Advanced Iridium-Catalyzed Synthesis of Tetrahydroquinoline Pyrans for Commercial Pharmaceutical Applications
Advanced Iridium-Catalyzed Synthesis of Tetrahydroquinoline Pyrans for Commercial Pharmaceutical Applications
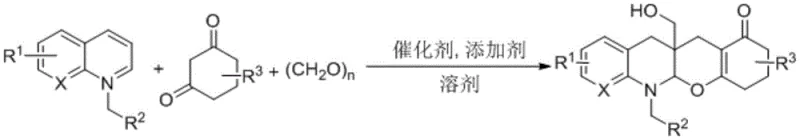
The landscape of organic synthesis is constantly evolving to meet the rigorous demands of modern pharmaceutical manufacturing, where efficiency, sustainability, and cost-effectiveness are paramount. A significant breakthrough in this domain is detailed in patent CN113234083B, which introduces a novel class of tetrahydroquinoline benzopyran compounds and a highly efficient preparation method thereof. This technology leverages an iridium-catalyzed hydrogen transfer cyclization reaction, utilizing azaarene salts, paraformaldehyde, and 1,3-cyclohexanedione compounds as key starting materials. Unlike traditional multi-step syntheses that often require harsh conditions and expensive reagents, this innovative approach employs paraformaldehyde as a dual-function agent, serving simultaneously as a hydrogen source and a coupling reagent. This unique mechanistic feature allows for the direct construction of complex nitrogen-oxygen heterocyclic frameworks in a single operational step, offering substantial advantages in terms of atom economy and diastereoselectivity. For global procurement and R&D teams, this represents a pivotal shift towards greener, more streamlined manufacturing processes for high-value pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of nitrogen-oxygen heterocyclic compounds, which are ubiquitous scaffolds in bioactive molecules, has been fraught with synthetic challenges. Conventional methodologies often necessitate complicated reaction sequences involving multiple isolation and purification steps, which inherently reduce overall yield and increase waste generation. Furthermore, many established routes require the use of additional external hydrogen sources or specialized additives to drive the cyclization, adding layers of complexity and cost to the process. The need for pre-functionalization of substrates is another common bottleneck, requiring chemists to install specific leaving groups or activating moieties before the core ring system can be formed. These factors collectively contribute to longer lead times, higher material costs, and increased environmental burden, making traditional methods less attractive for large-scale commercial production of complex heterocycles needed for drug development.
The Novel Approach
In stark contrast to these legacy methods, the technology disclosed in patent CN113234083B offers a transformative solution through a one-pot multicomponent reaction strategy. By utilizing an iridium catalyst, specifically dichloro(pentamethylcyclopentadienyl)iridium(III) dimer, the process facilitates a hydrogen transfer cyclization that bypasses the need for external hydrogen gas or complex reducing agents. The use of paraformaldehyde is particularly ingenious, as it acts as both the C1 building block and the hydride donor, effectively streamlining the reaction stoichiometry. This approach not only simplifies the operational workflow by combining multiple bond-forming events into a single vessel but also significantly enhances the step economy. The ability to synthesize diverse tetrahydroquinoline pyran derivatives by simply varying the structures of the azaarene salts and 1,3-cyclohexanediones provides a versatile platform for generating libraries of compounds for drug discovery without compromising on efficiency or scalability.
Mechanistic Insights into Iridium-Catalyzed Hydrogen Transfer Cyclization
The core of this synthetic innovation lies in the sophisticated catalytic cycle mediated by the iridium complex. The reaction initiates with the activation of the azaarene salt and the 1,3-cyclohexanedione derivative in the presence of a base, such as sodium tert-butoxide or potassium hydroxide. The iridium catalyst facilitates the transfer of hydrogen from the paraformaldehyde to the intermediate species, driving the cyclization forward with high diastereoselectivity. This hydrogen transfer mechanism is crucial as it avoids the safety hazards associated with high-pressure hydrogenation reactors while maintaining high conversion rates. The catalytic system is robust enough to tolerate various substituents on the aromatic rings, allowing for the synthesis of derivatives with furan, benzene, or alkyl groups without significant loss in performance. This mechanistic elegance ensures that the final tetrahydroquinoline pyran products are formed with high purity, minimizing the formation of difficult-to-remove impurities that often plague heterogeneous catalytic processes.
Furthermore, the reaction conditions are optimized to balance reactivity and selectivity, typically operating at moderate temperatures between 80°C and 90°C. This thermal window is sufficient to activate the paraformaldehyde and drive the equilibrium towards the product without causing thermal degradation of sensitive functional groups. The choice of solvent, ranging from methanol to 1,4-dioxane, plays a critical role in solubilizing the ionic azaarene salts and stabilizing the transition states. The result is a clean reaction profile where the target molecule is the predominant species, simplifying downstream processing. For R&D directors focused on impurity control, this high level of selectivity translates to reduced analytical burdens and faster regulatory approval timelines, as the impurity profile is inherently cleaner compared to stepwise synthetic routes.
How to Synthesize Tetrahydroquinoline Pyrans Efficiently
The practical implementation of this synthesis is straightforward and amenable to standard laboratory and pilot plant equipment. The process begins by charging a reaction vessel with the iridium catalyst, the specific azaarene salt, the 1,3-cyclohexanedione derivative, and paraformaldehyde in a molar ratio of approximately 1:1.5:10. An appropriate base and solvent are added, and the mixture is heated under a protective nitrogen or argon atmosphere to prevent oxidation of the catalyst or intermediates. After the reaction reaches completion, typically within 16 hours, the mixture is cooled, concentrated, and purified using standard column chromatography techniques. This operational simplicity underscores the method's suitability for rapid scale-up and commercial adoption.
