Revolutionizing (R)-Fluoxetine Production: A Cost-Effective Nickel-Catalyzed Asymmetric Hydrogenation Strategy
The pharmaceutical industry is constantly seeking more sustainable and cost-effective pathways for the synthesis of high-volume active pharmaceutical ingredients (APIs), particularly for widely prescribed antidepressants like (R)-Fluoxetine. A groundbreaking approach detailed in patent CN114605272A introduces a novel preparation method that fundamentally shifts the catalytic paradigm from expensive noble metals to earth-abundant Nickel. This technology leverages a sophisticated Nickel-catalyzed asymmetric hydrogenation system to construct the critical chiral gamma-aryl lactone intermediate with exceptional precision. By replacing traditional precious metal catalysts with a robust Nickel-perchlorate and chiral phosphine ligand complex, this method addresses long-standing economic and environmental challenges in chiral drug manufacturing. The significance of this development lies not only in its chemical elegance but also in its potential to drastically lower the cost of goods sold (COGS) for generic manufacturers while maintaining stringent quality standards required by global regulatory bodies.
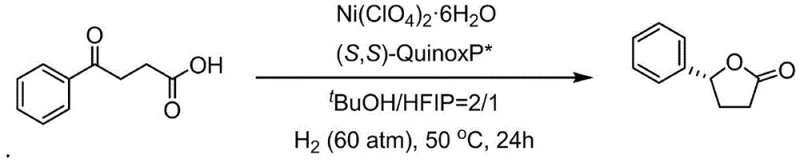
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of chiral intermediates for (R)-Fluoxetine has relied heavily on asymmetric hydrogenation using precious metal catalysts such as Rhodium, Ruthenium, or Iridium complexes. While these noble metal systems are known for their high activity, they present significant drawbacks for large-scale commercial operations. The primary limitation is the exorbitant cost of the catalysts themselves, which are subject to volatile market prices and geopolitical supply constraints. Furthermore, the removal of trace heavy metal residues from the final API is a rigorous and expensive process, often requiring additional purification steps like scavenging or recrystallization to meet strict ICH guidelines. Conventional routes may also suffer from moderate enantioselectivity, necessitating chiral resolution steps that inherently cap the maximum theoretical yield at 50% unless dynamic kinetic resolution is employed, thereby increasing waste and processing time.
The Novel Approach
The methodology disclosed in CN114605272A offers a transformative alternative by utilizing a Nickel-based catalytic system that operates with high efficiency and selectivity. This novel approach initiates with the asymmetric hydrogenation of aryl gamma-keto acids, a transformation that was previously difficult to achieve with base metals at this level of performance. The process employs Nickel perchlorate hexahydrate as the metal source and the (S,S)-QuinoxP* ligand to induce chirality, operating effectively in a mixed solvent system of tert-butanol and hexafluoroisopropanol. As illustrated in the reaction scheme below, this system successfully converts achiral keto acids into chiral gamma-aryl lactones with yields reaching up to 95% and enantiomeric excess values around 93-96%. This high level of stereocontrol eliminates the need for resolution, effectively doubling the material throughput compared to non-enantioselective routes.
Mechanistic Insights into Nickel-Catalyzed Asymmetric Hydrogenation
The core of this technological breakthrough lies in the unique coordination chemistry between the Nickel center and the chiral QuinoxP* ligand. In the catalytic cycle, the Nickel(II) species is reduced in situ to the active Nickel(0) or Nickel(II)-hydride species, which then coordinates with the carbonyl oxygen and the alkene moiety of the gamma-keto acid substrate. The bulky chiral environment created by the QuinoxP* ligand dictates the facial selectivity of the hydrogen addition, ensuring that the hydride transfer occurs exclusively to one pro-chiral face of the ketone. This precise spatial arrangement is critical for establishing the (R)-configuration at the benzylic position. Unlike noble metals that might rely on different electronic effects, this Nickel system leverages specific Lewis acidic interactions enhanced by the fluorinated alcohol solvents, which likely stabilize the transition state through hydrogen bonding networks. This mechanistic understanding allows for fine-tuning of reaction parameters, such as hydrogen pressure (optimized at 60 atm in examples) and temperature (50°C), to maximize turnover numbers.
From an impurity control perspective, the high enantioselectivity of this Nickel-catalyzed step is paramount for downstream processing. By generating the chiral lactone with an ee value exceeding 90% directly from the hydrogenation reactor, the burden on subsequent purification units is significantly reduced. In traditional syntheses where racemic mixtures are produced, half of the material is the unwanted enantiomer, which can act as a persistent impurity throughout the synthesis, complicating crystallization and potentially co-eluting in chromatography. The high fidelity of this asymmetric hydrogenation ensures that the impurity profile remains clean, facilitating easier isolation of the key oxazinone precursor in later steps. This purity advantage translates directly into higher overall process yields and reduced solvent consumption during workup, aligning with green chemistry principles.
How to Synthesize (R)-Fluoxetine Intermediates Efficiently
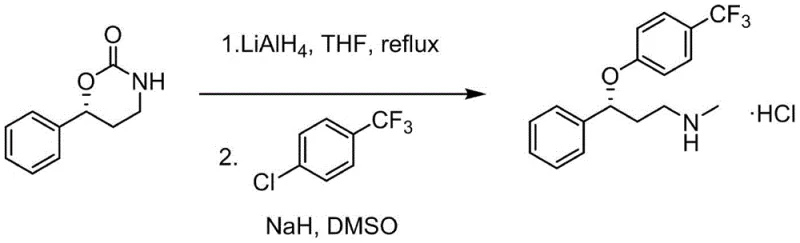
The synthesis of high-purity (R)-Fluoxetine intermediates via this patented route involves a streamlined sequence of transformations that are amenable to standard chemical manufacturing infrastructure. The process begins with the critical asymmetric hydrogenation step described previously, followed by a cascade of functional group manipulations including aminolysis, Hofmann rearrangement, and reduction. Each step has been optimized to minimize side reactions and maximize atom economy. For R&D teams looking to implement this technology, the protocol emphasizes the importance of maintaining anhydrous conditions during the catalyst preparation and utilizing specific solvent ratios to ensure optimal ligand coordination. The subsequent conversion of the lactone to the amine functionality is achieved through a robust two-step sequence involving ammonolysis to open the ring and oxidative rearrangement to contract it, setting the stage for the final reductive amination-like steps.
