Revolutionizing Quinone Production: Green Aqueous Catalysis for High-Purity Pharmaceutical Intermediates
Revolutionizing Quinone Production: Green Aqueous Catalysis for High-Purity Pharmaceutical Intermediates
The pharmaceutical and fine chemical industries are currently undergoing a significant paradigm shift towards sustainable manufacturing processes, driven by both regulatory pressure and economic necessity. Patent CN1824646A introduces a groundbreaking methodology for the synthesis of aryl or arylamino group substituted quinone compounds, utilizing pure water as the reaction medium. This innovation addresses critical pain points in traditional organic synthesis, specifically the reliance on hazardous organic solvents and expensive transition metal catalysts. By leveraging the unique physicochemical properties of water, such as hydrogen bonding and the hydrophobic effect, this technology enables the efficient 1,4-conjugate addition of aromatic nucleophiles to p-benzoquinone or naphthoquinone scaffolds. For R&D directors and procurement managers seeking a reliable pharmaceutical intermediate supplier, this water-mediated approach represents a substantial opportunity for cost reduction in fine chemical manufacturing while ensuring high purity standards required for downstream drug synthesis.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of aryl-substituted quinones has been fraught with significant technical and economic challenges that hinder scalable production. Traditional routes often rely on the oxidation of aromatic rings using stoichiometric amounts of transition metal oxidants, which are not only expensive but also generate heavy metal waste streams that are costly to treat and dispose of. Alternative methods, such as the Meerwein arylation of quinones with diazonium salts, frequently suffer from poor yields and the formation of complex mixtures that require difficult purification steps. Furthermore, palladium-catalyzed coupling reactions, while powerful, impose severe limitations on substrate scope and require rigorous exclusion of moisture and oxygen, increasing operational complexity. These conventional processes often necessitate the use of chlorinated or aromatic organic solvents, posing serious safety hazards and environmental liabilities that modern supply chains strive to eliminate.
The Novel Approach
In stark contrast to these legacy methods, the technology disclosed in CN1824646A utilizes a simple, robust, and environmentally benign protocol that operates efficiently in pure water. The core innovation lies in the discovery that amino-group or methoxy-group substituted aromatic compounds can undergo smooth conjugate addition to quinones under mild acid catalysis without the need for organic co-solvents. This method achieves impressive isolated yields ranging from 69% to 93%, demonstrating superior efficiency compared to many literature precedents. The reaction proceeds at room temperature, eliminating the energy costs associated with heating or cooling, and utilizes commercially available Lewis acids like Indium trifluoromethanesulfonate. This shift to aqueous media simplifies the workup procedure significantly, as the product can often be extracted directly, bypassing the need for complex protection and deprotection strategies common in organic phase synthesis.

Mechanistic Insights into Acid-Catalyzed Conjugate Addition in Aqueous Media
The mechanistic foundation of this synthesis relies on the activation of the quinone electrophile by a Lewis or Brønsted acid catalyst, which enhances the susceptibility of the beta-carbon to nucleophilic attack. In an aqueous environment, the hydrophobic effect plays a crucial role; non-polar organic reactants are forced into close proximity within the water matrix, effectively increasing their local concentration and accelerating the reaction rate beyond what is observed in organic solvents. The acid catalyst coordinates with the carbonyl oxygen of the quinone, lowering the LUMO energy level and facilitating the 1,4-addition of the electron-rich aromatic ring, such as N,N-dimethylaniline derivatives. This activation mode is highly selective, minimizing side reactions such as polymerization or over-oxidation that typically plague quinone chemistry in harsher conditions.
Furthermore, the use of water as a solvent provides a unique mechanism for impurity control and product stability. Unlike organic solvents that may participate in side reactions or stabilize reactive intermediates leading by-products, water acts as a heat sink and a stabilizing medium for the polar transition states involved in the catalytic cycle. The mild acidic conditions prevent the decomposition of sensitive functional groups on the aromatic substrates, ensuring a clean impurity profile that is critical for high-purity pharmaceutical intermediates. For instance, when N-methylaniline derivatives are used as nucleophiles, the nitrogen atom acts as the nucleophilic species, leading to arylamino-substituted products with high regioselectivity. This mechanistic understanding allows for precise tuning of reaction parameters to optimize the commercial scale-up of complex quinone derivatives.
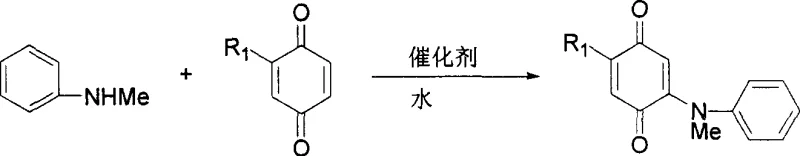
How to Synthesize Aryl-Substituted Quinones Efficiently
The practical implementation of this synthesis route is designed for ease of operation, making it highly attractive for both laboratory-scale optimization and industrial manufacturing. The process begins by simply mixing the aromatic nucleophile, such as an N,N-dimethylaniline derivative or an anisole derivative, with the quinone substrate in water at room temperature. The molar ratio is typically adjusted between 1:1 and 1:2 equivalents depending on whether mono- or di-substituted products are desired. Following the addition of a catalytic amount of acid (approximately 5 mol%), the mixture is stirred for a period ranging from 30 minutes to 24 hours. The detailed standardized synthesis steps, including specific workup and purification protocols, are outlined below to ensure reproducibility and quality control.
