Advanced Palladium-Catalyzed Kinetic Resolution for High-Purity Chiral Cyclophane Intermediates
Advanced Palladium-Catalyzed Kinetic Resolution for High-Purity Chiral Cyclophane Intermediates
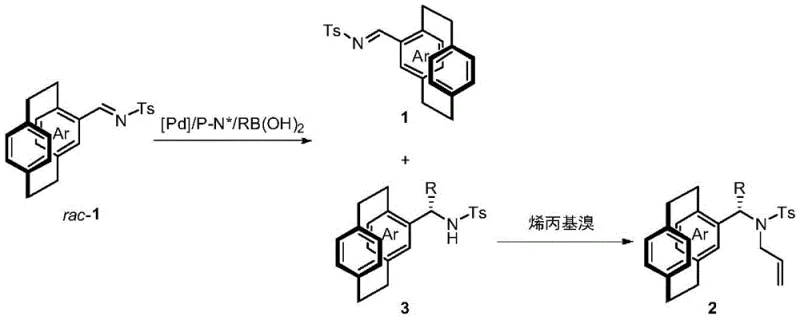
The field of asymmetric synthesis continues to evolve with the demand for highly enantioenriched building blocks, particularly those possessing planar chirality which are indispensable for advanced material science and pharmaceutical applications. Patent CN112538033A introduces a groundbreaking resolution method for naphthenic face chiral compounds, specifically targeting the challenging class of [2.2]paracyclophane derivatives. This technology leverages a sophisticated palladium-catalyzed homogeneous system utilizing chiral phosphine-nitrogen complexes to achieve kinetic resolution through an addition reaction with boronic acids. The significance of this innovation lies in its ability to simultaneously construct central chirality while resolving planar chirality, yielding products with exceptional optical purity. For R&D directors and process chemists, this represents a pivotal shift from traditional stoichiometric resolutions to catalytic processes that offer superior atom economy and operational simplicity. The method not only provides access to valuable chiral cyclophane imines but also their corresponding amine derivatives, which serve as critical precursors for chiral ligands and auxiliaries in complex organic synthesis.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the acquisition of optically pure [2.2]cyclophane compounds has been fraught with significant technical and economic hurdles that hinder efficient commercial manufacturing. Traditional approaches predominantly rely on classical chemical resolution using stoichiometric amounts of chiral resolving agents or preparative chiral chromatography, both of which suffer from inherent inefficiencies. Classical resolution is theoretically limited to a maximum yield of 50% for the desired enantiomer, necessitating the recycling or disposal of the unwanted isomer, which drastically impacts overall process mass intensity and cost efficiency. Furthermore, chiral chromatographic separation, while effective for analytical purposes, often proves prohibitively expensive and difficult to scale for multi-kilogram production due to the high cost of chiral stationary phases and the large volumes of solvents required. These legacy methods also frequently involve harsh reaction conditions or multiple purification steps that introduce impurities, complicating the downstream processing for high-value pharmaceutical intermediates. Consequently, there has been a persistent industry need for a catalytic method that can break the 50% yield barrier through dynamic processes or achieve high selectivity in kinetic resolution without excessive waste.
The Novel Approach
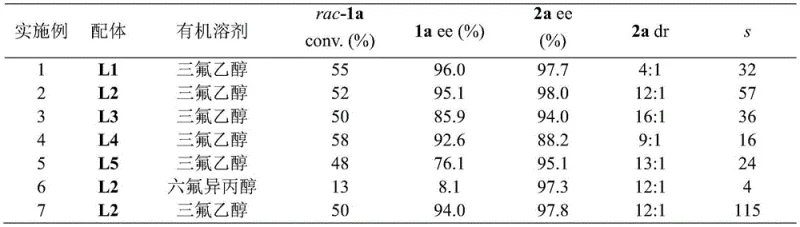
The methodology disclosed in the patent offers a transformative solution by employing a palladium-catalyzed asymmetric addition of arylboron reagents to cyclophane imines. Unlike traditional methods, this catalytic kinetic resolution approach allows for the differentiation of enantiomers based on their reaction rates with the chiral catalyst, enabling the isolation of both the unreacted imine and the newly formed amine product with high optical purity. The use of a chiral phosphine-nitrogen ligand complexed with palladium creates a highly defined chiral environment that effectively discriminates between the planar chiral faces of the substrate. This novel route operates under relatively mild conditions, typically around 60°C in trifluoroethanol, which significantly reduces energy consumption compared to high-temperature thermal resolutions. Moreover, the reaction exhibits a remarkably high resolution coefficient (s value), reaching up to 368 in optimized examples, which indicates an extremely efficient separation of enantiomers. This efficiency translates directly into higher yields of valuable chiral materials and reduced waste generation, aligning perfectly with modern green chemistry principles and cost-reduction strategies in fine chemical manufacturing.
