Advanced Beta-Lactam Route for Larotaxel: Enhancing Scalability and Purity for Global API Manufacturing
Introduction to Next-Generation Taxane Synthesis
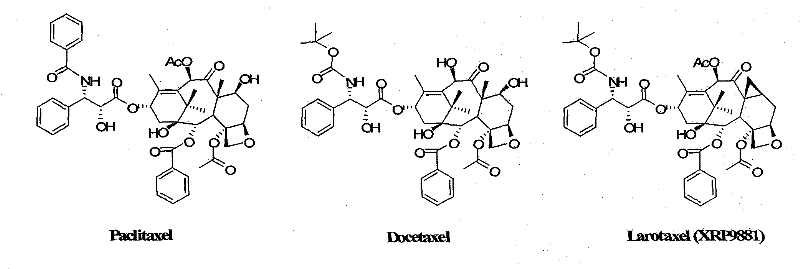
The pharmaceutical landscape for oncology treatments continues to evolve with the development of second-generation taxanes that offer improved efficacy and reduced toxicity profiles compared to first-generation agents like Paclitaxel and Docetaxel. Among these advanced therapeutics, Larotaxel (XRP9881) has emerged as a potent candidate, demonstrating superior activity against multidrug-resistant tumor lines. The technical foundation for producing this critical active pharmaceutical ingredient (API) is detailed in patent CN101863861A, which discloses a simple and highly efficient semisynthetic method. This patent represents a significant leap forward in process chemistry, moving away from labor-intensive chiral resolution methods toward a streamlined beta-lactam approach. By optimizing the construction of the C-13 side chain, the disclosed technology addresses key bottlenecks in yield and purity that have historically constrained the commercial availability of complex taxane analogues. For global supply chain stakeholders, understanding this technological shift is paramount, as it directly influences the reliability and cost structure of high-purity pharmaceutical intermediates required for clinical and commercial drug supply.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of the C-13 side chain for taxane derivatives has relied heavily on the use of optically pure starting materials, such as alpha-phenylethylamine, to establish the necessary stereochemistry early in the sequence. While chemically sound, these conventional routes suffer from severe economic and operational drawbacks that hinder large-scale adoption. The primary disadvantage lies in the exorbitant cost of chiral auxiliaries, which drastically inflates the raw material expenditure for the final API. Furthermore, traditional pathways often involve extended synthetic sequences with multiple protection and deprotection steps, leading to cumulative yield losses at every stage. Purification in these legacy methods frequently depends on column chromatography, a technique that is notoriously difficult to scale, consumes vast quantities of organic solvents, and creates significant waste disposal challenges. The instability of certain intermediates, particularly those involving triflate leaving groups in parent nucleus modifications, further complicates the manufacturing process, requiring stringent temperature controls and specialized handling that increase operational complexity and risk.
The Novel Approach
In stark contrast to these legacy constraints, the methodology outlined in patent CN101863861A introduces a robust and economically viable alternative centered on the [2+2] cycloaddition reaction. This innovative strategy utilizes readily available and inexpensive commodity chemicals, specifically benzaldehyde and p-anisidine, to construct the beta-lactam core. By initially forming a racemic intermediate and relying on downstream stereoselective crystallization and coupling dynamics, the process eliminates the need for costly chiral starting materials. The route is characterized by its brevity and operational simplicity, significantly shortening the overall preparation timeline. Crucially, the purification strategy has been re-engineered to favor recrystallization and precipitation over chromatography, which not only enhances throughput but also aligns with green chemistry principles by reducing solvent waste. This approach ensures that the key intermediates possess higher diastereomeric selectivity, facilitating a more efficient docking with the taxane parent nucleus and ultimately delivering a higher quality final product with improved process mass intensity.
