Scalable Synthesis of Cabazitaxel Core via Tandem Pummerer-Swern Oxidation for Global Pharma Supply
Introduction to Advanced Taxane Synthesis
The global demand for second-generation taxane anticancer agents continues to surge, driven by the critical need to overcome multidrug resistance (MDR) in oncology treatments. Patent CN103012330A introduces a groundbreaking preparation method for Cabazitaxel (XRP6258), a potent semi-synthetic analog of docetaxel designed specifically to retain efficacy against P-glycoprotein overexpressing tumor cells. Unlike conventional approaches that struggle with low regioselectivity and cumbersome protection strategies, this invention leverages a sophisticated tandem Pummerer rearrangement and Swern oxidation protocol. By utilizing 10-deacetylbaccatin III (10-DAB) as the starting material, the process achieves high-yield dual functionalization at the C-7 and C-10 positions while simultaneously oxidizing the C-13 hydroxyl group. This technical breakthrough not only simplifies the synthetic pathway but also establishes a robust foundation for the reliable supply of high-purity pharmaceutical intermediates required for next-generation chemotherapy formulations.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the semi-synthesis of Cabazitaxel has been plagued by significant inefficiencies that hinder cost-effective commercialization. Traditional routes, such as those described in WO9925704, attempt direct dimethyl etherification of 10-DAB using strong bases like sodium hydride and alkylating agents such as methyl iodide. However, these methods suffer from severe drawbacks, primarily due to the susceptibility of the C-7 hydroxyl group to retro-aldol condensation and epimerization under strongly basic conditions. Consequently, the conversion rate for C-7 alkylation remains dismally low, often ranging between 10% and 20%, necessitating complex preparative liquid chromatography for purification. Furthermore, alternative multi-step protection strategies involving triethylsilyl groups (as seen in WO9630356) result in excessively long synthetic sequences with total yields frequently falling below 10%. These inefficiencies create substantial bottlenecks in cost reduction in pharmaceutical manufacturing, making large-scale production economically unviable for many suppliers.
The Novel Approach
In stark contrast, the methodology disclosed in CN103012330A offers a paradigm shift by integrating protection and oxidation into a single, highly efficient operation. The core innovation lies in the synchronous execution of Pummerer rearrangement and Swern oxidation using dimethyl sulfoxide (DMSO) and acetic anhydride. This allows for the simultaneous installation of methylthiomethyl (MTM) groups at C-7 and C-10 alongside the oxidation of the C-13 hydroxyl to a ketone, achieving yields upwards of 85% with exceptional regioselectivity. 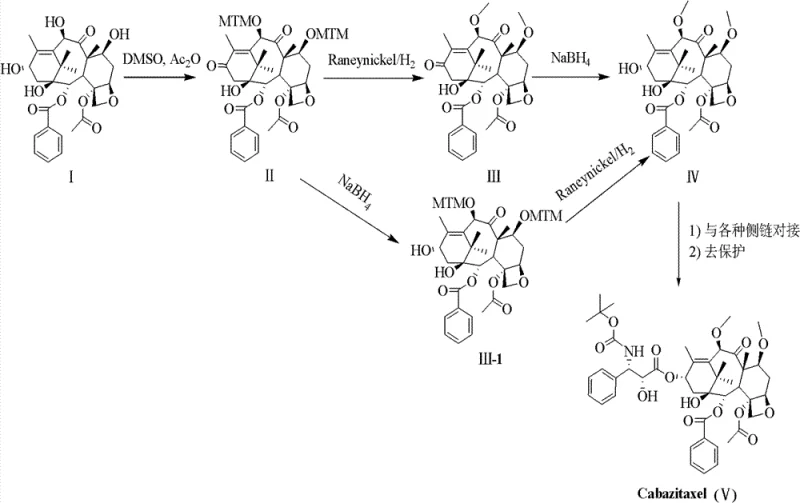 This streamlined approach drastically reduces the number of unit operations and eliminates the need for cryogenic conditions in the early stages. The resulting intermediates can often be carried forward as crude solids after simple workup, bypassing the need for resource-intensive column chromatography. This efficiency translates directly into enhanced supply chain reliability, ensuring that critical oncology intermediates can be produced consistently to meet rigorous global quality standards without the delays associated with low-yield legacy processes.
This streamlined approach drastically reduces the number of unit operations and eliminates the need for cryogenic conditions in the early stages. The resulting intermediates can often be carried forward as crude solids after simple workup, bypassing the need for resource-intensive column chromatography. This efficiency translates directly into enhanced supply chain reliability, ensuring that critical oncology intermediates can be produced consistently to meet rigorous global quality standards without the delays associated with low-yield legacy processes.
