Advanced Trifluoromethyl Reagent Synthesis for Commercial Pharmaceutical Intermediates Production
The integration of fluorine atoms into organic molecules represents a cornerstone of modern medicinal chemistry, with statistics indicating that a significant percentage of approved pharmaceuticals contain at least one fluorine atom to enhance metabolic stability and bioavailability. Within this critical domain, the trifluoromethyl group stands out as a particularly valuable moiety, featured in blockbuster drugs such as Celecoxib and Ponatinib for treating various cancers and inflammatory conditions. 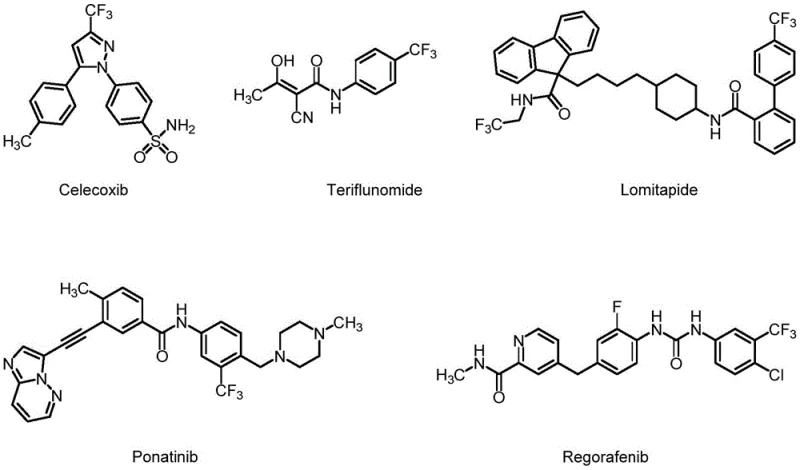 . Recent intellectual property developments, specifically patent CN113861240A published in late 2021, have introduced a novel trifluoromethyl reagent that addresses longstanding challenges in introducing this group efficiently. This technical insight report analyzes the synthesis and application of trifluoromethyl diphenyl methyl phosphine iodide, offering a strategic perspective for R&D and procurement leaders seeking reliable trifluoromethyl reagent suppliers. The disclosed technology promises to streamline the production of high-purity trifluoromethylated intermediates, which are essential for the next generation of agrochemical and pharmaceutical active ingredients.
. Recent intellectual property developments, specifically patent CN113861240A published in late 2021, have introduced a novel trifluoromethyl reagent that addresses longstanding challenges in introducing this group efficiently. This technical insight report analyzes the synthesis and application of trifluoromethyl diphenyl methyl phosphine iodide, offering a strategic perspective for R&D and procurement leaders seeking reliable trifluoromethyl reagent suppliers. The disclosed technology promises to streamline the production of high-purity trifluoromethylated intermediates, which are essential for the next generation of agrochemical and pharmaceutical active ingredients.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the introduction of trifluoromethyl groups into complex organic scaffolds has been plagued by significant technical and safety hurdles that impede efficient commercial scale-up of complex polymer additives and pharmaceutical intermediates. Traditional methodologies often rely on extremely harsh reaction conditions involving hazardous gaseous reagents such as hydrogen fluoride or sulfur tetrafluoride, which require specialized corrosion-resistant equipment and rigorous safety protocols. Furthermore, many existing electrophilic or nucleophilic trifluoromethylating agents suffer from limited substrate scope, poor stability during storage, or exorbitant costs that render them impractical for large-scale manufacturing processes. The reliance on transition metal catalysts in some modern approaches also introduces the risk of heavy metal contamination, necessitating expensive and time-consuming purification steps to meet stringent regulatory purity specifications for drug substances. These cumulative factors create substantial bottlenecks in the supply chain, leading to extended lead times and inflated production costs for downstream manufacturers.
The Novel Approach
In stark contrast to these legacy methods, the technology described in patent CN113861240A utilizes a straightforward addition reaction between diphenyl trifluoromethyl phosphine and methyl iodide to generate the target phosphonium salt. 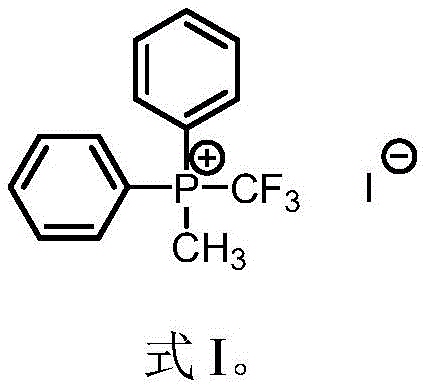 . This novel approach eliminates the need for hazardous gaseous fluorine sources and operates effectively in common organic solvents such as toluene or chlorobenzene at moderate temperatures ranging from 60°C to 120°C. The simplicity of the reaction setup allows for easy monitoring and control, while the resulting reagent exhibits remarkable stability and versatility in subsequent transformations. By avoiding complex catalytic systems and dangerous reagents, this method significantly reduces the operational complexity and safety risks associated with trifluoromethylation, thereby offering a robust pathway for cost reduction in pharmaceutical intermediates manufacturing. The ability to prepare this reagent in ten-gram quantities with high yield demonstrates its immediate potential for scaling to industrial levels without compromising on safety or efficiency.
