Revolutionizing Asymmetric Biaryl Manufacturing with Advanced Cyclopalladated Ferrocenylimine Catalysts
Introduction to Advanced Catalytic Solutions for Biaryl Synthesis
The landscape of organic synthesis for complex pharmaceutical intermediates is constantly evolving, driven by the need for more efficient and sustainable methodologies. Patent CN102125875A introduces a significant breakthrough in this domain through the application of a cyclopalladated ferrocenylimine-phosphine adduct in the synthesis of asymmetric biaryl compounds. This technology addresses critical bottlenecks in traditional cross-coupling reactions by enabling a highly efficient one-pot borylation and Suzuki-Miyaura cross-coupling (BSC) sequence. For R&D directors and process chemists, the implications are profound, offering a pathway to reduce operational complexity while maintaining rigorous purity standards. The core innovation lies in the unique structural properties of the ferrocenyl-based palladium catalyst, which provides superior stability and activity compared to conventional palladium sources. By leveraging this advanced catalytic system, manufacturers can achieve high yields of up to 95% under relatively mild reaction conditions, marking a substantial improvement over legacy processes that often require harsh conditions or excessive reagent usage.
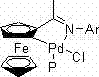
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional methods for synthesizing asymmetric biaryl compounds typically rely on a stepwise approach where the aryl boronic acid or ester must be synthesized, isolated, purified, and then subjected to the Suzuki-Miyaura coupling reaction. This multi-step process is not only labor-intensive but also incurs significant material losses at each isolation stage, leading to reduced overall yields and increased production costs. Furthermore, conventional catalytic systems often suffer from instability during the prolonged reaction times required for difficult substrates, necessitating the addition of fresh palladium catalyst mid-process to drive the reaction to completion. Another major drawback identified in prior art is the requirement for a large excess of the first halogenated arene to ensure sufficient generation of the boronate ester for the subsequent coupling. This excess leads to problematic self-coupling side reactions, creating impurities that are difficult to separate from the target product, thereby complicating downstream purification and compromising the quality of the final pharmaceutical intermediate.
The Novel Approach
The novel approach detailed in the patent utilizes a specialized ferrocenimine cyclopalladium-phosphine adduct to facilitate a seamless one-pot transformation. This methodology effectively merges the borylation and cross-coupling steps into a single continuous operation, eliminating the need for intermediate isolation and significantly streamlining the manufacturing workflow. The robust nature of this catalyst allows it to remain active throughout both stages of the reaction, removing the necessity for supplementary catalyst addition which is a common pain point in existing technologies. By optimizing the reaction stoichiometry and conditions, this system minimizes the formation of homocoupling byproducts, ensuring a cleaner reaction profile and higher selectivity for the desired asymmetric biaryl product. The ability to tolerate a wide range of functional groups and substrate types, including challenging heterocyclic halides, further underscores the versatility of this new catalytic protocol for diverse chemical applications.
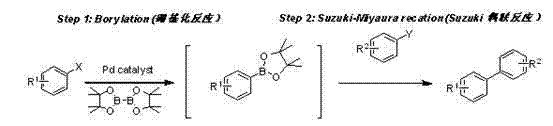
Mechanistic Insights into Ferrocenimine Cyclopalladium-Phosphine Catalysis
The exceptional performance of the ferrocenimine cyclopalladium-phosphine adduct can be attributed to its unique electronic and steric environment provided by the ferrocenyl backbone and the phosphine ligand. The rigid structure of the cyclopalladated complex enhances thermal stability, preventing the decomposition of the active palladium species even at elevated temperatures ranging from 80°C to 110°C. This stability is crucial for maintaining catalytic turnover numbers over the extended reaction periods required for the second coupling step, which can last up to 15 hours for less reactive substrates like chlorobenzenes. The phosphine moiety facilitates the oxidative addition of the aryl halide, while the ferrocenyl unit likely assists in stabilizing the transition states during the transmetallation and reductive elimination steps. This synergistic effect results in a highly efficient catalytic cycle that operates effectively at low catalyst loadings, typically between 0.5 mol% and 2 mol%, making it economically viable for large-scale production.
From an impurity control perspective, the mechanism inherently suppresses the formation of symmetric biaryl byproducts which often plague traditional Suzuki reactions. In conventional processes, the accumulation of free aryl boronic acid can lead to protodeboronation or homocoupling, but the in-situ generation and immediate consumption of the boronate species in this one-pot system mitigates these risks. The specific choice of bases, such as potassium acetate for the borylation step and potassium phosphate or cesium carbonate for the coupling step, is optimized to match the pKa requirements of each transformation without interfering with the catalyst integrity. This precise control over reaction parameters ensures that the impurity profile remains manageable, reducing the burden on purification teams and facilitating compliance with stringent regulatory standards for pharmaceutical ingredients.