Mechanistic Insights into Tandem Pummerer-Swern Functionalization
The chemical elegance of this synthesis is rooted in the dual reactivity of the DMSO/acetic anhydride system. Under acidic catalysis, typically using acetic acid or p-toluenesulfonic acid at temperatures between 25°C and 60°C, the reaction initiates a Pummerer rearrangement. This mechanism facilitates the nucleophilic attack of the C-7 and C-10 hydroxyl groups on the activated sulfur species, installing the MTM protecting groups with high fidelity. Concurrently, the system acts as a Swern-type oxidant for the C-13 secondary alcohol. The activation of DMSO generates a reactive sulfonium ion which, upon interaction with the C-13 hydroxyl and subsequent base-mediated elimination, converts the alcohol into a ketone. 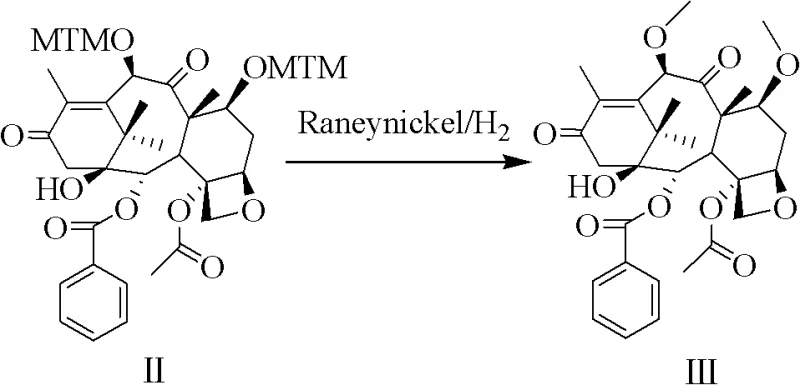 This tandem transformation is critical because it sets the stage for the subsequent stereocontrolled reduction. By oxidizing C-13 early in the sequence, the synthesis avoids the complications of differentiating between multiple hydroxyl groups later in the pathway. The use of MTM groups is particularly strategic; they are robust enough to withstand the oxidative conditions yet can be cleanly removed later via catalytic hydrogenolysis using Raney nickel. This mechanistic design ensures that the impurity profile remains manageable, as the high regioselectivity minimizes the formation of positional isomers that are notoriously difficult to separate in taxane chemistry.
This tandem transformation is critical because it sets the stage for the subsequent stereocontrolled reduction. By oxidizing C-13 early in the sequence, the synthesis avoids the complications of differentiating between multiple hydroxyl groups later in the pathway. The use of MTM groups is particularly strategic; they are robust enough to withstand the oxidative conditions yet can be cleanly removed later via catalytic hydrogenolysis using Raney nickel. This mechanistic design ensures that the impurity profile remains manageable, as the high regioselectivity minimizes the formation of positional isomers that are notoriously difficult to separate in taxane chemistry.
Following the formation of the key intermediate II, the process employs a gentle yet effective deprotection and reduction sequence. The removal of the MTM groups is achieved using Raney nickel under a hydrogen atmosphere, a method chosen for its scalability and safety profile compared to harsher chemical demethylation reagents. Subsequently, the C-13 ketone is reduced back to the hydroxyl group using sodium borohydride. This reduction step is highly stereoselective, favoring the formation of the desired beta-configured alcohol essential for biological activity. The ability to perform these transformations in common solvents like ethanol or tetrahydrofuran at near-ambient temperatures further underscores the practicality of the route. By controlling the reaction temperature between -10°C and 25°C during the borohydride reduction, the process maximizes the yield of the correct stereoisomer, thereby reducing the burden on downstream purification and ensuring the delivery of high-purity taxane intermediates suitable for final API synthesis.
How to Synthesize Cabazitaxel Core Efficiently
The synthesis of the XRP6258 parent nucleus (Intermediate IV) represents a critical milestone in the production of this life-saving medication. The patented procedure outlines a logical progression from readily available 10-DAB through a series of high-yielding transformations that prioritize operational simplicity. The initial functionalization creates a versatile scaffold that can be coupled with various side chains, offering flexibility in manufacturing. For detailed standard operating procedures, stoichiometry, and specific workup protocols required to replicate this high-efficiency route, please refer to the standardized synthesis guide below.
- Perform simultaneous Pummerer rearrangement and Swern oxidation on 10-deacetylbaccatin III using DMSO and acetic anhydride to yield the C-7/C-10 protected and C-13 oxidized intermediate.
- Execute catalytic hydrogenolysis using Raney nickel to remove methylthiomethyl groups, followed by stereoselective reduction of the C-13 carbonyl with sodium borohydride.