- Perform asymmetric hydrogenation of aryl gamma-keto acids using a Nickel perchlorate hexahydrate catalyst and (S,S)-QuinoxP* ligand under hydrogen pressure to obtain chiral gamma-aryl lactones.
- Subject the chiral lactone to aminolysis with ammonium hydroxide, followed by Hofmann rearrangement using iodobenzene diacetate to form the key oxazinone precursor.
- Reduce the oxazinone intermediate with lithium aluminum hydride and perform nucleophilic substitution with 1-chloro-4-(trifluoromethyl)benzene to yield the final (R)-Fluoxetine derivative.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this Nickel-catalyzed route represents a strategic opportunity to optimize the cost structure and resilience of the API supply chain. The shift from precious metals to base metals is not merely a technical adjustment but a significant financial lever that impacts the bottom line. By eliminating the dependency on Rhodium or Ruthenium, manufacturers can insulate themselves from the extreme price volatility associated with platinum group metals. Furthermore, the simplified purification requirements resulting from high enantioselectivity reduce the consumption of expensive chromatography media and solvents, leading to substantial operational expenditure savings. The use of commercially available and stable Nickel salts also simplifies inventory management and reduces the lead time for raw material acquisition.
- Cost Reduction in Manufacturing: The replacement of expensive noble metal catalysts with affordable Nickel salts results in a direct and significant decrease in raw material costs. Since the catalyst loading is low and the metal is inexpensive, the overall catalyst cost per kilogram of product is drastically reduced compared to traditional methods. Additionally, the high yield and selectivity minimize waste generation, lowering the costs associated with waste disposal and solvent recovery. The elimination of chiral resolution steps further enhances cost efficiency by ensuring that nearly all starting material is converted into the desired enantiomer, maximizing asset utilization.
- Enhanced Supply Chain Reliability: Nickel is an earth-abundant element with a stable and diverse global supply chain, unlike precious metals which are often sourced from geopolitically sensitive regions. This abundance ensures a consistent availability of the catalyst precursor, reducing the risk of supply disruptions that could halt production lines. The reagents used in the subsequent steps, such as ammonium hydroxide and lithium aluminum hydride, are commodity chemicals available from multiple suppliers worldwide. This diversification of the supply base enhances the overall resilience of the manufacturing process against market shocks.
- Scalability and Environmental Compliance: The process is designed for scalability, utilizing standard high-pressure hydrogenation reactors that are common in fine chemical plants. The absence of toxic heavy metals like Rhodium simplifies the regulatory compliance landscape, particularly regarding residual metal limits in the final drug substance. This ease of compliance accelerates the technology transfer from pilot scale to commercial production. Moreover, the reduced solvent usage and higher atom economy contribute to a smaller environmental footprint, aligning with corporate sustainability goals and increasingly stringent environmental regulations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this Nickel-catalyzed synthesis route. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing a clear picture of the technology's capabilities and limitations. Understanding these details is crucial for technical teams evaluating the feasibility of adopting this new methodology for their specific production needs.
Q: What is the primary advantage of using Nickel catalysis over traditional noble metals for (R)-Fluoxetine synthesis?
A: The primary advantage is substantial cost reduction and improved sustainability. Nickel is an earth-abundant base metal, significantly cheaper than traditional Rhodium, Ruthenium, or Iridium catalysts, while the patented system achieves comparable high yields and enantioselectivity without requiring expensive additives.
Q: How does this process ensure high optical purity for pharmaceutical applications?
A: The process utilizes a highly stereoselective Nickel catalyst system paired with the (S,S)-QuinoxP* chiral ligand. Patent data demonstrates that this combination consistently delivers chiral gamma-aryl lactones with excellent enantiomeric excess (ee) values, often exceeding 90%, which minimizes the need for costly downstream chiral resolution.
Q: Is this synthetic route suitable for large-scale commercial production?
A: Yes, the route is designed for scalability. It employs standard hydrogenation equipment and commercially available reagents like Nickel perchlorate and common solvents. The elimination of sensitive noble metals simplifies supply chain logistics and reduces the regulatory burden associated with heavy metal residues in the final API.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (R)-Fluoxetine Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this Nickel-catalyzed technology for the global antidepressant market. As a leading CDMO partner, we possess the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory processes are seamlessly translated into robust industrial operations. Our facility is equipped with state-of-the-art high-pressure hydrogenation units and rigorous QC labs capable of monitoring enantiomeric purity with precision. We are committed to delivering high-purity pharmaceutical intermediates that meet the stringent purity specifications required by major regulatory agencies, leveraging our deep expertise in chiral synthesis to support your drug development pipeline.
We invite you to collaborate with us to evaluate the feasibility of this cost-effective route for your specific requirements. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis comparing this Nickel-based method against your current supply chain metrics. Please contact us to request specific COA data and route feasibility assessments, and let us demonstrate how our advanced manufacturing capabilities can drive efficiency and reliability in your (R)-Fluoxetine supply chain.
