Advanced Chiral Iodoarene Catalysts for Scalable Spirolactone Manufacturing
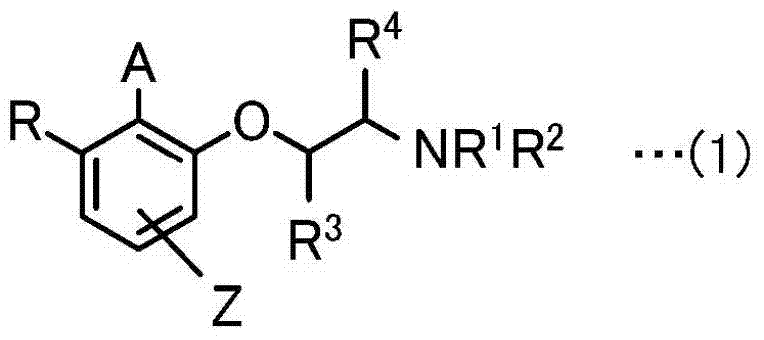
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies for constructing complex chiral scaffolds, particularly spirocyclic structures found in numerous bioactive natural products and drug candidates. Patent CN103415501A introduces a groundbreaking advancement in this domain by disclosing novel iodoarene derivatives that serve as highly efficient, stable precursors for hypervalent iodine catalysts. This technology addresses critical limitations in prior art regarding catalyst stability and enantioselectivity, offering a reliable pathway for the enantioselective dearomatization of phenols and naphthols. By utilizing 1,2-amino alcohols as chiral sources rather than traditional lactic acid derivatives, the invention ensures that the asymmetric center remains configurationally stable, avoiding racemization issues caused by keto-enol tautomerism. This stability is paramount for industrial applications where shelf-life and consistent batch-to-batch reproducibility are non-negotiable requirements for a reliable pharmaceutical intermediate supplier.
The significance of this innovation extends beyond mere academic interest; it provides a practical solution for the synthesis of optically active spirolactone compounds, which are valuable building blocks in medicinal chemistry. The ability to generate these complex architectures with high enantiomeric excess (ee) using catalytic amounts of the iodine reagent represents a substantial leap forward in green chemistry and process efficiency. For procurement managers and supply chain heads, this translates to a reduction in the reliance on stoichiometric oxidants and expensive chiral auxiliaries, thereby streamlining the manufacturing process. The patent details extensive experimental data confirming that these derivatives can be synthesized in short steps from commercially available starting materials like 2-iodoresorcinol, making them economically viable for large-scale production.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the enantioselective oxidative dearomatization of phenols has relied heavily on hypervalent iodine reagents. However, earlier generations of chiral hypervalent iodine precursors, such as those described in Non-Patent Document 1 (Angew. Chem. Int. Ed. 2010), faced significant stability challenges. These conventional precursors often utilized lactic acid as the chiral source, introducing a carbonyl group adjacent to the chiral carbon atom. This structural feature rendered them susceptible to racemization through keto-enol tautomerism, especially under varying storage conditions or during prolonged reaction times. For an R&D director, this instability poses a severe risk to process robustness, as the erosion of optical purity directly impacts the quality of the final active pharmaceutical ingredient (API). Furthermore, the synthesis of these older precursors often required multi-step sequences that were not easily scalable, leading to higher costs and supply chain vulnerabilities. The inability to recycle the iodine species efficiently also contributed to increased waste generation and higher environmental compliance burdens.
The Novel Approach
The methodology presented in CN103415501A overcomes these hurdles by employing 1,2-amino alcohols as the chiral source for the iodoarene derivatives. This strategic modification eliminates the labile carbonyl group adjacent to the stereocenter, thereby locking the configuration and preventing racemization. The resulting catalyst precursors, represented by Formula (1) in the patent, exhibit exceptional stability and can be stored without significant degradation. From a manufacturing perspective, this novel approach allows for the use of catalytic amounts (typically 5 mol% to 10 mol%) of the iodine species in the presence of a stoichiometric oxidant like m-CPBA. The reaction proceeds via an in situ generation of the active hypervalent iodine (III) species, which then mediates the dearomatization. This catalytic cycle not only improves atom economy but also simplifies the workup procedure. The flexibility in designing the side chains (R groups) allows chemists to fine-tune the steric and electronic properties of the catalyst to match specific substrate requirements, ensuring high yields and enantioselectivity across a diverse range of phenolic substrates.
