Scalable Synthesis of Octahydropyrrolopyrrole Intermediates for Global Pharmaceutical Manufacturing
Scalable Synthesis of Octahydropyrrolopyrrole Intermediates for Global Pharmaceutical Manufacturing
The pharmaceutical industry constantly seeks robust, scalable pathways for complex heterocyclic intermediates that serve as the backbone for next-generation therapeutics. A significant breakthrough in this domain is detailed in patent CN111620877B, which outlines a novel preparation method for 1-benzyl-5-(tert-butyloxycarbonyl)octahydropyrrolopyrrole-2-carboxylic acid. This specific compound, characterized by its fused pyrrolidine ring system, represents a critical building block in medicinal chemistry, yet historically suffered from a lack of viable industrial synthesis routes. The patented process addresses this gap by introducing a concise four-step sequence that transforms readily available starting materials into high-value intermediates with exceptional efficiency. For R&D directors and procurement strategists, this development signals a shift from laboratory curiosity to commercial viability, offering a reliable pathway to secure supply chains for complex API precursors.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to the innovations described in the referenced patent, the synthesis of (2S,3aS,6aS)-1-benzyl-5-(tert-butoxycarbonyl)octahydropyrrolo[3,4-b]pyrrole-2-carboxylic acid was plagued by significant technical hurdles. The background art indicates a scarcity of literature reports, suggesting that existing methods were likely convoluted, low-yielding, or reliant on exotic reagents unsuitable for tonnage production. Conventional approaches to constructing fused nitrogen-containing heterocycles often involve multiple protection-deprotection cycles that erode overall yield and generate substantial chemical waste. Furthermore, the stereochemical control required for the (2S,3aS,6aS) configuration typically demands chiral resolution steps or expensive chiral pool starting materials, driving up costs and extending lead times. These limitations created a bottleneck for pharmaceutical developers who required consistent, high-purity supplies of this scaffold for drug discovery programs, forcing them to rely on inefficient custom synthesis or face supply discontinuity risks.
The Novel Approach
The methodology disclosed in patent CN111620877B revolutionizes this landscape by employing a streamlined four-step strategy that prioritizes operational simplicity and high conversion rates. The process initiates with a thermal cyclization in anhydrous toluene, followed by a selective deprotection and subsequent Boc-protection sequence, culminating in a mild hydrolysis step. This route eliminates the need for complex chromatographic separations in the early stages, as evidenced by the reported 100% yield in the initial cyclization step. By utilizing common industrial solvents like toluene, methanol, and glacial acetic acid, the process aligns perfectly with standard manufacturing infrastructure, reducing the barrier to entry for commercial scale-up. The strategic use of benzyl glycine ethyl ester as a key synthon ensures that the nitrogen functionality is correctly positioned for the subsequent ring-closing events, demonstrating a sophisticated understanding of retrosynthetic analysis applied to practical manufacturing constraints.
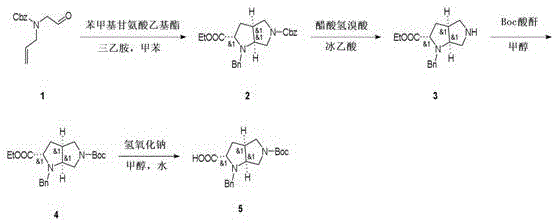
Mechanistic Insights into Intramolecular Cyclization and Boc Protection
The core of this synthetic achievement lies in the efficient construction of the octahydropyrrolopyrrole core through a thermally driven intramolecular cyclization. In the first step, the reaction between compound 1 and benzyl glycine ethyl ester in the presence of triethylamine facilitates a nucleophilic attack that closes the second pyrrolidine ring. The use of a Dean-Stark trap or similar water removal technique during the reflux at 110-130°C is critical, as it drives the equilibrium forward by removing the water byproduct, thereby pushing the reaction to completion. This mechanistic detail is vital for process chemists, as it highlights the importance of water management in achieving the reported quantitative conversion. The stereochemistry is inherently controlled by the geometry of the transition state during this ring closure, ensuring the formation of the desired cis-fused ring system without the need for external chiral catalysts.
Following the cyclization, the process employs a clever tandem deprotection and re-protection strategy to install the tert-butyloxycarbonyl (Boc) group, which is essential for downstream peptide coupling applications. The treatment with acetic acid hydrobromide serves to cleave the benzyl protecting group under acidic conditions, generating a free amine intermediate that is immediately trapped by Boc anhydride in methanol. This one-pot or telescoped approach minimizes the isolation of unstable free amine species, which are prone to oxidation or polymerization. The final hydrolysis step using sodium hydroxide in a methanol-water mixture is a classic saponification reaction that converts the ethyl ester to the free carboxylic acid. The mild conditions (25°C) prevent racemization of the chiral centers, preserving the optical purity of the final product, which is a non-negotiable requirement for pharmaceutical intermediates intended for biological activity.
How to Synthesize 1-Benzyl-5-(tert-butyloxycarbonyl)octahydropyrrolopyrrole-2-carboxylic acid Efficiently
Executing this synthesis requires precise control over reaction parameters to replicate the high yields observed in the patent examples. The process begins with the rigorous drying of toluene to prevent hydrolysis of the starting esters during the high-temperature reflux. Operators must monitor the reaction progress via TLC, looking for the complete disappearance of the starting amine and ester signals. Following the workup, which involves acid-base extractions to remove triethylamine salts, the crude oil can often be used directly in the next step, further streamlining the workflow. The subsequent acidic treatment requires careful temperature control, starting at 0°C to manage the exotherm upon addition of hydrobromic acid, before warming to room temperature to ensure complete deprotection.
