Scalable FeCl3-Catalyzed Synthesis of 5-Trifluoromethyl-1,2,4-Triazoles for Advanced Pharmaceutical Manufacturing
Scalable FeCl3-Catalyzed Synthesis of 5-Trifluoromethyl-1,2,4-Triazoles for Advanced Pharmaceutical Manufacturing
The landscape of heterocyclic chemistry is continually evolving to meet the rigorous demands of modern drug discovery, particularly for scaffolds that offer enhanced metabolic stability and bioavailability. Patent CN111978265B introduces a transformative preparation method for 5-trifluoromethyl substituted 1,2,4-triazole derivatives, a structural motif prevalent in high-value pharmaceuticals such as Maraviroc, Triazolam, Sitagliptin, and Deferasirox. This innovation addresses critical bottlenecks in traditional synthetic routes by leveraging a cost-effective ferric chloride (FeCl3) catalytic system that operates under mild, air-tolerant conditions. For R&D directors and process chemists, this represents a significant leap forward in accessing complex fluorinated heterocycles without the burden of exotic reagents or苛刻 reaction environments. The methodology not only streamlines the synthetic pathway but also ensures high purity profiles essential for regulatory compliance in API manufacturing.
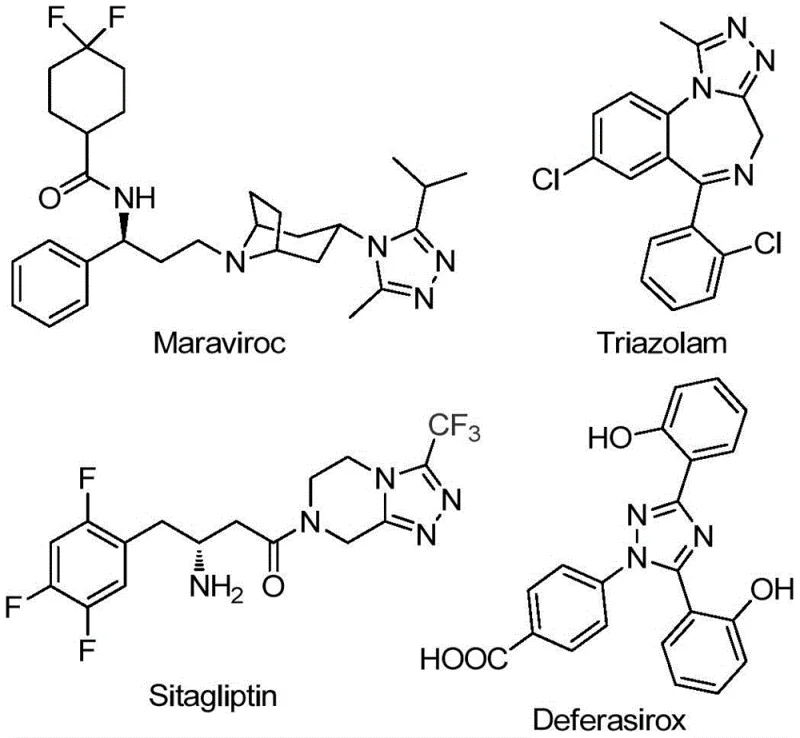
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the 1,2,4-triazole core bearing a trifluoromethyl group has been fraught with synthetic challenges that hinder large-scale production. Traditional literature methods often rely on harsh conditions, such as the condensation of 3,5-ditrifluoromethyl-1,3,4-oxadiazoles with primary amines or the cyclization of trifluoromethyl hydrazides with amidines. These legacy processes are frequently characterized by narrow substrate scopes, meaning they fail to accommodate diverse functional groups necessary for medicinal chemistry optimization. Furthermore, many conventional routes require stringent anhydrous and oxygen-free environments, necessitating expensive inert gas setups and specialized equipment that drive up capital expenditure. The multi-step nature of some older protocols also leads to cumulative yield losses and generates substantial chemical waste, creating environmental and disposal burdens that conflict with modern green chemistry principles.
The Novel Approach
In stark contrast, the novel approach detailed in the patent utilizes a tandem cyclization strategy starting from readily available trifluoroethylimide chlorides and hydrazides. This method eliminates the need for sensitive reagents and allows the reaction to proceed efficiently in common aprotic solvents like 1,4-dioxane. By employing a two-stage temperature protocol—initially at moderate temperatures for coupling followed by elevated temperatures for cyclization—the process achieves excellent conversion rates across a wide array of substrates. The use of inexpensive iron salts as promoters instead of precious metals drastically reduces raw material costs. This robustness makes the technology particularly attractive for the commercial scale-up of complex pharmaceutical intermediates, ensuring a reliable supply chain for critical drug substances.
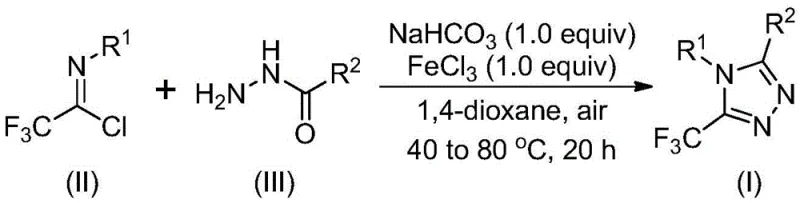
Mechanistic Insights into FeCl3-Catalyzed Cyclization
The mechanistic pathway proposed for this transformation involves a sophisticated interplay between base-mediated activation and Lewis acid catalysis. Initially, sodium bicarbonate acts as a mild base to promote the intermolecular formation of a carbon-nitrogen bond between the trifluoroethylimide chloride and the hydrazide nucleophile. This step generates a key trifluoroacetamidine intermediate under relatively mild thermal conditions (30-50°C). The subsequent addition of ferric chloride triggers the critical ring-closing event. As a Lewis acid, FeCl3 coordinates with the nitrogen atoms, facilitating an intramolecular dehydration condensation that aromatizes the five-membered ring to form the stable 1,2,4-triazole core. This dual-activation strategy ensures high regioselectivity and minimizes the formation of unwanted isomers or polymeric byproducts.