. This novel approach eliminates the need for hazardous gaseous fluorine sources and operates effectively in common organic solvents such as toluene or chlorobenzene at moderate temperatures ranging from 60°C to 120°C. The simplicity of the reaction setup allows for easy monitoring and control, while the resulting reagent exhibits remarkable stability and versatility in subsequent transformations. By avoiding complex catalytic systems and dangerous reagents, this method significantly reduces the operational complexity and safety risks associated with trifluoromethylation, thereby offering a robust pathway for cost reduction in pharmaceutical intermediates manufacturing. The ability to prepare this reagent in ten-gram quantities with high yield demonstrates its immediate potential for scaling to industrial levels without compromising on safety or efficiency.
Mechanistic Insights into Trifluoromethyl Diphenyl Methyl Phosphine Iodide Reactivity
The chemical versatility of this new reagent lies in its unique ability to function dually as both a radical and a nucleophilic trifluoromethyl source, depending on the specific reaction conditions employed. In radical pathways, the reagent can be activated under visible light irradiation, specifically using blue light at 450 nm, to generate trifluoromethyl radicals that add efficiently to electron-deficient alkenes such as enamides and acryloyl benzamides. This photocatalytic mode of action enables the construction of complex heterocyclic systems like oxindoles and isoquinolinediones, which are privileged structures in drug discovery, without the need for expensive external photocatalysts. Conversely, in the presence of a mild base such as cesium carbonate or DBU, the reagent releases a trifluoromethyl anion equivalent that attacks electrophilic carbonyl centers. 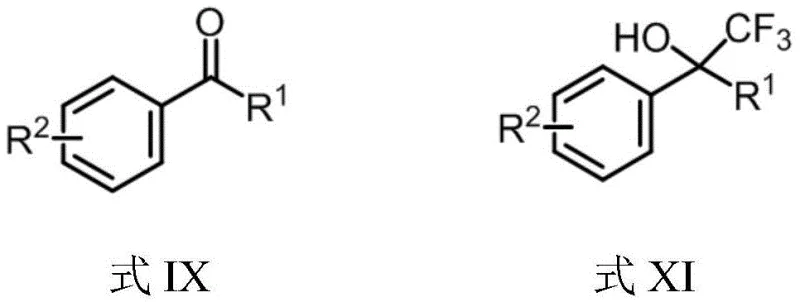 . This nucleophilic pathway is particularly valuable for synthesizing trifluoromethylated alcohols from readily available aldehydes and ketones, expanding the chemical space accessible to process chemists. The mechanistic flexibility ensures that a single reagent inventory can support multiple synthetic routes, optimizing warehouse management and reducing the need for diverse specialized reagents.
. This nucleophilic pathway is particularly valuable for synthesizing trifluoromethylated alcohols from readily available aldehydes and ketones, expanding the chemical space accessible to process chemists. The mechanistic flexibility ensures that a single reagent inventory can support multiple synthetic routes, optimizing warehouse management and reducing the need for diverse specialized reagents.
From an impurity control perspective, the mechanism of this reaction minimizes the formation of difficult-to-remove byproducts that often plague traditional fluorination chemistry. The phosphine oxide byproducts generated during the trifluoromethyl transfer are typically polar and can be easily separated from the non-polar organic products via standard aqueous workup or column chromatography. This inherent cleanliness of the reaction profile is critical for maintaining high purity standards required by regulatory bodies for pharmaceutical ingredients. Moreover, the reaction conditions are mild enough to preserve sensitive functional groups elsewhere in the molecule, preventing degradation or side reactions that could lower overall yield. The precise control over the trifluoromethyl incorporation allows for the consistent production of materials with well-defined impurity profiles, facilitating smoother regulatory filings and reducing the risk of batch rejection. Such predictability is a key factor for supply chain heads when evaluating the reliability of a new synthetic route for long-term production commitments.
How to Synthesize Trifluoromethyl Diphenyl Methyl Phosphine Iodide Efficiently
The synthesis protocol outlined in the patent provides a clear and reproducible method for generating the core reagent, serving as a foundational step for downstream applications in fine chemical synthesis. The process begins with the precise weighing of diphenyl trifluoromethyl phosphine and methyl iodide, which are combined in a dried reaction vessel under an inert atmosphere to prevent moisture interference. The mixture is then heated in a suitable solvent like toluene, where the temperature is carefully maintained to drive the addition reaction to completion over a period of 24 to 48 hours. Detailed standardized synthesis steps are provided below to ensure technical teams can replicate the high yields reported in the patent examples.