Mechanistic Insights into Hypervalent Iodine-Catalyzed Dearomatization
The core of this technology lies in the unique reactivity of the hypervalent iodine species generated in situ. The reaction mechanism begins with the oxidation of the monovalent iodoarene derivative (I) by a percarboxylic acid, such as m-chloroperbenzoic acid (m-CPBA), to form a trivalent hypervalent iodine (III) intermediate. This active species possesses two ligands coordinated to the iodine center, creating a highly electrophilic environment capable of activating the phenolic substrate. The phenol derivative, which contains a carboxylic acid side chain positioned appropriately for cyclization, attacks the activated iodine center. This interaction facilitates the transfer of oxygen to the aromatic ring, triggering a dearomatization event that converts the planar aromatic system into a three-dimensional spirocyclic structure. Crucially, the chiral environment provided by the amino alcohol-derived side chains on the iodine catalyst directs the facial selectivity of this oxidation, ensuring that one enantiomer of the spirolactone is formed preferentially over the other.
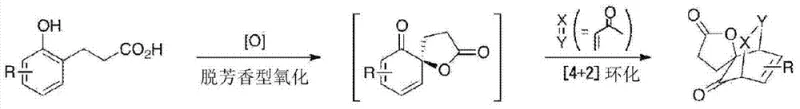
Following the oxidative dearomatization, the hypervalent iodine species is reduced back to its monovalent state, regenerating the catalyst precursor and completing the catalytic cycle. This regeneration is a key feature that distinguishes this method from stoichiometric approaches. The patent highlights that the nature of the ligands on the iodine atom can vary; they may be acyloxy groups derived from the oxidant or the substrate's carboxylic acid, or hydroxyl groups. The choice of solvent also plays a critical role in modulating the reaction outcome. For instance, the addition of alcohols like ethanol to halogenated solvents such as 1,2-dichloroethane (DCE) has been shown to enhance enantiomeric excess, likely by influencing the coordination sphere of the iodine center or stabilizing transition states. Understanding these mechanistic nuances allows process chemists to optimize reaction conditions for maximum efficiency and purity, ensuring that the final product meets the stringent specifications required for pharmaceutical applications.
How to Synthesize Optically Active Spirolactone Efficiently
The synthesis of these high-value chiral intermediates follows a streamlined protocol that balances reaction rate with stereocontrol. The process typically involves dissolving the phenolic substrate and the chiral iodoarene catalyst precursor in a dry organic solvent, followed by the controlled addition of the oxidant at low temperatures. Maintaining the reaction temperature between -20°C and 0°C is essential to suppress background non-catalyzed reactions that could lower the enantiomeric excess. After the reaction reaches completion, usually within 10 to 20 hours depending on the substrate, the mixture is quenched with a reducing agent like sodium thiosulfate to destroy excess oxidant. The product is then extracted into an organic phase, washed to remove acidic byproducts, and purified via column chromatography or recrystallization. The detailed standardized synthesis steps are provided in the guide below.
- Prepare the reaction mixture by combining the phenol derivative substrate, a catalytic amount of the chiral iodoarene derivative precursor (5-10 mol%), and m-CPBA (1.2 equivalents) in a suitable solvent such as DCE or chloroform.
- Maintain the reaction temperature between -20°C and 0°C to ensure high enantioselectivity while allowing the in situ generation of the active hypervalent iodine (III) species.
- Monitor the reaction progress via TLC or HPLC, then quench with sodium thiosulfate and bicarbonate solution, followed by extraction and purification to isolate the optically active spirolactone.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this catalytic technology offers compelling economic and operational benefits. The primary advantage lies in the drastic reduction of material costs associated with chiral reagents. By shifting from stoichiometric chiral auxiliaries to a catalytic system where the iodine species is recycled, the overall consumption of expensive chiral starting materials is significantly minimized. This reduction in reagent load directly translates to lower raw material costs per kilogram of the final product. Furthermore, the stability of the iodoarene precursors means they can be sourced in bulk and stored for extended periods without special handling requirements, reducing inventory turnover pressure and minimizing waste due to expiration. This reliability ensures a continuous supply of critical intermediates, mitigating the risk of production delays caused by reagent degradation.