From an impurity control perspective, this mechanism offers distinct advantages for process development. The stepwise addition of reagents and the specific temperature ramping allow for tight control over reaction kinetics, preventing runaway exotherms that often lead to decomposition. The tolerance of the catalytic system to various electronic environments on the aromatic rings (electron-donating methyl groups or electron-withdrawing halogens) suggests a robust catalytic cycle that is not easily poisoned by substrate variations. This predictability is vital for maintaining consistent quality attributes in batch-to-batch production, reducing the need for extensive re-optimization when scaling from gram to kilogram quantities.
How to Synthesize 5-Trifluoromethyl-1,2,4-Triazole Derivatives Efficiently
The operational simplicity of this synthesis makes it highly accessible for laboratory and pilot plant settings. The protocol begins by combining the starting materials in a standard reactor without the need for glovebox techniques. Detailed standardized synthesis steps follow the logical progression of mixing, heating, catalyst addition, and workup. Operators should note that the choice of solvent is critical, with 1,4-dioxane providing optimal solubility and reaction efficiency compared to other polar aprotic options. The following guide outlines the precise execution of this patented methodology.
- Mix sodium bicarbonate, trifluoroethylimide chloride, and hydrazide in an aprotic solvent like 1,4-dioxane.
- Heat the mixture at 30-50°C for 8-16 hours to facilitate intermolecular carbon-nitrogen bond formation.
- Add ferric chloride catalyst and increase temperature to 70-90°C for 6-10 hours to complete the intramolecular dehydration cyclization.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route translates directly into tangible operational efficiencies and risk mitigation. The shift away from precious metal catalysts and sensitive reagents creates a more resilient supply chain that is less susceptible to market volatility. By utilizing commodity chemicals like sodium bicarbonate and ferric chloride, manufacturers can secure raw materials from multiple global sources, ensuring continuity of supply even during geopolitical disruptions. Furthermore, the elimination of strict inert atmosphere requirements reduces the dependency on high-purity nitrogen or argon supplies, lowering utility costs and simplifying facility requirements.
- Cost Reduction in Manufacturing: The economic impact of this process is driven primarily by the substitution of expensive catalysts with abundant iron salts and the simplification of reaction conditions. Eliminating the need for specialized anhydrous solvents and inert gas purging significantly lowers the cost of goods sold (COGS). Additionally, the high yields reported across diverse substrates minimize raw material waste, further enhancing the overall process economics. This cost structure allows for competitive pricing strategies in the generic API market while maintaining healthy margins for innovator drugs.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions means that production schedules are less likely to be disrupted by equipment failures related to moisture or oxygen ingress. The use of stable, shelf-stable starting materials reduces inventory spoilage risks and simplifies warehousing logistics. This reliability is crucial for meeting just-in-time delivery commitments to downstream pharmaceutical clients who operate on tight production timelines. The ability to source precursors like acyl chlorides and hydrazides from established industrial suppliers further strengthens the supply chain integrity.
- Scalability and Environmental Compliance: Scaling this process from laboratory to commercial production is straightforward due to the absence of hazardous reagents and extreme pressure conditions. The simplified workup procedure, involving filtration and standard chromatography, reduces the volume of organic waste generated per kilogram of product. This aligns with increasingly stringent environmental regulations regarding solvent usage and waste disposal. The process inherently supports green chemistry initiatives by maximizing atom economy and minimizing the E-factor, making it an attractive option for companies aiming to reduce their carbon footprint.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and scope defined in the patent documentation, providing clarity for technical teams evaluating this route for potential integration into their manufacturing portfolios.
Q: Does this synthesis require strict anhydrous or oxygen-free conditions?
A: No, a key advantage of the method described in patent CN111978265B is that it operates effectively under air and does not require rigorous anhydrous conditions, significantly simplifying operational complexity.
Q: What is the substrate scope for the R1 and R2 groups in this triazole synthesis?
A: The method demonstrates broad tolerance, accommodating substituted or unsubstituted aryl groups for R1 and alkyl, alkenyl, or aryl groups for R2, including functional groups like methyl, methoxy, halogens, and trifluoromethyl.
Q: How is the final product purified after the reaction?
A: Post-processing involves simple filtration followed by silica gel mixing and standard column chromatography purification, avoiding complex extraction or distillation steps often required in traditional methods.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5-Trifluoromethyl-1,2,4-Triazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic value of efficient heterocycle synthesis in accelerating drug development pipelines. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from benchtop discovery to industrial manufacturing is seamless. We adhere to stringent purity specifications and utilize rigorous QC labs to guarantee that every batch of 5-trifluoromethyl-1,2,4-triazole derivative meets the highest international standards. Our commitment to quality assurance ensures that our clients receive materials that are ready for immediate use in critical synthetic sequences.
We invite you to collaborate with us to leverage this advanced synthetic technology for your next project. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our capabilities can optimize your supply chain and reduce time-to-market for your vital pharmaceutical products.
