Advanced Catalytic Synthesis of 1,2-Dihydroquinazolin-4(3H)-ones for Commercial Scale Production
Advanced Catalytic Synthesis of 1,2-Dihydroquinazolin-4(3H)-ones for Commercial Scale Production
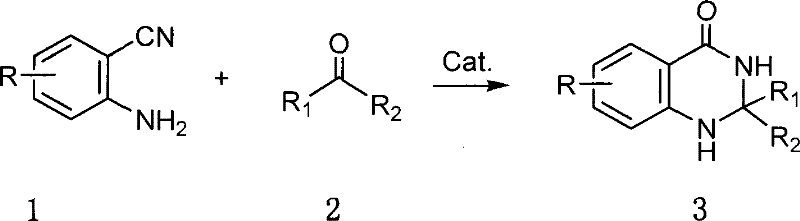
The pharmaceutical and agrochemical industries continuously demand efficient, scalable routes for nitrogen-containing heterocycles due to their prevalence in bioactive molecules. Patent CN101190899B introduces a transformative methodology for synthesizing 1,2-dihydroquinazolin-4(3H)-ones, a scaffold critical for antitumor, anti-inflammatory, and herbicidal applications. This innovation addresses long-standing inefficiencies in heterocyclic construction by utilizing a simple condensation between o-aminoaromatic nitriles and carbonyl compounds. Unlike traditional approaches that rely on harsh conditions or exotic catalysts, this method employs accessible Lewis acids such as anhydrous zinc chloride to drive the cyclization under mild thermal conditions. The strategic shift towards base-metal catalysis not only enhances reaction safety but also drastically simplifies the purification workflow, positioning this technology as a cornerstone for reliable pharmaceutical intermediates supplier networks seeking to optimize their manufacturing portfolios.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the quinazolinone core has been plagued by significant operational and economic hurdles that hinder industrial adoption. Classical pathways, such as the Niementowski reaction, necessitate the use of anthranilic acid derivatives and formamide under excessively high temperatures, often leading to thermal degradation and difficult-to-control exotherms. Alternative modern strategies have attempted to mitigate these issues by employing transition metal catalysts like samarium iodide, titanium tetrachloride-zinc mixtures, or ruthenium carbonyl complexes. While these methods offer milder conditions, they introduce prohibitive costs associated with precious metal procurement and complex catalyst preparation. Furthermore, the presence of heavy metals necessitates stringent downstream purification to meet regulatory limits for residual metals in active pharmaceutical ingredients, adding layers of complexity and expense to the overall manufacturing process that erode profit margins.
The Novel Approach
The methodology disclosed in the patent represents a paradigm shift by leveraging inexpensive, commercially abundant Lewis acids to facilitate the cyclocondensation reaction. By reacting o-aminoaromatic nitriles directly with aldehydes or ketones in the presence of catalysts like anhydrous zinc chloride or aluminum trichloride, the process achieves high conversion rates without the need for exotic reagents. This approach allows for the use of common polar solvents such as DMF or toluene and supports both conventional heating and microwave promotion, offering flexibility in process engineering. The robustness of this system is evident in its ability to tolerate a wide array of functional groups, enabling the synthesis of diverse derivatives from a single standardized protocol. This simplicity translates directly into operational excellence, reducing the barrier to entry for cost reduction in pharmaceutical intermediates manufacturing while maintaining high product integrity.
Mechanistic Insights into ZnCl2-Catalyzed Cyclization
The efficacy of this synthetic route lies in the specific activation of the nitrile functionality by the Lewis acid catalyst. Anhydrous zinc chloride coordinates with the nitrogen atom of the nitrile group, increasing the electrophilicity of the carbon atom and facilitating nucleophilic attack by the amine or the imine intermediate formed with the carbonyl compound. This activation lowers the energy barrier for the subsequent intramolecular cyclization, allowing the reaction to proceed efficiently at moderate temperatures ranging from 100°C to 160°C. The mechanism avoids the formation of unstable intermediates that often plague acid-catalyzed condensations, thereby minimizing the generation of polymeric byproducts. For R&D teams, understanding this mechanistic pathway is crucial for optimizing reaction parameters, as the stoichiometry of the catalyst (typically 1 to 1.5 equivalents) plays a pivotal role in driving the equilibrium towards the desired heterocyclic product without promoting side reactions.
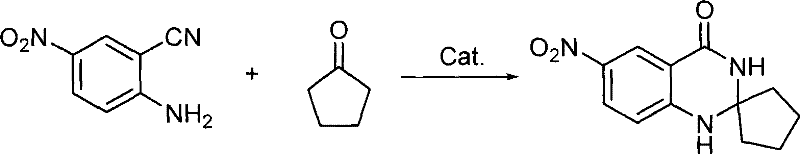
Impurity control is inherently superior in this system due to the mild nature of the catalytic environment. Unlike strong Brønsted acids which can induce hydrolysis of the nitrile group to the corresponding amide or carboxylic acid, the Lewis acid conditions preserve the integrity of the cyano moiety until cyclization occurs. The patent data illustrates that substrates bearing sensitive electron-withdrawing groups, such as nitro functionalities, remain intact throughout the process, yielding products with high spectral purity as confirmed by NMR and MS analysis. This selectivity is vital for producing high-purity OLED material precursors or pharmaceutical intermediates where trace impurities can compromise biological activity. The ability to utilize both aliphatic and aromatic ketones, including cyclic variants like cyclopentanone and cyclohexanone, further demonstrates the versatility of the mechanism in accommodating steric bulk without sacrificing yield.