- Cost Reduction in Manufacturing: The catalytic nature of this process fundamentally alters the cost structure of spirolactone production. Unlike traditional methods that require equimolar amounts of costly chiral oxidants, this system operates effectively with only 5 mol% to 10 mol% of the catalyst. This substantial decrease in reagent usage leads to direct savings on bill of materials. Additionally, the ability to use commercially available, lower-purity grades of oxidants like m-CPBA without compromising reaction performance further drives down input costs. The simplified workup procedure, which avoids complex metal removal steps often associated with transition metal catalysis, also reduces downstream processing expenses and solvent consumption, contributing to a more lean and cost-effective manufacturing operation.
- Enhanced Supply Chain Reliability: Supply chain continuity is often threatened by the instability of sensitive reagents. The iodoarene derivatives described in this patent are remarkably robust, resisting racemization and decomposition under standard storage conditions. This stability simplifies logistics, allowing for safer transportation and longer shelf lives, which in turn reduces the frequency of urgent replenishment orders. The synthesis of the catalyst precursors themselves relies on readily available commodity chemicals such as 2-iodoresorcinol and common amino alcohols, ensuring that the supply base is broad and resilient against market fluctuations. This accessibility makes the technology highly scalable, enabling suppliers to ramp up production volumes quickly to meet surging demand without being bottlenecked by exotic or scarce raw materials.
- Scalability and Environmental Compliance: From an environmental and regulatory standpoint, this organocatalytic approach offers a greener alternative to heavy metal-catalyzed processes. The absence of toxic transition metals eliminates the need for rigorous and costly metal scavenging steps to meet residual metal limits in pharmaceutical products. The reaction generates benign byproducts, primarily carboxylic acids, which are easier to treat and dispose of compared to heavy metal waste streams. This alignment with green chemistry principles facilitates smoother regulatory approvals and reduces the environmental footprint of the manufacturing site. The process has been demonstrated to work efficiently on various scales, from milligram screening to gram-scale synthesis, indicating strong potential for successful translation to multi-kilogram commercial production with minimal process redesign.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this chiral iodoarene technology. These answers are derived directly from the experimental data and embodiments disclosed in the patent documentation, providing clarity on substrate scope, reaction conditions, and product quality. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this route into their existing manufacturing pipelines.
Q: What distinguishes these iodoarene derivatives from previous hypervalent iodine precursors?
A: Unlike earlier precursors based on lactic acid which suffered from racemization via keto-enol tautomerism, these novel derivatives utilize 1,2-amino alcohols as chiral sources, providing superior stability and preventing loss of optical purity during storage or reaction.
Q: Can this catalytic system be applied to diverse phenolic substrates?
A: Yes, the patent demonstrates broad substrate scope including 1-naphthol and 2-naphthol derivatives with various substituents such as halogens, alkyl groups, and silyl ethers, consistently achieving high yields and enantiomeric excess.
Q: Is the catalyst recoverable for industrial scale-up?
A: The iodoarene derivatives function catalytically and are regenerated in the reaction cycle. Their high stability allows for potential recovery and reuse, significantly reducing material costs for large-scale pharmaceutical manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Spirolactone Compounds Supplier
The technological potential of chiral iodoarene-catalyzed dearomatization is immense, offering a pathway to complex molecular architectures that are otherwise difficult to access. NINGBO INNO PHARMCHEM stands at the forefront of translating such advanced academic discoveries into commercial reality. As a specialized CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our facilities are equipped with state-of-the-art rigorous QC labs capable of monitoring enantiomeric excess and impurity profiles with precision, ensuring that every batch meets stringent purity specifications required by global regulatory bodies. We understand the critical nature of chiral intermediates in drug development and are committed to delivering consistent quality.
We invite you to collaborate with us to leverage this innovative chemistry for your next project. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements. Please contact us to request specific COA data for our catalog intermediates or to discuss route feasibility assessments for your proprietary targets. Together, we can accelerate your development timeline and secure a robust supply chain for your critical pharmaceutical ingredients.