- Prepare the reaction vessel by drying thoroughly and adding diphenyl trifluoromethyl phosphine and methyl iodide in a 1: 2 molar ratio within an organic solvent like toluene.
- Heat the mixture to a temperature range of 60-120°C and maintain stirring for 24 to 48 hours to facilitate the addition reaction.
- Remove the solvent and excess methyl iodide via rotary evaporation to isolate the yellow solid trifluoromethyl diphenyl methyl phosphine iodide.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel trifluoromethylation technology presents a compelling opportunity to optimize cost structures and enhance supply resilience. The elimination of hazardous gaseous reagents and complex catalytic systems translates directly into reduced capital expenditure on specialized safety infrastructure and waste treatment facilities. Furthermore, the use of commodity chemicals like methyl iodide and common organic solvents ensures that raw material sourcing remains stable and unaffected by the volatility often seen with exotic fluorine sources. This stability is crucial for maintaining continuous production schedules and avoiding costly downtime caused by material shortages. The simplified workflow also reduces labor hours required for reaction monitoring and workup, contributing to overall operational efficiency and lower manufacturing overheads.
- Cost Reduction in Manufacturing: The streamlined synthesis route eliminates the need for expensive transition metal catalysts and the subsequent rigorous purification steps required to remove trace heavy metals from the final product. By utilizing a metal-free or low-metal approach, manufacturers can achieve substantial cost savings on both reagent procurement and analytical testing for residual metals. Additionally, the high yield reported in the patent examples suggests that less raw material is wasted per unit of product formed, further driving down the cost of goods sold. The ability to operate at moderate temperatures also reduces energy consumption compared to processes requiring cryogenic conditions or extreme heating, adding another layer of economic benefit to the overall production budget.
- Enhanced Supply Chain Reliability: Sourcing strategies benefit significantly from the use of widely available starting materials such as diphenyl trifluoromethyl phosphine and methyl iodide, which are produced by multiple global suppliers. This diversification of the supply base mitigates the risk of single-source dependency and ensures that production can continue even if one vendor faces disruptions. The robustness of the reaction conditions means that the process is less sensitive to minor variations in raw material quality, reducing the rate of batch failures and ensuring consistent output. For supply chain heads, this reliability translates into more accurate forecasting and the ability to commit to longer-term delivery schedules with confidence, strengthening relationships with downstream pharmaceutical clients.
- Scalability and Environmental Compliance: The process is inherently scalable, moving seamlessly from gram-scale laboratory experiments to multi-kilogram pilot runs and eventually to ton-scale commercial production without fundamental changes to the chemistry. The absence of highly toxic gases simplifies environmental permitting and reduces the burden on exhaust gas treatment systems, aligning with increasingly stringent global environmental regulations. Waste streams are easier to manage and treat, lowering disposal costs and minimizing the environmental footprint of the manufacturing site. This alignment with green chemistry principles not only ensures compliance but also enhances the corporate sustainability profile, which is becoming a key differentiator in B2B procurement decisions for major multinational corporations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this trifluoromethyl reagent technology in industrial settings. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation, providing a factual basis for decision-making. Understanding these aspects helps stakeholders evaluate the feasibility of integrating this new reagent into their existing manufacturing portfolios.
Q: What are the primary advantages of this trifluoromethyl reagent over conventional methods?
A: Unlike conventional methods requiring harsh conditions or hazardous gases like HF, this reagent offers a simpler process with high yield and operates under milder thermal conditions, significantly improving operational safety and scalability.
Q: Can this reagent be used for both radical and nucleophilic reactions?
A: Yes, the patent demonstrates dual functionality, allowing the reagent to serve as both a radical source under visible light irradiation and a nucleophilic source in the presence of a base, broadening its synthetic utility.
Q: Is the synthesis process suitable for large-scale commercial production?
A: The method is designed for scalability, utilizing common organic solvents and straightforward workup procedures like filtration and evaporation, which facilitates transition from gram-level laboratory synthesis to ton-scale manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Trifluoromethyl Diphenyl Methyl Phosphine Iodide Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this advanced trifluoromethylation technology for the global pharmaceutical and fine chemical industries. As a seasoned CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory discoveries are successfully translated into robust industrial processes. Our facility is equipped with state-of-the-art rigorous QC labs capable of meeting stringent purity specifications required for GMP-grade intermediates. We are committed to delivering high-purity trifluoromethyl reagents that empower our clients to accelerate their drug development timelines while maintaining the highest standards of quality and safety.
We invite procurement leaders and technical directors to engage with our technical procurement team to discuss how this specific reagent can optimize your current synthesis routes. By requesting a Customized Cost-Saving Analysis, you can gain detailed insights into the potential economic benefits of switching to this novel methodology for your specific projects. We encourage you to contact us to obtain specific COA data and route feasibility assessments tailored to your target molecules. Let us collaborate to engineer a more efficient and cost-effective supply chain for your critical fluorinated building blocks.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