Mechanistic Insights into Pd-Catalyzed Asymmetric Addition
The core of this technological breakthrough resides in the intricate interplay between the palladium metal center and the specialized chiral ligands, which dictates the stereochemical outcome of the reaction. The catalyst system utilizes a series of phosphine-oxazoline ligands, often featuring a ferrocene backbone, which provides a rigid and sterically demanding framework essential for high enantioselectivity. During the catalytic cycle, the palladium species activates the arylboronic acid through transmetallation, forming an organopalladium intermediate that subsequently attacks the imine double bond. The chiral ligand, such as the ferrocene-based derivatives shown in the patent data, imposes strict spatial constraints that favor the approach of the nucleophile to one specific face of the planar chiral imine. This precise control ensures that the addition reaction proceeds with high diastereoselectivity, generating a new stereocenter adjacent to the existing planar chirality. The result is the formation of chiral amines with defined (Sp, S) or (Rp, R) configurations, depending on the ligand handedness used. Understanding this mechanism is crucial for process optimization, as slight modifications to the ligand structure, such as changing the substituent on the oxazoline ring from tert-butyl to benzyl, can significantly alter the electronic and steric properties, thereby tuning the selectivity for different substrate classes.
Impurity control in this system is inherently managed by the high specificity of the catalyst, which minimizes the formation of side products such as homocoupling of boronic acids or non-selective background reactions. The patent data highlights that the reaction tolerates a wide range of functional groups on the arylboronic acid, including halogens, alkyls, and electron-withdrawing groups like trifluoromethyl, without compromising the enantiomeric excess. This robustness suggests that the catalytic cycle is resilient against potential poisoning by heteroatoms or steric bulk, a common issue in transition metal catalysis. Furthermore, the subsequent allylation step to protect the amine nitrogen is performed under basic conditions using sodium hydride, which is a standard and clean transformation that does not introduce racemization. The combination of high enantioselectivity in the resolution step and the stability of the chiral center during protection ensures that the final product maintains stringent purity specifications required for pharmaceutical applications. This level of control over the impurity profile simplifies the purification process, often allowing for direct column chromatography to isolate the target compounds in high purity, thereby reducing the need for extensive recrystallization campaigns.
How to Synthesize Chiral Cyclophane Derivatives Efficiently
The synthesis protocol outlined in the patent provides a clear and reproducible pathway for producing these high-value chiral intermediates, starting from readily available racemic cyclophane imines. The process begins with the in situ preparation of the active catalyst, where palladium trifluoroacetate is complexed with the chiral ligand in acetone, followed by solvent removal to ensure a homogeneous mixture before the addition of reactants. The resolution reaction is then conducted in trifluoroethanol at 60°C for approximately 15 hours, a timeframe that balances conversion and selectivity to maximize the resolution coefficient. Following the resolution, the crude mixture containing both the recovered imine and the amine product is separated via column chromatography. The amine fraction is then subjected to a protection step involving deprotonation with sodium hydride in DMF and subsequent alkylation with allyl bromide. This two-stage sequence—resolution followed by protection—is designed to be operationally simple, avoiding the need for inert atmosphere gloveboxes for the entire duration, although nitrogen protection is recommended for the catalyst preparation and reaction stages. The detailed standardized synthesis steps below provide the specific molar ratios and workup procedures necessary to replicate the high yields and selectivities reported in the patent examples.
- Prepare the palladium catalyst by stirring palladium trifluoroacetate and chiral phosphine-oxazoline ligand in acetone, then concentrate under vacuum.
- Conduct the kinetic resolution by reacting the cyclophane imine substrate with arylboronic acid in trifluoroethanol at 60°C for 15 hours using the prepared catalyst.
- Perform allylation on the resolved amine precursor using sodium hydride and allyl bromide in DMF to obtain the final protected chiral amine product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this palladium-catalyzed resolution technology offers substantial strategic advantages over legacy sourcing models for chiral cyclophanes. The primary benefit stems from the drastic simplification of the supply chain for chiral reagents; instead of relying on expensive, stoichiometric chiral resolving agents that generate equimolar waste, this method uses a catalytic amount of a commercially available ligand system. This shift significantly reduces the raw material cost per kilogram of the final product, as the chiral information is recycled through the catalyst rather than consumed in the reaction. Additionally, the use of arylboronic acids as coupling partners is highly advantageous from a sourcing perspective, as these reagents are commodity chemicals produced on a massive scale for Suzuki couplings, ensuring stable pricing and reliable availability from multiple global vendors. The mild reaction conditions, operating at moderate temperatures without the need for cryogenic cooling, further contribute to cost reduction by lowering energy utility costs and reducing the complexity of the reactor infrastructure required for production. These factors combined create a more resilient and cost-effective supply chain for high-purity chiral intermediates.
