Advanced Synthesis of Alpha-Aryl Nitriles via Mild Sulfoxide Activation for Commercial Scale-Up
Advanced Synthesis of Alpha-Aryl Nitriles via Mild Sulfoxide Activation for Commercial Scale-Up
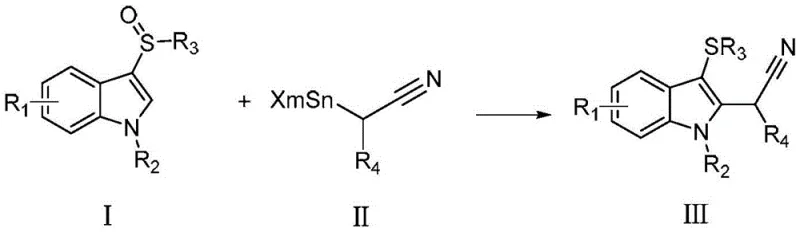
The pharmaceutical and fine chemical industries are constantly seeking robust, scalable methodologies for constructing complex molecular architectures, particularly those containing the versatile alpha-aryl nitrile motif. As detailed in the groundbreaking patent CN108409628B, a novel synthetic strategy has been developed that fundamentally shifts the paradigm for accessing these critical structural units. This technology leverages the unique reactivity of aryl sulfoxides coupled with alpha-tin substituted nitriles in the presence of acid anhydrides, offering a pathway that is both chemically elegant and commercially viable. For R&D directors and procurement specialists alike, this represents a significant opportunity to streamline the supply chain for key drug intermediates such as anastrozole and diphenoxylate derivatives. The method distinguishes itself by operating under exceptionally mild conditions, typically between -80°C and 0°C, thereby preserving sensitive functional groups that might otherwise degrade under traditional harsh synthetic protocols.
Furthermore, the versatility of this approach allows for the accommodation of a wide array of substituents on the aromatic ring, including halogens, alkyl groups, alkoxy groups, and even electron-withdrawing nitro or cyano groups. This broad substrate scope is crucial for medicinal chemists who require diverse libraries for structure-activity relationship (SAR) studies without being constrained by the limitations of previous methodologies. By utilizing readily available starting materials and avoiding the use of toxic metal cyanides or expensive noble metal catalysts, this process aligns perfectly with modern green chemistry principles while simultaneously addressing cost pressures. The following analysis delves deep into the mechanistic underpinnings and commercial implications of this transformative technology, positioning it as a cornerstone for future pharmaceutical intermediate manufacturing strategies.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the carbon-carbon bond adjacent to a nitrile group has been fraught with significant challenges that hinder efficient large-scale production. Traditional methods often rely on the coupling of aryl halides with cyano compounds under strongly basic conditions, a approach that severely restricts the scope of compatible substrates due to the sensitivity of many functional groups to strong bases. Alternatively, the cyanation of benzyl alcohols or halides, while sometimes effective, frequently necessitates the use of highly toxic metal cyanide sources, posing severe safety and environmental disposal risks that complicate regulatory compliance in GMP environments. Another common route involves palladium-catalyzed cross-coupling reactions, which, despite their utility, introduce the burden of expensive catalyst costs and the rigorous requirement for removing trace heavy metals from the final active pharmaceutical ingredient (API). These legacy methods often suffer from narrow substrate tolerance, requiring high temperatures that can lead to decomposition, or they involve multi-step sequences that erode overall yield and increase waste generation.
The Novel Approach
In stark contrast, the methodology disclosed in patent CN108409628B introduces a streamlined, one-pot transformation that bypasses these historical bottlenecks through the activation of aryl sulfoxides. By reacting structure (I) aryl sulfoxides with structure (II) alpha-tin substituted nitriles in the presence of an acid anhydride activator, the process achieves high conversion rates with exceptional selectivity. This novel route eliminates the need for transition metal catalysts entirely, thereby removing the costly and time-consuming steps associated with metal scavenging and validation. The reaction proceeds smoothly in common solvents like dichloromethane or acetonitrile at low temperatures, ensuring that thermally labile groups remain intact. Moreover, the byproducts generated are generally easier to separate than those from traditional cyanation reactions, facilitating a cleaner isolation of the target high-purity alpha-aryl nitrile compounds. This shift not only enhances operational safety but also drastically simplifies the downstream processing workflow, making it an ideal candidate for cost reduction in API manufacturing.
