Advanced Trivalent Iridium Catalysts: Enabling Green Scale-Up of Complex Pharmaceutical Intermediates
Advanced Trivalent Iridium Catalysts: Enabling Green Scale-Up of Complex Pharmaceutical Intermediates
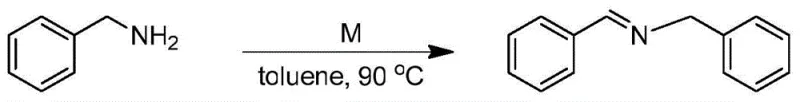
The landscape of pharmaceutical intermediate manufacturing is undergoing a significant transformation driven by the demand for greener, more efficient catalytic systems. A pivotal advancement in this domain is detailed in Chinese Patent CN108620130B, which discloses a novel trivalent iridium complex featuring an ortho-carborane (o-C2B10H10) structure. This innovation addresses critical bottlenecks in the synthesis of imine compounds, which are ubiquitous precursors in the production of active pharmaceutical ingredients (APIs), agrochemicals, and functional materials. By leveraging the unique electronic and steric properties of the carborane cage, this catalyst achieves exceptional stability and activity under mild conditions, utilizing air as a benign oxidant. For R&D directors and procurement strategists, this technology represents a viable pathway to streamline supply chains and reduce the environmental footprint of fine chemical manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the synthesis of imine compounds via the oxidative coupling of primary amines has relied heavily on stoichiometric oxidants or transition metal catalysts that suffer from significant drawbacks. Conventional methods often necessitate the use of hazardous oxidizing agents such as peroxides or hypervalent iodine species, which generate substantial amounts of toxic waste and pose severe safety risks during large-scale operations. Furthermore, many existing catalytic systems require harsh reaction conditions, including high temperatures and inert atmospheres, to achieve acceptable conversion rates. These factors not only inflate the cost of goods sold (COGS) due to expensive reagent consumption and waste disposal but also complicate the regulatory approval process for pharmaceutical intermediates due to residual impurity concerns. The lack of thermal stability in many organometallic catalysts further limits their utility in continuous flow processes or high-throughput manufacturing environments.
The Novel Approach
The technology described in CN108620130B offers a paradigm shift by introducing a robust trivalent iridium complex that operates efficiently under aerobic conditions. Unlike traditional catalysts that degrade rapidly upon exposure to oxygen or heat, this o-carborane-modified iridium species maintains its structural integrity and catalytic activity even at elevated temperatures up to 300°C. The process utilizes simple primary amines and atmospheric air as the oxidant, eliminating the need for costly and dangerous external oxidizing agents. As demonstrated in the patent examples, the system delivers high yields ranging from 82% to 95% across a diverse array of substrates, including those with sensitive functional groups. This approach not only simplifies the reaction workflow but also aligns perfectly with the principles of green chemistry, offering a sustainable alternative for the industrial production of high-value imine derivatives.
Mechanistic Insights into Carborane-Stabilized Iridium Catalysis
The exceptional performance of this catalyst stems from the unique integration of the closo-dodecaborate cluster into the coordination sphere of the iridium center. The o-carborane ligand acts as a bulky, electron-withdrawing scaffold that stabilizes the high-valent iridium(III) state against reductive decomposition. This stabilization is critical for sustaining the catalytic cycle during the oxidative dehydrogenation of amines. The steric bulk provided by the carborane cage likely prevents the formation of inactive dimeric species, ensuring that the active monomeric catalyst remains available throughout the reaction duration. Furthermore, the presence of selenium atoms in the coordination environment enhances the Lewis acidity of the metal center, facilitating the activation of the amine substrate and the subsequent abstraction of hydrogen atoms. This synergistic effect between the carborane ligand and the selenium donors creates a highly tuned electronic environment that promotes rapid turnover frequencies.