How to Synthesize Asymmetric Biaryl Compounds Efficiently
Implementing this advanced synthesis route requires careful attention to the sequential addition of reagents and the maintenance of an inert atmosphere to protect the sensitive catalytic species. The process begins with the combination of the ferrocenimine cyclopalladium-phosphine catalyst, the first aryl halide, a suitable base like potassium acetate, and bis(pinacolato)diboron in a solvent such as 1,4-dioxane. This mixture is heated to initiate the borylation, converting the aryl halide into the corresponding boronate ester. Once this initial conversion is complete, the reaction mixture is cooled slightly before the direct addition of the second aryl halide and a stronger base to trigger the cross-coupling event. Detailed standardized synthesis steps see the guide below.
- Combine the ferrocenimine cyclopalladium-phosphine catalyst, aryl halide Aryl1-X, base A, and pinacol diboronate in an organic solvent like 1,4-dioxane.
- Heat the mixture to 80-110°C for 2-6 hours to complete the borylation step, generating the aryl boronate ester in situ.
- Without isolation, add the second aryl halide Aryl2-X and base B directly to the reaction vessel and continue heating at 80-110°C for 3-15 hours to finalize the Suzuki-Miyaura coupling.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this catalytic technology presents compelling opportunities for cost optimization and risk mitigation. The elimination of intermediate isolation steps translates directly into reduced solvent consumption and lower waste disposal costs, contributing to a more sustainable and economical manufacturing process. The high efficiency of the catalyst means that less precious metal is required per kilogram of product, which helps stabilize raw material costs against fluctuations in the palladium market. Additionally, the robustness of the reaction conditions reduces the likelihood of batch failures due to catalyst deactivation, ensuring more predictable production schedules and reliable delivery timelines for downstream customers.
- Cost Reduction in Manufacturing: The one-pot nature of this process significantly lowers operational expenditures by removing the unit operations associated with filtering, drying, and purifying the intermediate boronate ester. This consolidation of steps reduces energy consumption and labor hours, leading to substantial cost savings in the overall production budget. Furthermore, the ability to use lower catalyst loadings without sacrificing yield minimizes the expenditure on expensive palladium complexes, enhancing the gross margin potential for high-volume commercial runs.
- Enhanced Supply Chain Reliability: The broad substrate scope of this catalytic system allows for flexibility in sourcing raw materials, as it can accommodate various substituted aryl halides including those with electron-withdrawing or donating groups. This versatility reduces dependency on specific niche starting materials that might have volatile supply chains. The mild reaction temperatures also mean that standard glass-lined or stainless steel reactors can be used without requiring specialized high-pressure or cryogenic equipment, simplifying facility requirements and increasing available manufacturing capacity.
- Scalability and Environmental Compliance: Scaling this process from laboratory to commercial production is facilitated by the straightforward protocol which avoids complex handling of unstable intermediates. The reduction in solvent usage and waste generation aligns with increasingly strict environmental regulations, minimizing the ecological footprint of the manufacturing site. The high selectivity of the reaction ensures that the final product meets purity specifications with minimal downstream processing, reducing the load on wastewater treatment facilities and lowering the overall environmental compliance burden.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and capabilities of this ferrocenyl-palladium catalytic system. These insights are derived directly from the experimental data and claims presented in the patent documentation to provide clarity for technical decision-makers. Understanding these nuances is essential for evaluating the feasibility of integrating this technology into existing production lines for fine chemicals and pharmaceutical intermediates.
Q: What are the primary advantages of the ferrocenimine cyclopalladium-phosphine adduct over traditional catalysts?
A: This catalyst offers exceptional stability and high catalytic activity, allowing for lower catalyst loadings while achieving yields up to 95%. It effectively eliminates the need for adding extra palladium catalyst during the second coupling step, which is a common requirement in conventional systems.
Q: Does this one-pot method require the isolation of the intermediate aryl boronate ester?
A: No, the process is designed as a true one-pot synthesis. The aryl boronate ester generated in the first borylation step reacts directly in the second Suzuki-Miyaura step without isolation, significantly simplifying the workflow and reducing solvent waste.
Q: What types of substrates are compatible with this catalytic system?
A: The system demonstrates broad substrate tolerance, successfully coupling various aryl halides including bromobenzenes, chlorobenzenes, and heterocyclic halides. It is compatible with diverse functional groups such as amines, carbonyls, and cyano groups.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Asymmetric Biaryl Compounds Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced catalytic technologies like the ferrocenimine cyclopalladium-phosphine adduct in driving innovation within the fine chemical sector. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that promising laboratory discoveries are successfully translated into robust industrial processes. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of asymmetric biaryl compounds meets the exacting standards required by the global pharmaceutical industry.
We invite you to collaborate with us to leverage these cutting-edge synthetic methods for your next project. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our expertise can optimize your supply chain and enhance your product quality.
