Scalable Manufacturing of S-N-Protected Azetidine-2-Carboxylic Acid via Novel Reduction Pathways
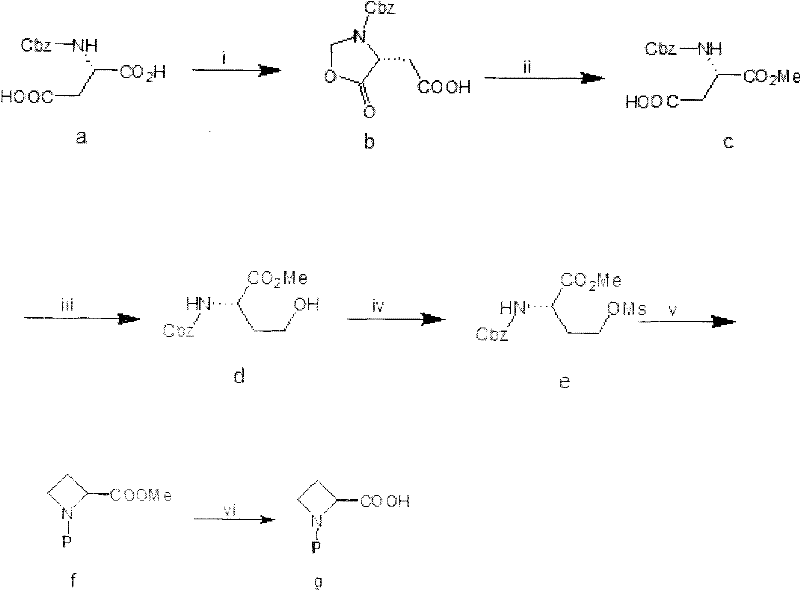
The pharmaceutical industry continuously seeks robust and scalable pathways for constructing constrained amino acid scaffolds, particularly azetidine derivatives which serve as critical building blocks for protease inhibitors and peptidomimetics. Patent CN102477007A introduces a transformative methodology for the preparation of S-N-protecting group-azetidine-2-carboxylic acid, addressing longstanding challenges associated with safety and cost in traditional synthetic routes. By utilizing N-protected-L-aspartic acid as a readily available chiral pool starting material, this novel process circumvents the need for hazardous reducing agents and complex purification steps that have historically plagued the manufacturing of these high-value intermediates. The strategic implementation of an oxazolidinone masking group allows for precise differentiation between carboxyl functionalities, enabling a streamlined sequence that culminates in the formation of the strained four-membered azetidine ring with high stereochemical integrity. This technological advancement represents a significant leap forward for reliable pharmaceutical intermediate supplier networks aiming to secure stable supply chains for complex heterocyclic acids.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of azetidine-2-carboxylic acid derivatives has been fraught with significant operational hazards and economic inefficiencies that hinder large-scale adoption. Traditional routes often rely heavily on lithium aluminum hydride (LiAlH4) for the reduction of ester or acid functionalities to the corresponding alcohols, a reagent known for its extreme pyrophoric nature and stringent requirement for anhydrous conditions. The handling of LiAlH4 necessitates specialized equipment, rigorous moisture control, and complex quenching procedures, all of which drastically inflate production costs and introduce substantial safety risks to the manufacturing facility. Furthermore, existing methodologies frequently suffer from poor selectivity, generating difficult-to-remove by-products that require labor-intensive column chromatography for purification, a technique that is notoriously impractical for ton-scale chemical production. These legacy processes also often involve multiple protection and deprotection steps that erode overall yield and generate excessive chemical waste, conflicting with modern green chemistry principles and increasing the environmental burden on cost reduction in pharmaceutical intermediates manufacturing.