- Couple the resulting core with protected side chains (beta-lactams or oxazolidines) and perform final deprotection to obtain Cabazitaxel.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel synthesis route offers compelling strategic advantages beyond mere technical novelty. The primary value proposition lies in the drastic simplification of the manufacturing workflow, which directly correlates to reduced operational expenditures and improved margin stability. By eliminating multiple protection and deprotection cycles that characterize older methodologies, the process significantly lowers the consumption of solvents, reagents, and labor hours. This lean manufacturing approach mitigates the risk of supply disruptions caused by complex purification bottlenecks, ensuring a more predictable flow of materials for downstream API production. Furthermore, the robustness of the chemistry allows for greater flexibility in sourcing raw materials, as the tolerance for minor variations in starting material quality is higher due to the effectiveness of the recrystallization steps.
- Cost Reduction in Manufacturing: The economic impact of this technology is profound, primarily driven by the elimination of expensive chromatographic purification steps for key intermediates. In traditional routes, the low yields necessitate processing larger volumes of starting material to achieve the same output, inflating costs exponentially. In this new method, the high conversion rates in the initial Pummerer/Swern step mean that less raw material is wasted. Additionally, the use of Raney nickel for desulfurization is a cost-effective alternative to precious metal catalysts or hazardous chemical reagents. The ability to use crude intermediates directly in subsequent reactions without intermediate isolation further compresses the production timeline and reduces utility costs associated with drying and solvent recovery, leading to substantial cost savings in the overall cost of goods sold (COGS).
- Enhanced Supply Chain Reliability: Supply continuity is paramount in the pharmaceutical industry, especially for oncology drugs where patient treatment schedules cannot be interrupted. This synthesis route enhances reliability by shortening the critical path of production. Fewer synthetic steps mean fewer opportunities for batch failures or quality deviations. The reagents employed, such as DMSO, acetic anhydride, and sodium borohydride, are commodity chemicals with stable global supply chains, reducing the risk of vendor lock-in or raw material shortages. Moreover, the simplified purification strategy, relying on recrystallization rather than column chromatography, makes the process inherently more scalable. This scalability ensures that suppliers can rapidly ramp up production volumes to meet sudden spikes in demand without compromising on the stringent purity specifications required for clinical-grade intermediates.
- Scalability and Environmental Compliance: From an environmental and regulatory perspective, this method aligns well with modern green chemistry principles. The reduction in solvent usage and the avoidance of heavy metal contaminants (by using Raney nickel which can be filtered and recycled) simplify waste stream management. The process operates largely at mild temperatures, reducing energy consumption for heating and cooling compared to cryogenic reactions. This lower environmental footprint facilitates easier regulatory approval and compliance with increasingly strict environmental, health, and safety (EHS) regulations. The straightforward nature of the unit operations—mixing, filtering, and crystallizing—makes the technology transfer to large-scale reactors seamless, supporting the commercial scale-up of complex oncology APIs from pilot plant quantities to multi-ton annual production capacities with minimal technical risk.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of this synthesis is vital for stakeholders evaluating its implementation. The following questions address common inquiries regarding the feasibility, purity, and scalability of the Cabazitaxel production method described in CN103012330A. These answers are derived directly from the patent's experimental data and technical disclosures to provide accurate guidance for R&D and procurement teams assessing this technology for their supply chains.
Q: How does this new method improve yield compared to traditional methylation routes?
A: Traditional direct methylation often suffers from low conversion (10-20%) due to C-7 epimerization. This patent utilizes a tandem Pummerer/Swern strategy that achieves high regioselectivity and yields exceeding 85% in the initial functionalization step, significantly boosting overall throughput.
Q: Is the process suitable for large-scale industrial manufacturing?
A: Yes, the method eliminates the need for complex multi-step column chromatography purifications for key intermediates. Operations such as recrystallization and filtration are prioritized, making the route highly amenable to commercial scale-up from 100 kgs to multi-ton production.
Q: What are the key cost-saving drivers in this synthesis?
A: Cost efficiency is driven by the use of inexpensive reagents like DMSO and acetic anhydride for dual functionalization, the avoidance of expensive silyl protecting groups in early stages, and the ability to use crude intermediates directly in subsequent steps without purification.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Cabazitaxel Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory innovation to commercial reality requires a partner with deep technical expertise and robust manufacturing capabilities. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising efficiencies of the Pummerer-based Cabazitaxel synthesis can be fully realized in an industrial setting. We maintain stringent purity specifications across all our product lines, supported by rigorous QC labs equipped with state-of-the-art analytical instrumentation to verify identity, potency, and impurity profiles. Our commitment to quality assurance means that every batch of high-purity taxane intermediates we deliver meets the exacting standards required by global regulatory bodies, providing you with the confidence needed to advance your drug development programs.
We invite you to collaborate with us to optimize your supply chain for next-generation anticancer therapeutics. By leveraging our advanced process chemistry capabilities, we can help you navigate the complexities of taxane synthesis and achieve significant operational efficiencies. Please contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our manufacturing excellence can support your strategic goals in the competitive oncology market.
