Scalable Metal-Free Synthesis of 5-Selenoquinoline Derivatives for Advanced Drug Discovery
Introduction to Regioselective Selenization Technology
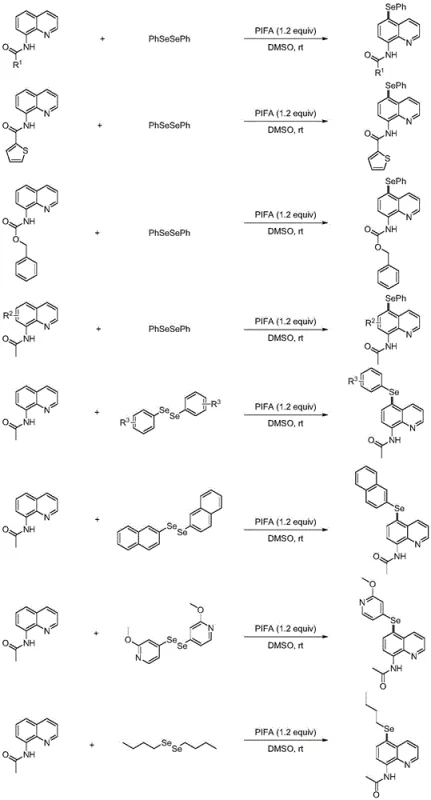
The development of efficient methods for the late-stage functionalization of heterocyclic cores remains a cornerstone of modern medicinal chemistry. Specifically, the introduction of selenium atoms into privileged scaffolds like quinoline can significantly enhance biological activity and metabolic stability. A groundbreaking approach detailed in patent CN112979545B describes a novel preparation method for 5-selenoquinoline amides and esters. This technology utilizes bis(trifluoroacetoxy)iodobenzene (PIFA) as a mild, metal-free oxidant to achieve direct C-H selenization at the challenging C-5 position. Unlike traditional transition-metal catalyzed processes, this protocol operates under ambient conditions, offering a streamlined pathway for generating high-value pharmaceutical intermediates. The ability to access these structurally complex molecules with high regioselectivity addresses a critical gap in the synthesis of selenium-containing bioactive compounds, providing a robust foundation for drug discovery programs targeting various therapeutic areas.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the direct functionalization of the quinoline nucleus at the C-5 position has been fraught with significant synthetic hurdles. Conventional strategies often rely on transition metal catalysts, particularly copper-based systems, which necessitate harsh reaction conditions such as elevated temperatures and controlled oxygen atmospheres. These rigorous requirements not only increase energy consumption but also introduce the risk of heavy metal contamination in the final product, a critical concern for pharmaceutical manufacturing. Furthermore, the geometric inaccessibility of the C-5 site often leads to poor regioselectivity, resulting in complex mixtures of isomers that are difficult and costly to separate. The reliance on stoichiometric metal salts and the need for inert gas protection further complicate the operational workflow, making scale-up economically unfeasible for many process chemistry teams seeking reliable supply chains for complex intermediates.
The Novel Approach
In stark contrast, the methodology disclosed in the patent leverages hypervalent iodine chemistry to overcome these barriers through a radical-mediated mechanism. By employing PIFA as the oxidant, the reaction proceeds efficiently at room temperature in an open air atmosphere, drastically simplifying the operational setup. The use of dimethyl sulfoxide (DMSO) for amides and acetonitrile (MeCN) for esters ensures optimal solubility and reaction kinetics without the need for exotic ligands or anhydrous conditions. This metal-free approach eliminates the downstream burden of metal scavenging, directly translating to cost reduction in pharmaceutical intermediate manufacturing. The protocol demonstrates exceptional tolerance for various functional groups on the diselenide substrate, allowing for the rapid generation of diverse chemical libraries. This shift from metal-catalysis to organo-oxidation represents a paradigm shift in how chemists approach the synthesis of 5-substituted quinolines.
Mechanistic Insights into PIFA-Mediated Radical Selenization
The success of this transformation lies in the unique reactivity profile of PIFA, which acts as a single-electron oxidant to generate reactive selenium species in situ. Upon interaction with the diselenide substrate, PIFA facilitates the homolytic cleavage of the Se-Se bond, generating a selenyl radical and a trifluoroacetoxyiodobenzene radical. This selenyl radical serves as the key electrophilic species that attacks the electron-rich quinoline ring. Due to the specific electronic distribution and the directing effect of the amide or ester group at the 8-position, the radical addition occurs selectively at the C-5 position. This regioselectivity is driven by the stabilization of the resulting radical intermediate through resonance with the adjacent nitrogen atom and the carbonyl group, ensuring that the reaction pathway favors the desired isomer over other potential sites of functionalization on the heterocyclic core.
Following the initial radical addition, the resulting carbon-centered radical intermediate undergoes a second oxidation event mediated by the iodine(III) species present in the reaction mixture. This step converts the neutral radical into a cationic selenonium intermediate, which is highly unstable. The final step involves a spontaneous deprotonation at the C-5 position to restore aromaticity, yielding the stable 5-selenoquinoline product. This mechanistic pathway avoids the formation of stable organometallic complexes that often plague transition metal catalysis, thereby minimizing side reactions and impurity formation. The clean conversion profile observed in the patent examples, with yields reaching up to 96%, underscores the efficiency of this radical relay process. Understanding this mechanism is crucial for process optimization, as it highlights the importance of maintaining the correct oxidant stoichiometry to drive the reaction to completion without over-oxidation.