How to Synthesize 1,2-Dihydroquinazolin-4(3H)-ones Efficiently
Implementing this synthesis requires careful attention to reagent quality and thermal management to maximize yield and minimize cycle time. The process begins with the precise weighing of o-aminoaromatic nitriles and the chosen carbonyl partner, ensuring a molar ratio that favors product formation, typically between 1:1 and 1:2. The addition of the anhydrous zinc chloride catalyst must be performed under inert conditions to prevent moisture deactivation, followed by heating the mixture to reflux. Detailed operational parameters, including specific solvent volumes and cooling rates for crystallization, are critical for reproducibility. For a comprehensive guide on executing this transformation with optimal safety and efficiency, please refer to the standardized protocol outlined below which encapsulates the critical steps derived from the patent examples.
- Mix o-aminoaromatic nitrile and aldehyde/ketone in a polar solvent like DMF or toluene with anhydrous ZnCl2 catalyst.
- Heat the reaction mixture to reflux (100-160°C) for 1-10 hours or utilize microwave promotion for accelerated kinetics.
- Cool the mixture, neutralize with alkali, filter the solid, and purify the crude product via recrystallization or column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement perspective, this technology offers substantial strategic benefits by decoupling production costs from volatile precious metal markets. The substitution of expensive ruthenium or samarium catalysts with commodity-grade zinc chloride results in a drastic reduction in raw material expenditure, directly impacting the bottom line. Additionally, the simplified workup procedure, which involves basic aqueous neutralization and filtration, eliminates the need for specialized scavenger resins or complex extraction trains required for heavy metal removal. This streamlining of the downstream process not only accelerates batch turnover but also reduces the consumption of organic solvents and auxiliary chemicals, contributing to a leaner, more sustainable manufacturing operation that aligns with modern green chemistry principles.
- Cost Reduction in Manufacturing: The economic model of this synthesis is fundamentally superior due to the elimination of high-cost catalytic systems. By utilizing zinc chloride, a bulk chemical available at a fraction of the cost of transition metal complexes, manufacturers can achieve significant savings on bill of materials. Furthermore, the avoidance of expensive ligand systems and the reduction in catalyst loading requirements mean that the overall cost per kilogram of the final API intermediate is substantially lowered. This cost efficiency allows for more competitive pricing strategies in the global market without compromising on the quality or purity specifications required by regulatory bodies.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of universally available starting materials and catalysts. Unlike specialized organometallic reagents that may suffer from long lead times or geopolitical supply constraints, zinc chloride and common aromatic nitriles are produced at scale by multiple vendors worldwide. This redundancy ensures continuous production capability even during market disruptions. Moreover, the robustness of the reaction conditions means that the process is less sensitive to minor variations in raw material quality, reducing the risk of batch failures and ensuring a consistent flow of high-purity pharmaceutical intermediates to downstream customers.
- Scalability and Environmental Compliance: The process is inherently designed for commercial scale-up of complex heterocyclic intermediates, featuring straightforward thermal profiles that are easily managed in large reactors. The absence of toxic heavy metals simplifies waste treatment protocols, as the effluent does not require specialized processing for metal recovery or containment. This ease of compliance with environmental regulations reduces the administrative and operational burden on manufacturing sites. The ability to recycle solvents and the high atom economy of the condensation reaction further enhance the environmental profile, making this a preferred route for companies aiming to reduce their carbon footprint while scaling production capacities.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. These insights are derived directly from the experimental data and claims within the patent documentation, providing a factual basis for decision-making. Understanding these nuances helps stakeholders evaluate the feasibility of adopting this technology for their specific product pipelines and ensures alignment with quality and regulatory expectations.
Q: What are the primary advantages of using ZnCl2 over transition metal catalysts for this synthesis?
A: Zinc chloride is significantly more cost-effective and readily available compared to expensive transition metal complexes like Ruthenium or Samarium iodide. Furthermore, it eliminates the need for rigorous heavy metal removal steps, simplifying downstream processing and reducing environmental compliance costs.
Q: Can this method accommodate diverse substrate substituents?
A: Yes, the patent demonstrates excellent tolerance for both electron-withdrawing groups (such as nitro and halogens) and electron-donating groups (like alkyl and alkoxy) on the aromatic ring, allowing for the synthesis of a wide library of derivatives without modifying reaction conditions.
Q: Is the purification process scalable for industrial manufacturing?
A: The workup involves simple aqueous neutralization and filtration, followed by standard recrystallization or chromatography. This avoids complex extraction sequences, making the process highly amenable to large-scale commercial production with minimal waste generation.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1,2-Dihydroquinazolin-4(3H)-one Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of robust synthetic routes in the development of next-generation therapeutics. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, which employ advanced analytical techniques to verify identity and assay. By leveraging the efficiencies of the ZnCl2-catalyzed method, we can offer our partners a reliable source of high-quality intermediates that support rapid drug development timelines.
We invite potential collaborators to engage with our technical procurement team to discuss how this innovative synthesis can be tailored to your specific needs. Whether you require a Customized Cost-Saving Analysis for an existing product line or need specific COA data for a new project, our experts are ready to provide detailed route feasibility assessments. Partnering with us ensures access to cutting-edge chemical technology combined with the reliability and scale necessary to support your global supply chain demands effectively.