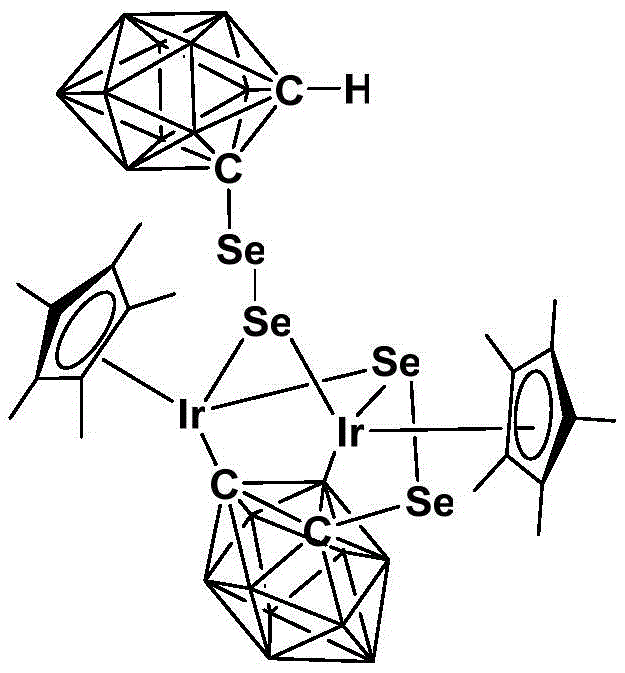
From an impurity control perspective, the robustness of the catalyst translates directly into cleaner reaction profiles. Because the catalyst does not decompose into metallic iridium black or other insoluble particulates under the reaction conditions (80-120°C), the final product mixture contains minimal metal contamination. This is a decisive advantage for pharmaceutical applications where strict limits on heavy metal residues are enforced by regulatory bodies like the FDA and EMA. The homogeneous nature of the reaction allows for precise control over selectivity, minimizing the formation of over-oxidized byproducts such as nitriles or amides, which are common side reactions in less selective systems. The ability to operate with air as the oxidant also precludes the introduction of halogenated or sulfur-based impurities that often accompany the use of chemical oxidants, thereby simplifying downstream purification and reducing the overall impurity burden on the final API intermediate.
How to Synthesize Trivalent Iridium Complex Efficiently
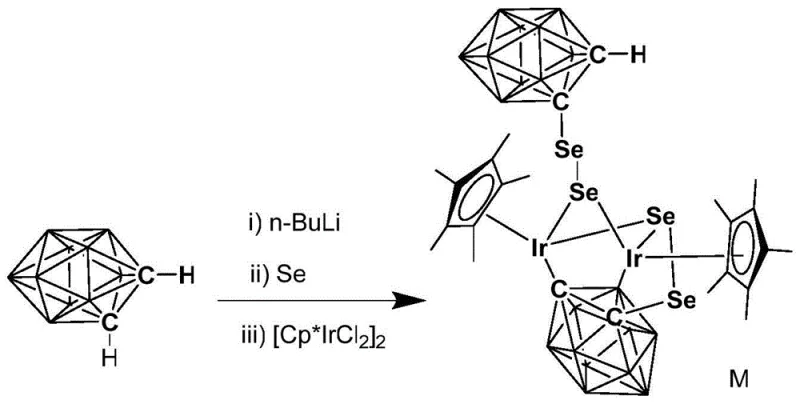
The preparation of this high-performance catalyst is designed for scalability and operational simplicity, avoiding the need for exotic reagents or ultra-low temperature cryogenics beyond standard laboratory capabilities. The synthesis begins with the lithiation of ortho-carborane, followed by selenylation to generate the reactive ligand precursor in situ. This intermediate is then reacted with a commercially available binuclear iridium chloride dimer to assemble the final complex. The entire process can be conducted in standard glassware under inert atmosphere initially, transitioning to ambient conditions for the final isolation. The crude product is easily purified via silica gel column chromatography using common solvent systems like petroleum ether and tetrahydrofuran. This straightforward synthetic route ensures that the catalyst can be produced in multi-kilogram quantities without prohibitive costs, making it a viable option for industrial adoption.
- Lithiation of o-Carborane: Dropwise add n-BuLi in n-hexane to ortho-carborane at -78°C, stir for 40-70 min, then warm to room temperature and react for another 40-70 min.
- Selenylation: Add elemental selenium to the lithiated carborane solution and react continuously at room temperature for 1.5-2.5 hours to form the selenolate intermediate.