Mechanistic Insights into Sulfoxide-Tin Coupling Activation
The success of this synthetic route lies in the precise activation of the sulfoxide moiety, which serves as a latent electrophile upon treatment with an acid anhydride such as trifluoroacetic anhydride (TFAA). The mechanism initiates with the oxygen atom of the sulfoxide attacking the anhydride, generating a highly reactive sulfonium intermediate. This activated species is then susceptible to nucleophilic attack by the alpha-carbon of the organotin nitrile reagent. The tin group, acting as a stabilizing element for the carbanion character during the transition state, facilitates the formation of the new carbon-carbon bond with high stereochemical control. Following the nucleophilic addition, the elimination of the tin-containing leaving group occurs, driven by the stability of the resulting nitrile functionality and the restoration of aromaticity or conjugation within the system. This concerted sequence ensures that the reaction proceeds with minimal side reactions, such as polymerization or over-alkylation, which are common pitfalls in other alkylation strategies.
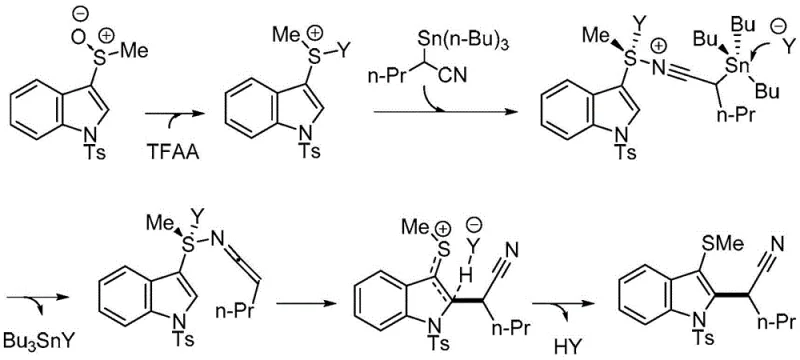
From an impurity control perspective, this mechanism offers distinct advantages because the reaction pathway is highly defined and does not generate radical species that could lead to complex impurity profiles. The use of the tin reagent is carefully balanced; the molar ratio of aryl sulfoxide to the alpha-tin substituted nitrile is optimized between 1:1.1 and 1:2, with a preferred ratio of 1:1.5 to ensure complete consumption of the limiting reagent while minimizing excess organometallic waste. The acid anhydride is similarly dosed at a ratio of 1.1 to 2 equivalents relative to the sulfoxide, ensuring sufficient activation without promoting excessive acid-mediated degradation of the product. Understanding these stoichiometric nuances is vital for process chemists aiming to replicate the high yields reported in the patent, which range from moderate to excellent depending on the electronic nature of the substituents. The ability to tune the reaction by selecting different anhydrides (e.g., TFAA, methanesulfonic anhydride) further provides a handle for optimizing kinetics and solubility profiles for specific substrates.
How to Synthesize Alpha-Aryl Nitrile Efficiently
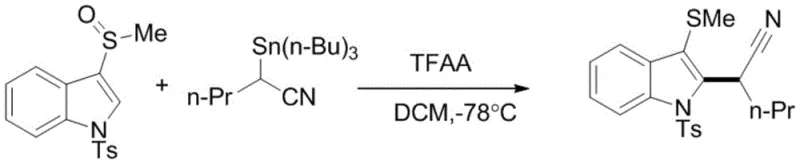
Implementing this synthesis requires strict adherence to the controlled low-temperature conditions and inert atmosphere protocols outlined in the experimental examples. The process begins with the preparation of dry, distilled solvents to prevent moisture-induced hydrolysis of the acid anhydride activator. Operators must ensure that the reaction mixture is maintained under nitrogen protection throughout the addition of reagents to avoid oxidation of the sensitive tin species. The addition sequence is critical: typically, the sulfoxide and tin reagent are dissolved first, followed by the slow introduction of the anhydride to manage the exotherm and control the formation of the reactive intermediate. Monitoring the reaction progress via thin-layer chromatography (TLC) is essential, as reaction times can vary from as short as 10 minutes to as long as 15 hours depending on the steric hindrance of the substrates. Upon completion, the quenching step with saturated sodium bicarbonate must be performed carefully to neutralize acidic byproducts before proceeding to extraction and purification.
- Prepare the reaction vessel under inert atmosphere (N2) and cool the solvent (DCM or MeCN) to temperatures between -80°C and 0°C.
- Add the aryl sulfoxide substrate and the alpha-tin substituted nitrile compound to the solvent, maintaining the molar ratio of sulfoxide to tin reagent at approximately 1: 1.5.