Mechanistic Insights into Beta-Lactam [2+2] Cycloaddition
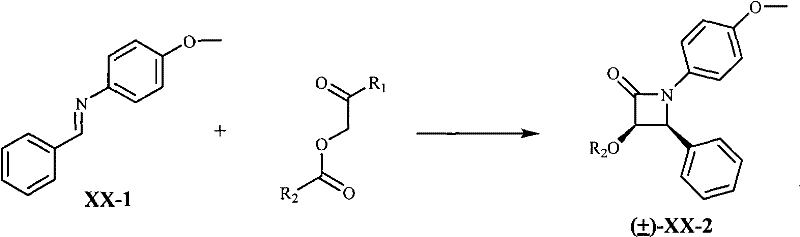
The cornerstone of this optimized synthesis is the formation of the beta-lactam ring via the Staudinger [2+2] cycloaddition reaction. In this critical transformation, a Schiff base derived from benzaldehyde and p-anisidine reacts with an alpha-acyloxy acyl halide in the presence of a tertiary amine base. The reaction proceeds through a ketene intermediate generated in situ, which undergoes a concerted cycloaddition with the imine functionality. Although this step initially yields a racemic mixture of beta-lactam diastereomers, the process design cleverly leverages subsequent purification steps to enrich the desired cis-isomer. The use of specific solvents like dichloromethane and controlled low-temperature conditions (-78°C to 25°C) is vital for maximizing the ratio of the target stereoisomer while minimizing side reactions. This mechanistic pathway is advantageous because it decouples the stereochemical control from the initial raw material cost, allowing manufacturers to use achiral, bulk chemicals while still achieving the high optical purity required for biological activity through physical separation methods later in the sequence.
Following the formation of the beta-lactam core, the synthesis employs a sophisticated protecting group strategy to ensure high fidelity during the final coupling stage. The hydroxyl group at the C-3 position of the beta-lactam is protected as a bulky silyl ether, typically using tert-butyldimethylsilyl (TBS) chloride, while the nitrogen atom is protected with a tert-butoxycarbonyl (Boc) group. These bulky substituents play a dual role: they protect sensitive functional groups from unwanted side reactions and, more importantly, exert significant steric influence during the nucleophilic attack on the taxane parent nucleus. When the side chain anion, generated using a strong base like LiHMDS, attacks the C-13 position of the Larotaxel parent nucleus (compound A-3), the steric bulk of the TBS and Boc groups directs the approach of the nucleophile, enforcing a high degree of diastereoselectivity. This "steric steering" mechanism ensures that the coupling reaction predominantly yields the biologically active (2'R, 3'S) configuration, effectively resolving the racemic nature of the earlier intermediate without the need for complex chiral chromatography. The final deprotection step using fluoride sources cleanly removes the silyl group to reveal the active hydroxyl moiety, completing the assembly of the Larotaxel molecule with high structural integrity.
How to Synthesize Larotaxel Efficiently
The synthesis of Larotaxel via this optimized beta-lactam route involves a convergent strategy where the side chain and the parent nucleus are prepared separately and then coupled in the final stages. The process begins with the condensation of benzaldehyde and p-anisidine to form the Schiff base, followed by cycloaddition to generate the beta-lactam ring. Subsequent steps involve selective hydrolysis, silylation, and oxidative removal of the p-methoxyphenyl group to install the necessary Boc and TBS protecting groups. Parallel to this, the Larotaxel parent nucleus is derived from 10-DAB through selective acetylation and cyclization to form the oxetane ring. The two fragments are then joined using lithium hexamethyldisilazide (LiHMDS) as the base to facilitate the esterification at the C-13 position. Detailed standard operating procedures for each reaction step, including specific molar ratios, temperature profiles, and workup protocols, are provided in the structured guide below to ensure reproducibility and safety in a GMP environment.
- Condense benzaldehyde and p-anisidine to form Schiff base XX-1, followed by [2+2] cycloaddition with alpha-acyloxy acyl halide to generate racemic beta-lactam.
- Perform selective deprotection and silylation to install TBS and Boc protecting groups, yielding the key intermediate (±)-XX-6.
- Couple the side chain intermediate with the Larotaxel parent nucleus (A-3) using LiHMDS, followed by desilylation to obtain the final product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of the synthesis method described in patent CN101863861A offers transformative advantages that extend far beyond simple chemical yield improvements. The shift from expensive chiral starting materials to commodity chemicals like benzaldehyde fundamentally alters the cost basis of the API, insulating the supply chain from the volatility associated with specialty fine chemicals. By eliminating the reliance on optically pure alpha-phenylethylamine, manufacturers can secure raw material supplies from a much broader vendor base, significantly enhancing supply continuity and reducing the risk of production stoppages due to single-source dependencies. Furthermore, the simplification of purification processes—replacing column chromatography with crystallization and precipitation—drastically reduces the consumption of organic solvents and silica gel. This reduction in material usage translates directly into lower waste disposal costs and a smaller environmental footprint, which is increasingly critical for meeting modern regulatory and sustainability standards in pharmaceutical manufacturing.