- React compound 1 with benzyl glycine ethyl ester and triethylamine in anhydrous toluene at 110-130°C for 12 hours to obtain compound 2.
- Dissolve compound 2 in glacial acetic acid and treat with acetic acid hydrobromide at 0°C, then stir at 25°C for 2 hours to form compound 3.
- React compound 3 with Boc anhydride in methanol at room temperature for 12 hours to yield the protected intermediate compound 4.
- Hydrolyze compound 4 using sodium hydroxide in a water and methanol mixture at 25°C for 2 hours to obtain the final carboxylic acid product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition from a theoretical synthesis to the robust process described in CN111620877B offers tangible strategic benefits. The primary advantage lies in the accessibility of raw materials; benzyl glycine ethyl ester and the requisite anhydrides are commodity chemicals available from multiple global suppliers, mitigating the risk of single-source dependency. This abundance ensures that production schedules are not held hostage by the availability of niche reagents, a common pain point in the synthesis of complex heterocycles. Furthermore, the elimination of transition metal catalysts or cryogenic conditions significantly reduces the operational expenditure (OPEX) associated with specialized equipment and hazardous waste disposal. The process relies on standard glass-lined reactors and conventional heating/cooling systems, making it adaptable to existing manufacturing suites without requiring capital-intensive retrofitting.
- Cost Reduction in Manufacturing: The economic viability of this route is underpinned by the high atom economy and the avoidance of expensive chiral auxiliaries. By achieving near-quantitative yields in the initial cyclization and final hydrolysis steps, the process minimizes the loss of valuable intermediates, directly translating to lower cost per kilogram of the final API intermediate. The use of recyclable solvents like toluene and methanol further enhances the cost profile, allowing for solvent recovery loops that reduce both material costs and environmental impact. Additionally, the simplified workup procedures, which rely on standard aqueous extractions rather than preparative HPLC, drastically reduce labor hours and consumable costs associated with purification.
- Enhanced Supply Chain Reliability: Supply continuity is bolstered by the robustness of the reaction conditions, which tolerate minor variations in reagent quality better than sensitive catalytic processes. The short four-step sequence reduces the cumulative cycle time, enabling manufacturers to respond more agilely to fluctuations in market demand. Since the intermediates generated in steps 2 and 3 are relatively stable, they can potentially be stocked as buffer inventory, decoupling the production of the final acid from the immediate availability of the starting cyclization precursors. This flexibility is crucial for maintaining just-in-time delivery schedules for downstream pharmaceutical clients who operate on tight development timelines.
- Scalability and Environmental Compliance: From an EHS (Environment, Health, and Safety) perspective, the process avoids the generation of heavy metal waste streams, simplifying regulatory compliance and wastewater treatment protocols. The reactions are conducted at moderate temperatures and pressures, reducing the energy footprint of the manufacturing campaign. The scalability is evidenced by the use of bulk solvents and the absence of dilution-dependent steps that often hinder scale-up. This makes the technology transfer from pilot plant to commercial production seamless, ensuring that the quality attributes established at the gram scale are maintained at the metric ton level, a key requirement for GMP manufacturing environments.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of this specialized pharmaceutical intermediate. These insights are derived directly from the experimental data and beneficial effects outlined in the patent documentation, providing clarity on the feasibility and advantages of this specific synthetic route. Understanding these details is essential for stakeholders evaluating the integration of this intermediate into their broader supply chain strategies.
Q: What are the primary advantages of this synthesis route for industrial scale-up?
A: The process utilizes readily available raw materials such as benzyl allyl (2-oxyethylene ethyl) amino methyl ester and benzyl glycine ethyl ester. It features a short four-step route with high yields in key stages (up to 100% in step 1 and 4), making it highly suitable for large-scale commercial production without complex purification bottlenecks.
Q: How does this method address the lack of existing industrial synthesis methods?
A: Prior to this invention, there were few reports on the synthesis of this specific octahydropyrrolopyrrole derivative, creating a supply gap. This patent provides a robust, controllable reaction sequence that solves the technical problem of unavailable industrial methods, ensuring a stable supply chain for downstream drug development.
Q: What are the specific reaction conditions required for the cyclization step?
A: The critical cyclization step involves refluxing the reactants in anhydrous toluene with triethylamine at temperatures between 110-130°C for 12 hours. This thermal condition ensures complete consumption of the starting material, as verified by TLC, driving the equilibrium towards the desired bicyclic structure efficiently.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1-Benzyl-5-(tert-butyloxycarbonyl)octahydropyrrolopyrrole-2-carboxylic acid Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of novel therapeutics depends on the reliability and quality of the underlying chemical supply chain. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project needs are met with precision and consistency. Our facilities are equipped with stringent purity specifications and rigorous QC labs capable of validating the structural integrity and optical purity of complex intermediates like the octahydropyrrolopyrrole derivatives discussed herein. We are committed to delivering products that meet the highest international standards, providing you with the confidence to advance your drug candidates through clinical trials and into the market.
We invite you to leverage our technical expertise to optimize your sourcing strategy for this critical building block. Our team is prepared to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how our optimized processes can enhance your project's economic viability. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments, allowing us to collaborate on securing a sustainable and cost-effective supply of high-purity pharmaceutical intermediates for your organization.