How to Synthesize 5-Selenoquinoline Amides Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires adherence to specific stoichiometric ratios and solvent choices to maximize yield and purity. The general procedure involves dissolving the 8-aminoquinoline amide substrate and the corresponding diselenide in DMSO, followed by the portion-wise addition of PIFA. Reaction monitoring via TLC indicates that most transformations reach completion within a short timeframe, typically ranging from 0.5 to 12 hours depending on the electronic nature of the substituents. Workup procedures are straightforward, involving aqueous quenching and extraction with dichloromethane, followed by standard purification techniques. For detailed operational parameters and safety guidelines regarding the handling of hypervalent iodine reagents, please refer to the standardized synthesis steps provided below.
- Prepare the reaction mixture by combining 8-aminoquinoline amide substrate and diselenide compound in a molar ratio of 1: 1 in dimethyl sulfoxide (DMSO) solvent.
- Add PIFA (bis(trifluoroacetoxy)iodobenzene) as the oxidant in a molar ratio of 1.2 equivalents relative to the substrate to initiate the radical selenization process.
- Stir the reaction at room temperature under air atmosphere until completion, then perform aqueous workup and column chromatography to isolate the pure 5-selenoquinoline product.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this PIFA-mediated protocol offers compelling advantages that align with the strategic goals of modern pharmaceutical supply chains. The elimination of transition metal catalysts removes a major bottleneck in the purification process, significantly reducing the cost associated with metal scavengers and analytical testing for residual metals. Furthermore, the ability to run reactions at room temperature under air atmosphere lowers the energy footprint of the manufacturing process, contributing to more sustainable production practices. The use of commercially available and inexpensive starting materials, such as simple diselenides and quinoline derivatives, ensures a stable and cost-effective supply of raw materials. This robustness makes the technology highly attractive for large-scale production where consistency and cost control are paramount.
- Cost Reduction in Manufacturing: The primary economic driver of this technology is the complete avoidance of precious or base metal catalysts. In traditional copper-catalyzed routes, the cost of the catalyst combined with the expensive ligands and the subsequent metal removal resins can account for a significant portion of the COGS. By switching to a PIFA-based system, manufacturers can eliminate these line items entirely. Additionally, the simplified workup procedure reduces solvent consumption and labor hours required for purification. The high yields reported across a broad substrate scope mean that less raw material is wasted, further enhancing the overall atom economy and financial efficiency of the process for high-purity pharmaceutical intermediates.
- Enhanced Supply Chain Reliability: Supply chain resilience is often compromised by the reliance on specialized catalysts that may have long lead times or limited suppliers. PIFA and diselenides are commodity chemicals available from multiple global vendors, mitigating the risk of supply disruption. The mild reaction conditions also reduce the dependency on specialized high-pressure or high-temperature reactor equipment, allowing for greater flexibility in manufacturing site selection. This adaptability ensures that production can be scaled up rapidly to meet market demand without the need for significant capital investment in new infrastructure, thereby reducing lead time for high-purity intermediates.
- Scalability and Environmental Compliance: Scaling chemical processes often introduces safety and environmental challenges, particularly when dealing with exothermic metal-catalyzed reactions or oxygen-sensitive protocols. This room-temperature, air-stable method inherently possesses a safer thermal profile, making it easier to scale from gram to kilogram quantities. The absence of heavy metals simplifies waste stream management, as the effluent does not require specialized treatment for metal recovery or disposal. This alignment with green chemistry principles not only reduces environmental compliance costs but also enhances the corporate sustainability profile of the manufacturing entity, a key factor for partnerships with major multinational pharmaceutical companies.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and scope of this selenization technology. These insights are derived directly from the experimental data and comparative analysis presented in the patent documentation. Understanding these nuances is essential for R&D teams evaluating this route for their specific pipeline candidates. The answers provided reflect the current state of the art as defined by the intellectual property.
Q: Why is the C-5 position of quinoline difficult to functionalize compared to other positions?
A: From a geometric perspective, the carbon-5 position of the quinoline core is sterically less accessible than the C-2, C-3, C-4, and C-8 positions. Traditional electrophilic substitution often favors more activated sites, making direct C-5 selective functionalization a significant synthetic challenge that requires specialized directing groups or specific catalytic systems.
Q: What are the advantages of using PIFA over traditional copper catalysts for this selenization?
A: Traditional methods often rely on copper catalysts which require high temperatures and oxygen atmospheres, leading to potential metal contamination and higher energy costs. The PIFA-mediated method operates at room temperature in air, eliminating the need for expensive transition metals and complex removal steps, thereby simplifying the purification process and reducing environmental impact.
Q: Can this method be applied to synthesize 5-selenoquinoline esters as well as amides?
A: Yes, the methodology is versatile. While 8-aminoquinoline amides react efficiently in DMSO, the protocol can be adapted for 8-hydroxyquinoline esters by switching the solvent to acetonitrile (MeCN). This flexibility allows for the synthesis of a broad library of selenium-containing quinoline derivatives for medicinal chemistry applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5-Selenoquinoline Amide Supplier
The technological advancements described in patent CN112979545B represent a significant opportunity for the pharmaceutical industry to access high-quality selenium-containing building blocks more efficiently. At NINGBO INNO PHARMCHEM, we pride ourselves on our ability to translate such innovative academic and patent methodologies into robust commercial processes. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project can move seamlessly from discovery to market. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of 5-selenoquinoline amide meets the highest standards required for clinical and commercial applications.
We invite you to collaborate with us to leverage this metal-free synthesis for your next drug development program. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements. Please contact us today to request specific COA data and route feasibility assessments for your target molecules. Let us help you optimize your supply chain with reliable, cost-effective, and high-purity chemical solutions.
