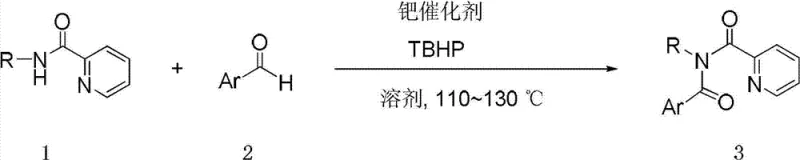
Advanced Palladium-Catalyzed Synthesis of Secondary Diimide Derivatives for Pharmaceutical Applications
The landscape of organic synthesis for nitrogen-containing heterocycles is undergoing a significant transformation, driven by the urgent need for greener, more efficient methodologies in the production of high-value intermediates. A pivotal advancement in this domain is detailed in Chinese Patent CN102731386B, which discloses a robust preparation method for secondary diimide derivatives. This technology represents a paradigm shift from classical acylation strategies to a direct, palladium-catalyzed carbon-hydrogen and nitrogen-hydrogen functionalization protocol. By leveraging the unique coordinating ability of the pyridyl group, this invention enables the coupling of N-substituted-2-pyridinecarboxamides with aromatic aldehydes under remarkably mild and neutral conditions. For R&D directors and process chemists seeking to modernize their synthetic routes, this patent offers a compelling solution that bypasses the limitations of moisture-sensitive reagents, providing a streamlined pathway to structurally complex scaffolds essential for pharmaceutical and agrochemical development.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of secondary imide frameworks has relied heavily on the nucleophilic attack of secondary amides upon activated acylating agents, most notably acid chlorides. While chemically straightforward in theory, this conventional approach suffers from severe practical drawbacks that hinder its applicability in large-scale industrial settings. Acid chlorides are inherently unstable and prone to rapid hydrolysis upon exposure to atmospheric moisture, necessitating stringent anhydrous conditions and specialized handling equipment that drive up operational costs. Furthermore, the generation of stoichiometric amounts of corrosive hydrogen chloride byproduct requires additional neutralization steps and creates significant waste disposal challenges. These harsh acidic environments often prove incompatible with acid-sensitive functional groups, limiting the structural diversity of the final products and forcing chemists to employ cumbersome protecting group strategies that reduce overall atom economy and process efficiency.
The Novel Approach
In stark contrast to the legacy methods, the technology described in CN102731386B introduces a revolutionary oxidative coupling strategy that utilizes readily available aromatic aldehydes as the acyl source. This novel approach eliminates the need for pre-activation of carboxylic acids into acid chlorides, thereby removing a complete synthetic step and its associated hazards. The reaction proceeds under neutral conditions, which preserves the integrity of sensitive functional groups such as esters, ethers, and halides that might otherwise degrade in acidic media. By employing a palladium catalyst in conjunction with tert-butyl peroxide (TBHP) as a green oxidant, the system achieves direct C-H and N-H bond functionalization. This not only simplifies the operational procedure to a simple mix-and-heat protocol but also significantly enhances the safety profile of the manufacturing process, making it an ideal candidate for cost reduction in API manufacturing where safety and simplicity are paramount.
Mechanistic Insights into Pd-Catalyzed C-H/N-H Functionalization
The success of this transformation hinges on the sophisticated interplay between the palladium catalyst and the directing group embedded within the substrate. The N-substituted-2-pyridinecarboxamide serves a dual role: it acts as the nitrogen source for the imide formation and, crucially, the pyridyl nitrogen acts as an intramolecular directing group. This coordination facilitates the activation of the proximal amide N-H bond by the palladium center, lowering the energy barrier for metallation. Once the Pd-N species is formed, it undergoes oxidative addition or insertion with the aldehyde component, mediated by the TBHP oxidant which regenerates the active catalytic species. This mechanistic pathway avoids the high-energy intermediates typical of radical chain reactions, leading to a cleaner reaction profile with fewer side products.
Furthermore, the choice of ligands and oxidation state management is critical for maintaining catalytic turnover. The patent specifies the use of phosphine-ligated palladium complexes, such as bis(triphenylphosphine)palladium acetate, which provide the necessary electronic and steric environment to stabilize the active catalytic cycle. The oxidant, TBHP, plays a vital role in re-oxidizing the reduced palladium species back to its active state, ensuring that only catalytic quantities of the expensive metal are required. This efficient recycling of the catalyst minimizes metal contamination in the final product, a key concern for pharmaceutical intermediates. The neutral nature of the medium ensures that the electron density on the nitrogen remains unperturbed by protonation, thereby sustaining its nucleophilicity towards the activated aldehyde species throughout the reaction duration.
How to Synthesize N-propyl-N-(4-chlorobenzoyl)pyridinecarboxamide Efficiently
To demonstrate the practical viability of this methodology, the patent provides detailed experimental procedures that serve as a blueprint for process optimization. The synthesis of specific derivatives, such as the model compound 3a, highlights the reproducibility and robustness of the system. Operators can achieve high conversion rates by strictly adhering to the specified molar ratios and temperature profiles. The following guide outlines the standardized operational parameters derived from the patent examples, ensuring that technical teams can replicate the high yields reported in the literature while maintaining strict quality control standards suitable for GMP environments.