The Novel Approach
The methodology disclosed in CN102477007A fundamentally reengineers the synthetic landscape by replacing dangerous reagents with benign, industrially viable alternatives while maintaining high stereocontrol. A pivotal innovation in this route is the substitution of lithium aluminum hydride with sodium borohydride (NaBH4) for the critical reduction step, facilitated by the activation of the carboxyl group as a mixed anhydride. This switch not only eliminates the fire hazards associated with hydride reagents but also allows the reaction to proceed in protic or semi-protic solvents like methanol and THF, significantly simplifying the operational protocol. The process leverages a clever cyclization strategy where paraformaldehyde reacts with the starting amino acid to form a transient oxazolidinone ring, effectively differentiating the two carboxyl groups without the need for additional protecting group manipulations. Subsequent mesylation and intramolecular nucleophilic substitution under basic conditions facilitate the closure of the four-membered azetidine ring with excellent efficiency. This approach ensures that the production of high-purity pharmaceutical intermediates can be achieved with simplified work-up procedures, such as extraction and crystallization, rather than chromatography, thereby enhancing throughput and economic viability.
Mechanistic Insights into Chemoselective Reduction and Cyclization
The core mechanistic brilliance of this synthesis lies in the chemoselective reduction of the mono-esterified intermediate using sodium borohydride, a transformation that is typically challenging due to the presence of other reducible functionalities like carbamates. In this specific pathway, the carboxyl group of the mono-methyl ester is first activated using ethyl chloroformate in the presence of an organic base to form a reactive mixed anhydride species. This activation lowers the energy barrier for hydride attack, allowing the relatively mild sodium borohydride to reduce the activated carbonyl to a primary alcohol while leaving the methyl ester and the Cbz-protected amine intact. This selectivity is crucial for preserving the chiral integrity of the molecule and avoiding side reactions that could lead to racemization or over-reduction. The resulting hydroxy-ester intermediate is then subjected to mesylation, converting the hydroxyl group into an excellent leaving group (mesylate), which sets the stage for the final ring-closing event. The mechanistic pathway ensures that impurities arising from non-selective reduction are minimized, leading to a cleaner crude product profile that is essential for downstream processing in API synthesis.
Furthermore, the cyclization mechanism involves a tandem deprotection and intramolecular substitution sequence that elegantly constructs the azetidine core. Following the removal of the Cbz protecting group via catalytic hydrogenation, the liberated amine acts as a nucleophile that attacks the carbon bearing the mesylate group. This intramolecular SN2 reaction is driven by the formation of the thermodynamically stable, albeit strained, four-membered ring system. The reaction conditions are carefully tuned to promote this cyclization over intermolecular polymerization or elimination side reactions, often utilizing bicarbonate bases in alcoholic solvents to maintain a pH that favors nucleophilic attack without causing ester hydrolysis prematurely. This precise control over the reaction environment ensures that the final commercial scale-up of complex pharmaceutical intermediates proceeds with high fidelity, minimizing the formation of oligomeric by-products and ensuring that the stereochemistry at the alpha-carbon is retained throughout the transformation.
How to Synthesize S-N-Protected Azetidine-2-Carboxylic Acid Efficiently
The execution of this synthesis requires careful attention to the activation steps and the sequential addition of reagents to maximize yield and safety. The process begins with the condensation of N-Cbz-L-aspartic acid with paraformaldehyde to generate the cyclic oxazolidinone intermediate, followed by selective esterification. The critical reduction step involves the generation of the mixed anhydride at low temperatures prior to the addition of the borohydride source. Detailed standard operating procedures regarding stoichiometry, temperature profiles, and quenching protocols are essential for reproducibility. For the complete technical breakdown and standardized synthesis steps, please refer to the guide below.
- Cyclization of N-Cbz-L-aspartic acid with paraformaldehyde to form the oxazolidinone intermediate.
- Mono-esterification followed by chemoselective reduction of the carboxyl group using sodium borohydride.