- Cost Reduction in Manufacturing: The transition from stoichiometric resolution to catalytic kinetic resolution fundamentally alters the cost structure of producing chiral cyclophanes. By eliminating the need for large quantities of chiral acids or bases and avoiding the 50% yield ceiling of classical resolution, the process maximizes the throughput of the manufacturing facility. The high turnover number of the palladium catalyst means that the cost contribution of the precious metal and the chiral ligand is amortized over a much larger quantity of product. Furthermore, the simplified workup procedure, which often requires only standard column chromatography rather than complex fractional crystallizations, reduces labor hours and solvent consumption. This streamlined workflow translates directly into lower operating expenses and a more competitive price point for the final API intermediate, allowing pharmaceutical companies to optimize their bill of materials for complex drug candidates.
- Enhanced Supply Chain Reliability: Dependence on single-source suppliers for exotic chiral auxiliaries poses a significant risk to production continuity, but this method mitigates that risk by utilizing widely available boronic acids and standard palladium precursors. The robustness of the reaction across a broad substrate scope means that a single catalytic platform can be adapted to produce a variety of different chiral derivatives without requiring extensive re-optimization or new supplier qualification. This flexibility allows manufacturers to respond quickly to changes in demand or to pivot production to different analogues as drug development programs evolve. Moreover, the stability of the intermediates and the final allyl-protected products ensures that inventory can be held without significant degradation, providing a buffer against supply disruptions. This reliability is critical for maintaining the uninterrupted flow of materials needed for clinical trials and commercial drug launches.
- Scalability and Environmental Compliance: Scaling chemical processes from the laboratory to commercial production often reveals hidden bottlenecks, but this catalytic system is designed with scalability in mind. The use of trifluoroethanol as a solvent, while requiring appropriate handling, is manageable on a large scale, and the reaction does not generate hazardous gaseous byproducts or require extreme pressures. The high selectivity of the reaction minimizes the formation of difficult-to-remove impurities, which simplifies the purification train and reduces the volume of waste solvent generated per unit of product. This alignment with green chemistry principles not only lowers waste disposal costs but also ensures compliance with increasingly stringent environmental regulations regarding heavy metal residues and solvent emissions. The ability to produce these complex chiral molecules with a smaller environmental footprint enhances the sustainability profile of the supply chain, a key metric for modern pharmaceutical procurement strategies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this resolution technology, derived from the specific data points and experimental results detailed in the patent documentation. These insights are intended to clarify the capabilities of the method for potential partners evaluating its suitability for their specific synthetic routes. Understanding the nuances of substrate compatibility, selectivity metrics, and operational parameters is essential for making informed decisions about integrating this chemistry into existing manufacturing workflows. The answers provided reflect the proven performance of the system as demonstrated in the patent examples, offering a realistic expectation of the technology's potential.
Q: What is the enantioselectivity achieved in this resolution method?
A: The method achieves exceptionally high enantiomeric excess (ee), reaching up to 99.9% for the recovered imine and 99.4% for the amine product, with a resolution coefficient (s) as high as 368.
Q: Which substrates are compatible with this palladium-catalyzed system?
A: The system demonstrates broad substrate scope, successfully resolving various cyclophane imines substituted with phenyl, naphthyl, and halogenated or alkoxy-substituted benzene rings.
Q: Is this process suitable for large-scale manufacturing?
A: Yes, the process utilizes commercially available catalysts and mild reaction conditions (60°C), avoiding cryogenic temperatures or exotic reagents, which facilitates straightforward scale-up for industrial production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Cyclophane Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-purity chiral intermediates play in the development of next-generation therapeutics and advanced functional materials. Our team of expert process chemists has extensively evaluated the palladium-catalyzed resolution technology described in CN112538033A and possesses the technical capability to implement this route effectively. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial manufacturing is seamless and efficient. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, including chiral HPLC analysis to guarantee the enantiomeric excess levels required for your specific application. We are committed to delivering consistent quality and reliability, leveraging our deep understanding of asymmetric catalysis to optimize yields and minimize costs for our global partners.
We invite you to collaborate with us to explore the full potential of this innovative synthesis method for your project needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis that details how adopting this catalytic resolution can improve your overall process economics. Please contact us to request specific COA data for our chiral cyclophane portfolio or to discuss route feasibility assessments for your target molecules. By partnering with NINGBO INNO PHARMCHEM, you gain access to a supply chain that is not only robust and compliant but also driven by a commitment to technical excellence and continuous improvement in chiral synthesis.