- Dissolve N-substituted-2-pyridinecarboxamide, aromatic aldehyde, and palladium catalyst (e.g., bis(triphenylphosphine)palladium acetate) in a solvent such as acetonitrile or toluene.
- Dropwise add tert-butyl peroxide (TBHP) into the reaction system at 10-30°C while stirring to ensure homogeneous mixing before heating.
- Raise the temperature to 110-130°C and stir for 1-100 hours until reaction completion, followed by separation and purification via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this palladium-catalyzed technology translates into tangible strategic benefits that extend beyond mere chemical elegance. The primary advantage lies in the drastic simplification of the raw material supply chain. By replacing acid chlorides with aromatic aldehydes, manufacturers can source starting materials from a much broader and more stable global market, as aldehydes are commodity chemicals with established production scales. This shift mitigates the risk of supply disruptions often associated with specialized, hazardous reagents. Additionally, the elimination of the acid chloride preparation step reduces the overall processing time and energy consumption, leading to substantial cost savings in utility and labor. The neutral reaction conditions also reduce wear and tear on reactor vessels, extending equipment lifespan and lowering maintenance overheads.
- Cost Reduction in Manufacturing: The economic impact of this process is profound due to the removal of multiple unit operations. Traditional routes require separate reactors for acid chloride generation, often involving thionyl chloride or oxalyl chloride, followed by a distinct coupling step. This new method consolidates these into a single pot, significantly reducing solvent usage and waste treatment costs. Although palladium is a precious metal, its usage is catalytic (as low as 10 mol% or less), and the high turnover number ensures that the cost per kilogram of product remains competitive. The avoidance of corrosive byproducts further lowers the cost of waste neutralization and disposal, contributing to a leaner manufacturing budget without compromising on output quality.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of stable, shelf-stable starting materials. Aromatic aldehydes and N-substituted amides do not require the cold chain logistics or inert atmosphere storage that acid chlorides demand. This simplifies warehousing requirements and reduces the risk of raw material degradation during transit. Furthermore, the broad substrate scope means that if a specific aldehyde becomes unavailable, structurally similar alternatives can often be substituted with minimal process re-validation, providing flexibility in sourcing strategies. This adaptability is crucial for maintaining continuous production schedules in the face of global market volatility.
- Scalability and Environmental Compliance: From an environmental and scalability perspective, this technology aligns perfectly with modern green chemistry principles. The reaction operates at moderate temperatures (110-130°C), which are easily achievable in standard stainless steel reactors without the need for extreme cooling or heating infrastructure. The use of TBHP as an oxidant generates tert-butanol as a byproduct, which is less hazardous than the HCl gas produced in conventional methods. This makes the process easier to scale from pilot plant to commercial production (100 kgs to 100 MT) while meeting increasingly stringent environmental regulations regarding emissions and effluent treatment.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation of this synthesis method. These answers are derived directly from the experimental data and claims within the patent documentation, providing clarity on reaction scope, optimization, and troubleshooting. Understanding these nuances is essential for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios.
Q: What are the advantages of this neutral condition synthesis over traditional acid chloride methods?
A: Traditional methods using acid chlorides are limited by hydrolysis sensitivity and harsh acidic conditions. This novel Pd-catalyzed approach operates under neutral conditions, offering wide functional group tolerance and eliminating the need for pre-activating carboxylic acids, thereby simplifying the workflow and reducing waste.
Q: What is the optimal catalyst loading and oxidant ratio for this transformation?
A: According to the patent data, the optimal molar ratio involves a catalytic amount of palladium (0.01-0.5 equivalents relative to aldehyde) and tert-butyl peroxide (TBHP) as the oxidant. Specifically, a ratio of Aromatic Aldehyde : Amide : TBHP : Pd catalyst of 1 : 2 : 2 : 0.1 has shown superior yields.
Q: Can this method tolerate diverse substituents on the aromatic ring?
A: Yes, the method demonstrates excellent substrate scope. It tolerates various substituents including halogens (F, Cl, Br), alkyl groups (methyl), and alkoxy groups (methoxy) on both the aromatic aldehyde and the N-substituent, making it highly versatile for generating diverse chemical libraries.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Secondary Diimide Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the palladium-catalyzed synthesis route described in CN102731386B for the production of high-purity secondary diimide derivatives. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from laboratory discovery to market supply is seamless. Our facility is equipped with state-of-the-art rigorous QC labs capable of monitoring trace metal residues and impurity profiles, guaranteeing that every batch meets stringent purity specifications required by top-tier pharmaceutical clients. We are committed to leveraging this advanced chemistry to deliver cost-effective solutions that enhance your supply chain efficiency.
We invite you to collaborate with our technical procurement team to explore how this innovative methodology can be tailored to your specific project needs. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic benefits of switching to this neutral, one-pot synthesis. We encourage you to contact us today to discuss your requirements, obtain specific COA data for our catalog compounds, and receive comprehensive route feasibility assessments that will accelerate your development timelines and secure your position in the competitive global market.