- Cost Reduction in Manufacturing: The economic impact of this process is driven by the substitution of high-cost chiral auxiliaries with low-cost achiral building blocks. In traditional synthesis, the cost of the chiral pool starting material can constitute a significant portion of the total COGS; replacing this with benzaldehyde and p-anisidine results in substantial raw material savings. Additionally, the avoidance of column chromatography reduces the operational expenditure related to solvent purchase, recovery, and hazardous waste treatment. The higher overall yield resulting from fewer synthetic steps and more robust purification further amplifies these cost benefits, allowing for a more competitive pricing structure for the final Larotaxel API without compromising on quality margins.
- Enhanced Supply Chain Reliability: Supply chain resilience is significantly bolstered by the use of universally available starting materials. Benzaldehyde and p-anisidine are produced on a massive industrial scale for various applications, ensuring that supply shortages are highly unlikely even during periods of global market stress. This contrasts sharply with specialized chiral intermediates, which often have limited production capacity and long lead times. The robustness of the synthetic route also means that the process is less sensitive to minor variations in reaction conditions, leading to more consistent batch-to-batch quality. This reliability allows supply chain planners to maintain leaner inventory levels while still guaranteeing on-time delivery to downstream formulation partners, thereby optimizing working capital and reducing storage costs.
- Scalability and Environmental Compliance: The design of this synthesis route is inherently scalable, addressing one of the most common failure points in translating laboratory chemistry to commercial production. Techniques such as precipitation and recrystallization are easily adapted to large-scale reactors, unlike chromatography which poses significant engineering challenges at the multi-kilogram or ton scale. The process also generates less hazardous waste due to the reduced solvent load and the elimination of silica gel disposal. This alignment with green chemistry principles facilitates smoother regulatory approvals and reduces the burden on environmental health and safety teams. The ability to run larger batch sizes efficiently means that manufacturers can respond more agilely to surges in demand, ensuring a stable supply of this critical oncology medication to patients worldwide.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of Larotaxel using the beta-lactam methodology. These insights are derived directly from the technical specifications and experimental data provided in the patent literature, offering clarity on the feasibility and advantages of this specific synthetic route. Understanding these details is essential for technical teams evaluating potential manufacturing partners or considering technology transfer opportunities. The answers highlight the balance between chemical innovation and practical manufacturing constraints, demonstrating how modern process chemistry solves historical bottlenecks in complex molecule synthesis.
Q: How does the new beta-lactam method improve cost efficiency compared to traditional chiral auxiliary routes?
A: The novel method utilizes inexpensive starting materials like benzaldehyde and p-anisidine instead of costly optically pure alpha-phenylethylamine. Furthermore, it replaces complex column chromatography purification with simple recrystallization and precipitation steps, significantly reducing solvent consumption and processing time.
Q: What ensures the high diastereoselectivity during the coupling of the racemic side chain?
A: High cis-selectivity is achieved through the steric hindrance provided by the bulky tert-butyldimethylsilyl (TBS) ether protecting group at the C-3 position and the tert-butoxycarbonyl (Boc) group at the N-4 position of the beta-lactam ring during the ring-opening docking reaction with the parent nucleus.
Q: Is this synthesis route suitable for large-scale industrial production?
A: Yes, the process is specifically designed for scalability. It features shorter reaction sequences, avoids unstable intermediates like triflates where possible, and employs robust purification techniques like precipitation and recrystallization that are easily transferable from laboratory to multi-ton manufacturing scales.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Larotaxel Supplier
As the demand for next-generation taxanes continues to grow, partnering with a manufacturer that possesses deep technical expertise in complex semi-synthesis is critical for success. NINGBO INNO PHARMCHEM stands at the forefront of this field, leveraging advanced process technologies like the beta-lactam route to deliver high-purity Larotaxel intermediates and APIs. Our facility is equipped with state-of-the-art reactor systems capable of handling the stringent temperature and atmosphere controls required for [2+2] cycloadditions and organometallic couplings. We pride ourselves on our extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with consistency and precision. Our rigorous QC labs employ advanced analytical techniques to verify stringent purity specifications, guaranteeing that every batch meets the exacting standards required for clinical and commercial pharmaceutical applications.
We invite you to collaborate with us to optimize your supply chain for Larotaxel and related taxane derivatives. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. By leveraging our optimized synthetic routes, we can help you reduce lead times and lower total acquisition costs. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us demonstrate how our manufacturing excellence can support your long-term strategic goals in the oncology sector.
