Advanced Photocatalytic Route for High-Purity 3-Trifluoromethylselenochromone Manufacturing
Advanced Photocatalytic Route for High-Purity 3-Trifluoromethylselenochromone Manufacturing
The pharmaceutical and fine chemical industries are constantly seeking more efficient, cost-effective, and environmentally benign synthetic routes for complex heterocyclic scaffolds. A significant breakthrough in this domain is detailed in patent CN113735813B, which discloses a novel photocatalytic synthesis method for 3-trifluoromethylselenochromone compounds. This technology represents a paradigm shift from traditional stoichiometric metal-mediated reactions to a sustainable, visible-light-driven catalytic process. By utilizing o-hydroxy phenyl enaminoketone compounds and p-toluenesulfonic acid trifluoromethylselenate (TsSeCF3) in a one-pot reaction, this method achieves high selectivity and excellent yields under remarkably mild conditions. For R&D directors and procurement managers alike, this innovation offers a compelling solution for accessing these biologically active intermediates with reduced operational complexity and significantly lowered material costs.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to this invention, the synthesis of 3-trifluoromethylthio chromone derivatives relied heavily on harsh and economically inefficient protocols. As illustrated in the background art, conventional methods typically employed a large excess of expensive silver trifluoromethanethiolate (AgSCF3), often requiring up to 3 stoichiometric equivalents, alongside 1.5 equivalents of trichloroisocyanuric acid (TCCA) to generate electrophilic species in situ. 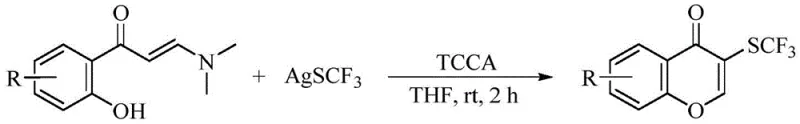 This stepwise approach not only suffers from poor atom economy due to the generation of substantial silver waste but also incurs prohibitive raw material costs that hinder commercial scalability. Furthermore, the requirement for strong oxidants and the multi-step nature of the operation introduce significant safety hazards and purification challenges, making the traditional route unsuitable for the reliable supply chain demands of modern API manufacturing.
This stepwise approach not only suffers from poor atom economy due to the generation of substantial silver waste but also incurs prohibitive raw material costs that hinder commercial scalability. Furthermore, the requirement for strong oxidants and the multi-step nature of the operation introduce significant safety hazards and purification challenges, making the traditional route unsuitable for the reliable supply chain demands of modern API manufacturing.
The Novel Approach
In stark contrast, the patented photocatalytic methodology streamlines the synthesis into a single, elegant pot reaction driven by visible light energy. By replacing the costly silver reagent with p-toluenesulfonic acid trifluoromethylselenate, the process drastically reduces the cost of goods sold (COGS) while improving the environmental profile of the synthesis. The reaction proceeds smoothly at room temperature without the need for external heating or cryogenic cooling, utilizing a simple blue LED light source to activate the catalyst. This transition from a stoichiometric metal-based oxidation to a catalytic radical pathway exemplifies the principles of green chemistry, offering a robust alternative for the cost reduction in pharmaceutical intermediate manufacturing. The simplicity of the workup procedure further enhances its appeal for industrial adoption, ensuring a consistent supply of high-quality intermediates.
Mechanistic Insights into Visible Light Photocatalytic Cyclization
The core of this technological advancement lies in the intricate radical mechanism facilitated by the Eosin Y photocatalyst under blue light irradiation. Upon exposure to visible light, the p-toluenesulfonic acid trifluoromethylselenate undergoes homolytic cleavage to generate highly active trifluoromethyl seleno radicals in situ. These radicals subsequently dimerize to form a ditrifluoromethyl diseleno-ether intermediate, which serves as the key electrophilic species for the subsequent transformation. 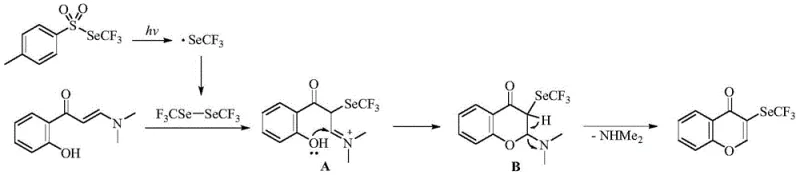 This intermediate then engages in an addition reaction with the o-hydroxy phenyl enaminone substrate to form a transient intermediate A. Through a carefully orchestrated sequence involving intramolecular nucleophilic cyclization to form intermediate B, followed by elimination of dimethylamine, the final 3-trifluoromethylselenochromone scaffold is constructed. This mechanistic pathway avoids the formation of heavy metal byproducts and ensures high regioselectivity, which is critical for maintaining the purity profile required by regulatory standards.
This intermediate then engages in an addition reaction with the o-hydroxy phenyl enaminone substrate to form a transient intermediate A. Through a carefully orchestrated sequence involving intramolecular nucleophilic cyclization to form intermediate B, followed by elimination of dimethylamine, the final 3-trifluoromethylselenochromone scaffold is constructed. This mechanistic pathway avoids the formation of heavy metal byproducts and ensures high regioselectivity, which is critical for maintaining the purity profile required by regulatory standards.
From an impurity control perspective, the mildness of the reaction conditions plays a pivotal role in minimizing side reactions. Traditional methods involving strong oxidants like TCCA often lead to over-oxidation or degradation of sensitive functional groups on the aromatic ring. However, the photocatalytic cycle operates under neutral and ambient conditions, preserving the integrity of diverse substituents such as halogens, alkoxy groups, and alkyl chains. The patent data indicates that varying the R1 substituent has no obvious negative influence on the reaction outcome, with yields consistently remaining above 86% across a broad substrate scope. This functional group tolerance simplifies the purification process, as fewer side products are generated, thereby reducing the burden on downstream processing and quality control laboratories.