- Mix aromatic compounds (such as N,N-dimethylaniline derivatives) and benzoquinone derivatives in water at a molar ratio of 1: 1 to 1:2 at room temperature.
- Add an acid catalyst such as Indium trifluoromethanesulfonate (5 mol%) to the mixture and stir for 30 minutes to 24 hours depending on substrate reactivity.
- Extract the aqueous reaction mixture with dichloromethane, dry the organic phase over anhydrous sodium sulfate, and purify via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this aqueous synthesis technology translates directly into tangible operational benefits and risk mitigation. The elimination of expensive transition metal catalysts like palladium removes a significant cost driver from the bill of materials, while also simplifying the supply chain by reducing dependency on scarce precious metals. Moreover, the use of water as the primary solvent drastically reduces the volume of hazardous waste generated, leading to lower disposal fees and a smaller environmental footprint. This aligns perfectly with the sustainability goals of major multinational corporations, enhancing the marketability of the final product. The simplicity of the reaction conditions also means that the process can be easily transferred to existing manufacturing facilities without requiring specialized high-pressure or cryogenic equipment.
- Cost Reduction in Manufacturing: The economic advantages of this process are driven primarily by the substitution of costly organic solvents and precious metal catalysts with inexpensive water and earth-abundant Lewis acids. By operating at room temperature, the process eliminates the energy consumption associated with heating reactors or maintaining low-temperature conditions, resulting in significant utility savings. Additionally, the high yields reported (up to 93%) minimize raw material waste, ensuring that a greater proportion of input costs are converted into saleable product. The simplified workup procedure, which avoids complex distillation or crystallization steps often required to remove organic solvents, further reduces labor and processing time, contributing to a lower overall cost of goods sold.
- Enhanced Supply Chain Reliability: Relying on water as a solvent mitigates the risks associated with the volatility and flammability of traditional organic solvents, improving plant safety and reducing insurance premiums. The raw materials required, such as substituted anilines and benzoquinones, are commodity chemicals with stable global supply chains, ensuring consistent availability and price stability. The robustness of the reaction conditions means that production schedules are less likely to be disrupted by equipment failures or stringent environmental controls, allowing for more reliable lead times. This reliability is crucial for maintaining continuous production flows in the pharmaceutical sector, where delays can have cascading effects on downstream drug development timelines.
- Scalability and Environmental Compliance: The inherent safety of running reactions in water makes this process highly scalable from kilogram to multi-ton production levels without encountering the thermal runaway risks often seen in organic exothermic reactions. The absence of VOC emissions during the reaction phase simplifies compliance with increasingly strict environmental regulations regarding air quality and solvent emissions. Waste treatment is streamlined since the aqueous waste stream is easier to treat than mixed organic waste, reducing the burden on effluent treatment plants. This environmental compatibility positions manufacturers as responsible partners in the green chemistry initiative, potentially unlocking incentives and preferential status with eco-conscious clients.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this aqueous quinone synthesis technology. These answers are derived directly from the experimental data and scope defined in the patent literature, providing a factual basis for decision-making. Understanding these nuances is essential for evaluating the feasibility of integrating this route into your current manufacturing portfolio. We encourage technical teams to review these details to assess the alignment with their specific project requirements.
Q: What are the primary advantages of using water as a solvent for quinone synthesis?
A: Using water eliminates the need for volatile organic compounds (VOCs), significantly reducing environmental impact and disposal costs. Additionally, the hydrophobic effect in water can accelerate reaction rates and improve selectivity compared to organic solvents.
Q: Which catalysts are effective for this aqueous conjugate addition reaction?
A: The patent specifies Lewis acids and protonic acids such as Indium trifluoromethanesulfonate (In(OTf)3), Scandium trifluoromethanesulfonate (Sc(OTf)3), and Trifluoromethanesulfonic acid (TfOH) as highly effective catalysts.
Q: Can this method produce di-substituted quinone derivatives?
A: Yes, by adjusting the feed ratio of the aromatic compound to the quinone derivative to 1:1, the process can selectively produce 2,6-diaryl substituted or 2,5-diarylamino substituted p-benzoquinone compounds.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Aryl-Substituted Quinones Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of green chemistry technologies like the one described in CN1824646A for the production of high-value quinone intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from lab bench to market is seamless and efficient. Our state-of-the-art facilities are equipped to handle aqueous chemistries with the highest standards of safety and quality, supported by rigorous QC labs that guarantee stringent purity specifications for every batch. We are committed to delivering high-purity pharmaceutical intermediates that meet the exacting demands of the global healthcare industry.
We invite you to collaborate with us to leverage this innovative synthesis route for your next project. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how this water-mediated process can optimize your budget. Please contact our technical procurement team today to request specific COA data for our quinone portfolio and to discuss route feasibility assessments for your target molecules. Together, we can drive efficiency and sustainability in the manufacture of complex chemical building blocks.