- Combine the azaarene salt compound, 1,3-cyclohexanedione compound, paraformaldehyde, iridium catalyst, additive, and solvent in a reaction vessel under a protective atmosphere.
- Heat the reaction mixture to a temperature between 80°C and 90°C, typically 85°C, and maintain stirring for 10 to 20 hours to ensure complete conversion.
- Cool the reaction to room temperature, concentrate the mixture under vacuum, and purify the crude product via column chromatography to isolate the target tetrahydroquinoline pyran.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this iridium-catalyzed methodology offers tangible strategic benefits that extend beyond mere chemical novelty. The elimination of external hydrogen sources and the use of inexpensive, commodity-grade chemicals like paraformaldehyde drastically simplify the raw material sourcing strategy. This reduction in material complexity directly correlates to enhanced supply chain reliability, as the risk of bottlenecks associated with specialty reagents is minimized. Moreover, the one-pot nature of the reaction reduces the number of unit operations required, leading to significant reductions in energy consumption and solvent usage. These factors collectively contribute to a more sustainable and cost-effective manufacturing footprint, aligning with the increasing corporate mandates for green chemistry and environmental stewardship in the fine chemical sector.
- Cost Reduction in Manufacturing: The economic viability of this process is driven by the high atom economy and the avoidance of expensive stoichiometric reductants. By utilizing paraformaldehyde as a dual-purpose reagent, the method eliminates the cost associated with purchasing and handling separate hydrogen donors or metal hydrides. Additionally, the simplified post-treatment process, which often requires only concentration and chromatography, reduces labor costs and waste disposal fees. The ability to achieve high diastereoselectivity also means less material is lost to unwanted isomers, maximizing the yield of the desired pharmaceutical intermediate and optimizing the overall cost per kilogram of the final product.
- Enhanced Supply Chain Reliability: The reliance on cheap and easily obtainable raw materials ensures a stable supply chain that is less susceptible to market volatility. Azaarene salts and 1,3-cyclohexanediones are widely available from multiple global suppliers, reducing the risk of single-source dependency. The robustness of the catalytic system allows for consistent production quality even with slight variations in feedstock, providing a buffer against supply disruptions. This reliability is critical for maintaining continuous production schedules for active pharmaceutical ingredients, ensuring that downstream drug manufacturing processes are not delayed by intermediate shortages.
- Scalability and Environmental Compliance: From an environmental perspective, this green synthesis method generates significantly less waste compared to traditional multi-step routes. The absence of harsh reaction conditions and toxic byproducts simplifies effluent treatment and reduces the environmental impact of the manufacturing facility. The process is inherently scalable, having been designed with industrial production in mind, allowing for seamless transition from gram-scale laboratory synthesis to ton-scale commercial manufacturing. This scalability ensures that as demand for the final drug product grows, the supply of the intermediate can be ramped up quickly without the need for extensive process re-engineering or new capital investment.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis and application of these tetrahydroquinoline pyran compounds. The answers are derived directly from the technical specifications and beneficial effects outlined in the patent literature, providing clarity on the method's capabilities and limitations. Understanding these details is essential for evaluating the feasibility of integrating this technology into existing production pipelines or new drug development programs.
Q: What is the primary advantage of this iridium-catalyzed method over conventional synthesis?
A: The primary advantage is the utilization of paraformaldehyde as both a hydrogen source and a coupling reagent in a one-pot multicomponent reaction. This eliminates the need for additional external hydrogen sources or complex pre-functionalization steps, resulting in high atom economy and simplified post-processing suitable for industrial scale-up.
Q: What are the potential therapeutic applications of these tetrahydroquinoline pyran compounds?
A: According to the patent data, these nitrogen-oxygen heterocyclic compounds exhibit significant potential in the pharmaceutical field, specifically noted for their application in preparing medicaments for treating Parkinson's disease, highlighting their biological relevance as active pharmaceutical ingredients or key intermediates.
Q: Is this synthetic route scalable for commercial manufacturing?
A: Yes, the method is explicitly designed for expanded industrial production. It features mild reaction conditions (80-90°C), uses cheap and easily obtainable raw materials, and avoids harsh additives, making it highly suitable for the commercial scale-up of complex heterocycles with consistent quality.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Tetrahydroquinoline Pyrans Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced catalytic technologies like the iridium-catalyzed hydrogen transfer cyclization described in patent CN113234083B. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from benchtop discovery to full-scale manufacturing is seamless and efficient. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications required for pharmaceutical intermediates, guaranteeing that every batch of tetrahydroquinoline pyrans delivered meets the highest industry standards for quality and consistency.
We invite you to collaborate with our technical team to explore how this innovative synthesis route can optimize your supply chain and reduce costs. Contact us today to request a Customized Cost-Saving Analysis tailored to your specific project needs. Our experts are ready to provide specific COA data and comprehensive route feasibility assessments to help you accelerate your drug development timeline. Partner with NINGBO INNO PHARMCHEM for a reliable, scalable, and cost-effective solution for your complex heterocyclic intermediate requirements.