- Complexation: Add the binuclear iridium compound [Cp*IrCl2]2 to the system, react for 3-5 hours, filter, remove solvent, and purify via column chromatography (Petroleum Ether/THF) to obtain the red target product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this catalytic technology offers tangible strategic benefits that extend beyond mere chemical efficiency. The primary value driver is the drastic simplification of the raw material portfolio. By replacing expensive, hazardous stoichiometric oxidants with free atmospheric air, manufacturers can significantly reduce direct material costs and eliminate the logistical complexities associated with storing and handling dangerous chemicals. This shift also mitigates supply chain risks related to the volatility of oxidant prices and availability. Furthermore, the high thermal stability of the catalyst implies a longer shelf life and reduced sensitivity to transportation conditions, ensuring consistent quality upon delivery. The simplified workup procedure, which avoids complex extraction protocols needed to remove oxidant byproducts, leads to faster batch cycle times and higher throughput in existing reactor infrastructure.
- Cost Reduction in Manufacturing: The elimination of stoichiometric oxidants represents a major cost saving opportunity. In traditional processes, the cost of oxidants and the subsequent waste treatment can account for a significant portion of the variable costs. By utilizing air, this expense is effectively reduced to zero. Additionally, the high yields (often exceeding 90%) mean that less starting amine material is wasted, improving the overall atom economy of the process. The catalyst loading is low (0.1-0.3 mol%), which minimizes the consumption of the precious iridium metal, further optimizing the cost structure for large-scale production runs.
- Enhanced Supply Chain Reliability: The robustness of the o-carborane iridium complex ensures consistent performance across different batches, reducing the risk of production delays caused by catalyst failure or variability. Since the catalyst is stable at high temperatures and in air, it does not require specialized cold-chain logistics for storage or transport, simplifying inventory management. The use of widely available starting materials like ortho-carborane and binuclear iridium dimers ensures that the supply of the catalyst itself is not constrained by niche sourcing issues, providing a stable foundation for long-term manufacturing planning.
- Scalability and Environmental Compliance: The process generates minimal waste, primarily consisting of water as the byproduct of the oxidative coupling, which drastically reduces the burden on wastewater treatment facilities. This aligns with increasingly stringent environmental regulations and corporate sustainability goals. The simplicity of the reaction setup allows for easy scale-up from pilot plant to commercial production volumes without the need for specialized high-pressure equipment, as the reaction proceeds efficiently at atmospheric pressure. This ease of scale-up accelerates time-to-market for new pharmaceutical intermediates relying on this chemistry.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this trivalent iridium catalyst technology. These insights are derived directly from the experimental data and comparative analysis presented in the patent literature, providing a clear understanding of the operational parameters and expected outcomes for potential adopters.
Q: What are the thermal stability limits of this trivalent iridium catalyst?
A: According to patent CN108620130B, the complex exhibits exceptional thermal stability, remaining structurally intact up to 300°C as confirmed by thermogravimetric analysis, which is crucial for high-temperature industrial processes.
Q: Can this catalyst handle substrates with strong electron-withdrawing groups?
A: Yes, the catalyst demonstrates broad substrate tolerance. It successfully catalyzes the oxidative coupling of primary amines containing strong electron-withdrawing groups like nitro (-NO2) and trifluoromethyl (-CF3) with high yields (92-94%) under air oxidation.
Q: Is the use of toxic oxidants required for this imine synthesis?
A: No, a key advantage of this technology is the use of molecular oxygen (air) as the sole oxidant. This eliminates the need for hazardous stoichiometric oxidants, significantly reducing waste generation and improving process safety.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Trivalent Iridium Complex Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced organometallic catalysis in modern pharmaceutical synthesis. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative technologies like the o-carborane iridium complex can be seamlessly integrated into your supply chain. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of catalyst or intermediate delivered meets the highest international standards. We understand the critical importance of consistency and reliability in the fine chemical industry, and our dedicated technical team is prepared to support your specific process optimization needs.
We invite you to explore how this cutting-edge catalytic system can enhance your manufacturing efficiency and reduce your environmental impact. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific imine synthesis requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to help you make informed decisions about adopting this green and efficient technology for your next project.