- Mesylation of the hydroxyl group, followed by catalytic hydrogenation and base-mediated cyclization to form the azetidine ring.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the adoption of this novel synthetic route offers profound advantages that directly impact the bottom line and operational resilience of chemical manufacturing organizations. By eliminating the dependency on lithium aluminum hydride, companies can significantly reduce the costs associated with specialized storage, handling equipment, and safety training, while also lowering the price point of the reducing agent itself. The use of common solvents like toluene, methanol, and ethyl acetate further simplifies the supply chain logistics, as these materials are widely available and do not require the same level of regulatory scrutiny as ether-based solvents often used in traditional hydride reductions. Moreover, the avoidance of column chromatography in favor of extraction and crystallization techniques dramatically increases the throughput capacity of existing manufacturing facilities, allowing for faster batch turnover times and reduced solvent consumption. These factors collectively contribute to a more robust and cost-effective supply chain capable of meeting the demanding volume requirements of the global pharmaceutical market.
- Cost Reduction in Manufacturing: The replacement of expensive and hazardous lithium aluminum hydride with sodium borohydride results in substantial raw material cost savings and reduces the overhead associated with safety compliance. Additionally, the simplified purification process eliminates the need for expensive silica gel and large volumes of chromatographic solvents, leading to a drastic reduction in waste disposal costs and overall production expenses. The ability to perform reactions under less stringent anhydrous conditions also lowers energy consumption related to solvent drying and inert atmosphere maintenance, further enhancing the economic efficiency of the process.
- Enhanced Supply Chain Reliability: Utilizing readily available starting materials like N-Cbz-L-aspartic acid and common reagents ensures a stable supply chain that is less susceptible to disruptions caused by the scarcity of specialized chemicals. The robustness of the reaction conditions means that production can be easily transferred between different manufacturing sites without the need for highly specialized infrastructure, thereby diversifying supply sources and mitigating risk. This reliability is critical for maintaining continuous production schedules for key drug intermediates, ensuring that downstream API manufacturing is not delayed by raw material shortages or quality issues.
- Scalability and Environmental Compliance: The process is inherently designed for scalability, with reaction steps that translate seamlessly from laboratory bench scale to multi-ton industrial reactors. The reduction in hazardous waste generation, particularly the avoidance of aluminum salts and the minimization of solvent usage, aligns with increasingly strict environmental regulations and corporate sustainability goals. This environmental compatibility not only reduces the regulatory burden on manufacturers but also enhances the marketability of the final product to eco-conscious pharmaceutical clients who prioritize green chemistry in their supply chains.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis and application of S-N-protected azetidine-2-carboxylic acid. These answers are derived directly from the technical specifications and advantageous effects described in the patent literature, providing clarity on the feasibility and benefits of this manufacturing route. Understanding these details is vital for stakeholders evaluating the integration of this intermediate into their broader drug development pipelines.
Q: Why is sodium borohydride preferred over lithium aluminum hydride in this synthesis?
A: Sodium borohydride is significantly safer, cheaper, and easier to handle than lithium aluminum hydride. It allows for reduction under milder conditions without requiring strictly anhydrous environments, reducing operational complexity and safety risks in large-scale manufacturing.
Q: What is the primary advantage of the oxazolidinone protection strategy?
A: The formation of the five-membered oxazolidinone ring selectively protects one carboxyl group while leaving the other available for functionalization. This avoids the need for complex orthogonal protection schemes and simplifies the purification process.
Q: Is this process suitable for multi-kilogram production?
A: Yes, the patent explicitly highlights the use of common industrial reagents and mild reaction conditions. The elimination of hazardous reagents like lithium aluminum hydride and the avoidance of column chromatography make it highly suitable for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable S-N-Protected Azetidine-2-Carboxylic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-quality intermediates in the development of next-generation therapeutics, and we are uniquely positioned to support your needs with our advanced manufacturing capabilities. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements with consistent quality and timely delivery. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of S-N-protected azetidine-2-carboxylic acid meets the highest industry standards, providing you with the confidence needed to advance your clinical programs. Our commitment to process optimization allows us to offer competitive pricing without compromising on the integrity of the final product.
We invite you to collaborate with us to leverage these technological advancements for your specific projects. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your volume needs. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our optimized synthesis of S-N-protected azetidine-2-carboxylic acid can enhance the efficiency and profitability of your supply chain.