How to Synthesize 3-Trifluoromethylselenochromone Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires adherence to specific optimized parameters to ensure reproducibility and maximum efficiency. The protocol utilizes a straightforward mixing of reagents in a polar aprotic solvent, followed by irradiation with a specific wavelength of light. The choice of solvent is critical, with dimethyl sulfoxide (DMSO) identified as the optimal medium, outperforming acetonitrile and DMF in terms of yield and reaction rate.  The detailed standardized synthesis steps below outline the precise molar ratios, catalyst loading, and irradiation times necessary to achieve the reported high yields, serving as a foundational guide for process chemists looking to adopt this technology for commercial scale-up of complex pharmaceutical intermediates.
The detailed standardized synthesis steps below outline the precise molar ratios, catalyst loading, and irradiation times necessary to achieve the reported high yields, serving as a foundational guide for process chemists looking to adopt this technology for commercial scale-up of complex pharmaceutical intermediates.
- Combine o-hydroxyphenylenaminone (0.40 mmol), trifluoromethylselenate (0.44 mmol), and Eosin Y catalyst (0.20 mmol) in DMSO solvent within a quartz reaction tube.
- Stir the mixture at room temperature while irradiating with a 10 W blue LED light source for approximately 6 hours, monitoring progress via TLC.
- Quench the reaction with water, extract with ethyl acetate, wash with saturated brine, dry, concentrate, and purify via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this photocatalytic process offers tangible strategic advantages beyond mere technical novelty. The primary driver for adoption is the substantial cost savings achieved by eliminating the reliance on precious metal reagents. By removing the need for stoichiometric amounts of silver salts, the raw material expenditure is drastically simplified, allowing for more competitive pricing structures in the final supply of intermediates. Additionally, the use of commodity chemicals like TsSeCF3 and organic dyes ensures a stable and resilient supply chain, mitigating the risks associated with the volatility of noble metal markets. This stability is crucial for long-term project planning and securing reliable contracts for multi-ton production campaigns.
- Cost Reduction in Manufacturing: The economic impact of this process is profound, primarily driven by the substitution of expensive silver reagents with cost-effective organic selenium sources. The elimination of silver not only reduces the direct cost of raw materials but also removes the downstream expenses associated with silver recovery and waste disposal, which are significant operational burdens in traditional synthesis. Furthermore, the high atom economy of the one-pot reaction minimizes solvent usage and waste generation, leading to lower overall processing costs. The ability to run the reaction at room temperature also translates to significant energy savings, as no heating or cooling infrastructure is required, further enhancing the cost-efficiency profile of the manufacturing process.
- Enhanced Supply Chain Reliability: Supply chain continuity is bolstered by the use of widely available and stable starting materials. Unlike specialized organometallic reagents that may have long lead times or limited suppliers, the reagents used in this photocatalytic method are commercially accessible from multiple vendors. This diversification of the supply base reduces the risk of production stoppages due to raw material shortages. Moreover, the robustness of the reaction conditions means that the process is less sensitive to minor fluctuations in environmental parameters, ensuring consistent batch-to-batch quality and reliable delivery schedules for downstream customers relying on these critical building blocks for drug development.
- Scalability and Environmental Compliance: The scalability of this method is exceptional, owing to its simple operational requirements and lack of hazardous reagents. The absence of strong oxidants and heavy metals simplifies the regulatory compliance landscape, making it easier to obtain necessary permits for large-scale production. The one-pot nature of the reaction reduces the number of unit operations, which facilitates easier scale-up from gram to kilogram and ton scales without significant re-engineering of the process. Additionally, the reduced waste stream and lower energy consumption align perfectly with modern sustainability goals and environmental regulations, positioning manufacturers who adopt this technology as leaders in green chemistry and responsible production practices.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this photocatalytic synthesis method. These answers are derived directly from the experimental data and comparative studies presented in the patent documentation, providing clarity on the practical aspects of the technology. Understanding these details is essential for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios.
Q: Why is the photocatalytic method superior to the traditional silver-mediated synthesis?
A: The traditional method requires 3 equivalents of expensive AgSCF3 and 1.5 equivalents of TCCA, resulting in poor atom economy and high costs. The new photocatalytic route uses cheaper TsSeCF3 reagents under mild visible light, eliminating the need for stoichiometric silver and harsh oxidants.
Q: What are the optimal reaction conditions for maximum yield?
A: The patent specifies that using DMSO as the solvent, Eosin Y as the photocatalyst, and a 10 W blue LED light source at room temperature for 6 hours provides the highest yields, often exceeding 90% for various substrates.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process operates at room temperature without external heating or cooling, uses readily available reagents, and involves a simple one-pot procedure with easy purification, making it highly scalable for commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Trifluoromethylselenochromone Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this photocatalytic technology in advancing the synthesis of complex heterocyclic intermediates. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the benefits of this innovative method are fully realized at an industrial level. Our state-of-the-art facilities are equipped with advanced photoreactors and rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of 3-trifluoromethylselenochromone delivered meets the highest international standards for pharmaceutical applications.
We invite global partners to collaborate with us to leverage this cost-effective and sustainable synthesis route for their drug discovery and development programs. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to reach out today to discuss specific COA data and route feasibility assessments, ensuring a seamless integration of this high-performance intermediate into your supply chain.