- Introduce the acid anhydride activator (such as TFAA) slowly, stir for the specified duration (10min to 15h), and quench with saturated bicarbonate solution before purification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this sulfoxide-tin coupling technology presents a compelling value proposition centered on risk mitigation and operational efficiency. By transitioning away from palladium-catalyzed routes, manufacturers can eliminate the volatility associated with precious metal pricing and the supply chain disruptions often linked to rare earth sourcing. The reliance on bulk commodity chemicals like acid anhydrides and standard organic solvents ensures a stable and predictable raw material supply base, reducing the risk of production stoppages due to reagent shortages. Furthermore, the simplified workup procedure, which avoids complex filtration steps for catalyst removal, translates directly into reduced cycle times and lower labor costs per batch. This efficiency gain is particularly impactful when scaling from pilot plant to full commercial production, where even minor reductions in unit operation time can result in substantial throughput increases.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts removes a significant line item from the bill of materials, while the avoidance of toxic cyanide salts reduces the costs associated with specialized hazardous waste disposal and safety infrastructure. The high atom economy of the reaction, combined with the ability to recover and potentially recycle solvent streams, contributes to a leaner manufacturing cost structure. Additionally, the mild reaction conditions reduce energy consumption compared to processes requiring prolonged heating or cryogenic cooling below -80°C for extended periods, further driving down utility expenses. These cumulative savings allow for a more competitive pricing strategy in the global market for pharmaceutical intermediates.
- Enhanced Supply Chain Reliability: The robustness of the reaction across a wide range of substrates means that a single manufacturing platform can be adapted to produce multiple derivatives, increasing asset utilization and flexibility. The use of stable, shelf-stable reagents minimizes the need for just-in-time delivery of sensitive materials, allowing for larger inventory buffers that protect against supply shocks. The simplicity of the purification process, often achievable through standard column chromatography or crystallization, reduces dependency on specialized contract manufacturing organizations for difficult separations. This self-sufficiency strengthens the supply chain resilience, ensuring consistent delivery schedules to downstream API manufacturers.
- Scalability and Environmental Compliance: The process generates fewer hazardous byproducts compared to traditional cyanation methods, aligning with increasingly stringent environmental regulations and corporate sustainability goals. The absence of heavy metals simplifies the environmental impact assessment and permitting process for new production lines. Scalability is supported by the exothermic nature of the activation step, which can be managed effectively in larger reactors through controlled dosing, ensuring safe commercial scale-up of complex heterocyclic intermediates. The overall greener profile of the synthesis enhances the brand reputation of the manufacturer as a responsible partner in the pharmaceutical value chain.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis route. These insights are derived directly from the experimental data and claims within the patent documentation, providing a factual basis for decision-making. Understanding these details helps stakeholders evaluate the feasibility of integrating this technology into their existing production portfolios. It clarifies the operational boundaries and highlights the specific advantages that differentiate this method from legacy processes currently in use.
Q: What are the primary advantages of this sulfoxide-tin coupling method over traditional palladium catalysis?
A: This method eliminates the need for expensive transition metal catalysts like palladium, significantly reducing raw material costs and simplifying the removal of heavy metal impurities, which is critical for pharmaceutical compliance.
Q: Does this process require harsh reaction conditions or strong bases?
A: No, the process operates under mild conditions ranging from -80°C to 0°C using acid anhydrides as activators, avoiding the safety hazards and substrate limitations associated with strong base systems.
Q: Is this synthesis route suitable for large-scale industrial production?
A: Yes, the reaction demonstrates high selectivity and yields up to 96% in laboratory settings, with simple workup procedures involving standard extraction and chromatography, indicating strong potential for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Alpha-Aryl Nitrile Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the sulfoxide-tin coupling methodology for the next generation of pharmaceutical intermediates. As a leading CDMO partner, we possess the technical expertise and infrastructure to translate this laboratory-scale innovation into robust, GMP-compliant manufacturing processes. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the high yields and purity profiles demonstrated in the patent are maintained at an industrial level. We operate rigorous QC labs equipped with advanced analytical instrumentation to verify stringent purity specifications, guaranteeing that every batch of alpha-aryl nitrile meets the exacting standards required by global regulatory bodies. Our commitment to quality assurance extends to every step of the synthesis, from raw material qualification to final product release.
We invite forward-thinking pharmaceutical companies to collaborate with us to leverage this cost-effective and environmentally friendly synthesis route. By partnering with our technical team, you can access a Customized Cost-Saving Analysis tailored to your specific molecule, identifying opportunities to optimize your supply chain and reduce overall COGS. We encourage you to contact our technical procurement team today to request specific COA data and comprehensive route feasibility assessments. Let us help you secure a reliable supply of high-quality intermediates while driving innovation and efficiency in your drug development programs.
