoichiometry of the ketene dimer and the halo-alcohol, as well as strict temperature control during the exothermic cyclization phase. The patent outlines a standardized procedure where the initial aldol reaction is conducted under reflux to ensure complete conversion of the ketene dimer, followed by a cooling phase before the addition of the base. Operators must ensure that the mixing is uniform to prevent localized hot spots that could trigger premature cyclization or decomposition. The choice of solvent, such as toluene or glycol dimethyl ether, plays a critical role in solubilizing the intermediate and facilitating heat transfer during the base addition. Detailed standard operating procedures for this specific transformation are essential for maintaining batch-to-batch consistency and achieving the reported high yields.
- Conduct an aldol reaction between ketene dimer and 2-chloroethanol using sodium alkoxide catalyst at 75°C.
- Add solvent to the resulting ester mixture and cool to 15-30°C.
- Perform intramolecular Claisen-Schmidt condensation with an alkali catalyst to cyclize into the final lactone product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this novel synthetic route offers compelling economic and logistical benefits that extend beyond simple yield improvements. The elimination of ethylene oxide and metallic sodium from the supply chain removes the need for specialized storage facilities and hazardous material handling certifications, which directly translates to reduced overhead costs and simplified logistics. Sourcing 2-chloroethanol and ketene dimer is generally more stable and less prone to market volatility compared to the highly regulated and dangerous reagents used in conventional methods. This stability ensures a more reliable supply of raw materials, reducing the risk of production stoppages due to supplier compliance issues or transportation restrictions on hazardous goods. Furthermore, the significant increase in yield from roughly 60% to over 85% means that less raw material is required to produce the same amount of finished product, effectively lowering the variable cost of goods sold. This efficiency gain is compounded by the reduction in waste disposal costs, as the process generates fewer hazardous byproducts and avoids the use of benzene, a solvent with expensive disposal requirements.
- Cost Reduction in Manufacturing: The economic impact of this process is driven primarily by the drastic simplification of the safety infrastructure required for production. By removing the need for explosion-proof reactors designed for ethylene oxide or inert atmosphere systems for sodium metal, capital expenditure for new plants can be significantly optimized. Additionally, the higher conversion rates mean that energy consumption per unit of product is lower, as less material needs to be heated, cooled, and distilled. The avoidance of benzene also eliminates the costly recovery and incineration steps associated with aromatic solvents, further enhancing the margin profile. Qualitatively, the reduction in safety incidents and insurance premiums associated with handling less hazardous chemicals contributes to a leaner operational cost structure. This makes the final alpha-acetyl-gamma-butyrolactone product more price-competitive in the global market while maintaining healthy profit margins for the manufacturer.
- Enhanced Supply Chain Reliability: Supply chain resilience is greatly improved by the use of commodity chemicals that are widely available from multiple global suppliers. Unlike metallic sodium, which requires careful moisture-free transport and has limited suppliers due to safety regulations, 2-chloroethanol is a standard industrial chemical with a robust distribution network. This diversification of the supply base reduces the risk of single-source bottlenecks and allows for more flexible procurement strategies. The milder reaction conditions also mean that the manufacturing process is less susceptible to disruptions caused by utility failures, such as loss of cooling water, since the reaction is not violently exothermic. Consequently, lead times for high-purity pharmaceutical intermediates can be shortened as the production schedule becomes more predictable and less prone to emergency shutdowns. This reliability is crucial for downstream customers who depend on just-in-time delivery for their own API synthesis campaigns.
- Scalability and Environmental Compliance: From an environmental standpoint, this process aligns perfectly with green chemistry principles by reducing the E-factor through higher yields and safer solvents. The absence of heavy metals and persistent organic pollutants like benzene simplifies the wastewater treatment process, making it easier to meet stringent discharge regulations in various jurisdictions. Scalability is enhanced because the reaction kinetics are manageable at large volumes, avoiding the heat transfer limitations that often plague batch processes involving highly reactive metals. The ability to run the cyclization at near-ambient temperatures reduces the energy load on the plant's HVAC and chilling systems, contributing to a lower carbon footprint. For companies aiming to achieve sustainability certifications or reduce their Scope 1 and Scope 2 emissions, adopting this technology provides a clear pathway to greener manufacturing without sacrificing productivity or quality.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthetic technology. These answers are derived directly from the experimental data and comparative analysis provided in the patent documentation to ensure accuracy and relevance for potential partners. Understanding these details is essential for evaluating the feasibility of integrating this route into existing production lines or new facility designs.
Q: What are the primary safety advantages of this new synthesis method?
A: The novel process eliminates the use of highly hazardous ethylene oxide and reactive metallic sodium, significantly reducing explosion and fire risks associated with traditional manufacturing.
Q: How does the yield of this method compare to conventional epoxy methods?
A: While conventional epoxy methods typically achieve yields around 60%, this patented process demonstrates an associating yield exceeding 85%, offering substantial material efficiency.
Q: Can this process be scaled for industrial production?
A: Yes, the use of stable liquid reagents like 2-chloroethanol and mild reaction temperatures (15-30°C for cyclization) makes the process highly suitable for large-scale commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Alpha-Acetyl-Gamma-Butyrolactone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic technologies to meet the evolving demands of the global pharmaceutical market. Our team of expert chemists has thoroughly analyzed the potential of the route described in CN103387556A and is fully prepared to implement this safer, high-yield process at commercial scale. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab bench to reactor is seamless and efficient. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of alpha-acetyl-gamma-butyrolactone meets the highest industry standards. By leveraging our expertise in process optimization, we can help you secure a stable supply of this key intermediate while minimizing your environmental footprint and operational risks.
We invite you to contact our technical procurement team to discuss how this innovative synthesis method can benefit your specific project requirements. We are ready to provide a Customized Cost-Saving Analysis that quantifies the potential economic advantages of switching to this safer route for your supply chain. Please reach out to request specific COA data and route feasibility assessments tailored to your volume needs. Partnering with us ensures access to cutting-edge chemical manufacturing solutions that prioritize safety, quality, and sustainability.
