Advanced Rhodium-Catalyzed Synthesis of 1-Trifluoroethoxy Indan-Succinimide Compounds for Commercial Pharmaceutical Applications
Advanced Rhodium-Catalyzed Synthesis of 1-Trifluoroethoxy Indan-Succinimide Compounds for Commercial Pharmaceutical Applications
The pharmaceutical industry continuously seeks robust and efficient synthetic routes for constructing complex heterocyclic scaffolds that serve as critical building blocks for bioactive molecules. Patent CN111217737B introduces a groundbreaking methodology for the synthesis of 1-trifluoroethoxy indan-succinimide compounds, utilizing a sophisticated yet operationally simple Rhodium(III)-catalyzed tandem reaction. This technology addresses the longstanding challenges associated with constructing indane-containing tricyclic skeletons, which are prevalent in numerous natural products and synthetic drugs exhibiting potent pharmacological activities, such as antihypertensive agents. By leveraging the unique reactivity of dichloropentamethylcyclopentadienyl rhodium dimer, this process enables the direct functionalization of aromatic aldehydes through ortho carbon-hydrogen bond activation, followed by a cascade of conjugate additions and cyclizations. For R&D directors and procurement specialists, this patent represents a significant leap forward in process chemistry, offering a pathway to high-purity intermediates with reduced operational complexity and enhanced scalability.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of indane-containing tricyclic skeleton compounds has been fraught with synthetic difficulties that hinder large-scale production and cost-effectiveness. Traditional literature methods often rely on intramolecular cyclization reactions or intermolecular tandem cyclizations that suffer from significant drawbacks. For instance, earlier approaches developed by researchers such as Fang-Lin Zhang utilized strong bases like 1,8-diazabicyclo[5.4.0]undec-7-ene to catalyze intramolecular Aldol condensations, which frequently require strict anhydrous conditions and exhibit limited tolerance for sensitive functional groups. Other reported strategies involving Ruthenium(II) or Rhodium(III) catalysts for [3+2] annulations often necessitate the pre-functionalization of substrates, such as the dehydration of hydroxyl-substituted isoindolinones to generate reactive N-acyl ketimines in situ. These multi-step sequences not only increase the overall production time but also introduce additional purification burdens and reduce the overall atom economy. Furthermore, many existing protocols demonstrate narrow substrate applicability, failing to accommodate diverse substitution patterns on the aromatic ring or the nitrogen atom of the succinimide moiety, thereby restricting their utility in the rapid generation of compound libraries for drug discovery.
The Novel Approach
In stark contrast to these cumbersome traditional routes, the methodology disclosed in CN111217737B offers a streamlined, one-pot solution that dramatically simplifies the synthetic landscape. This novel approach employs a tandem reaction sequence initiated by the conjugate addition of the ortho carbon-hydrogen bond of an aromatic aldehyde to the double bond of a maleimide. This is seamlessly followed by an intramolecular Aldol condensation and a subsequent Michael addition with trifluoroethanol, all occurring within a single reaction vessel. The use of trifluoroethanol serves a dual purpose as both the solvent and a reactant, facilitating the introduction of the trifluoroethoxy group which can enhance the metabolic stability and lipophilicity of the final drug candidate. The reaction proceeds efficiently under an air atmosphere at moderate temperatures ranging from 90°C to 120°C, eliminating the need for expensive inert gas setups or cryogenic conditions. This operational simplicity translates directly into lower capital expenditure and reduced energy consumption for manufacturing facilities.
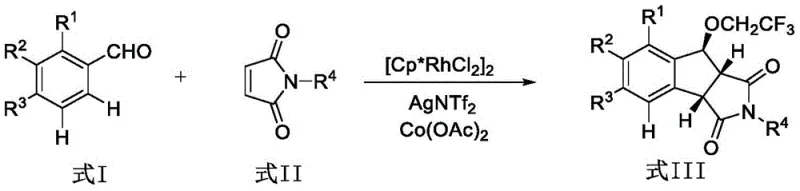
The versatility of this new protocol is exemplified by its broad substrate scope, accommodating a wide array of aromatic aldehydes bearing various substituents such as methyl, ethyl, isopropyl, tert-butyl, fluoro, and bromo groups at different positions on the benzene ring. Additionally, the method tolerates diverse maleimide derivatives, including those substituted with ethyl, benzyl, tert-butyl, phenyl, and cyclohexyl groups on the nitrogen atom. This flexibility allows medicinal chemists to rapidly explore structure-activity relationships without being constrained by synthetic feasibility. The ability to construct these complex tricyclic frameworks in a single step from cheap and easily obtained raw materials represents a paradigm shift in the synthesis of indane-based pharmaceutical intermediates, providing a reliable foundation for the development of next-generation therapeutic agents.
Mechanistic Insights into Rhodium(III)-Catalyzed C-H Activation and Tandem Cyclization
At the heart of this transformative synthesis lies a sophisticated catalytic cycle driven by the Cp*Rh(III) species generated in situ from the dimeric precursor [Cp*RhCl2]2. The mechanism initiates with the coordination of the rhodium catalyst to the carbonyl oxygen of the aromatic aldehyde, which directs the activation of the ortho carbon-hydrogen bond through a concerted metalation-deprotonation (CMD) pathway. This step is facilitated by the acetate additive, which acts as a base to abstract the proton while stabilizing the resulting organometallic intermediate. The resulting five-membered rhodacycle is highly reactive and undergoes migratory insertion into the electron-deficient double bond of the maleimide substrate. This conjugate addition step forms a new carbon-carbon bond and generates an alkyl-rhodium species that is poised for the subsequent cyclization events. The presence of silver salts, specifically silver bistrifluoromethanesulfonimide, plays a crucial role in halide abstraction, generating the cationic rhodium species necessary for high catalytic turnover and preventing catalyst deactivation through chloride binding.
Following the initial C-H activation and insertion, the reaction proceeds through an intricate cascade involving intramolecular Aldol condensation. The aldehyde functionality, now positioned in proximity to the newly formed enolate-like intermediate, undergoes nucleophilic attack to close the five-membered indane ring. This step establishes the core tricyclic skeleton characteristic of the target molecules. The final stage of the tandem sequence involves the Michael addition of trifluoroethanol to the remaining unsaturated bond or an intermediate electrophilic center, installing the trifluoroethoxy group with high regioselectivity. The cobalt acetate co-catalyst is believed to assist in the re-oxidation of the rhodium center or facilitate the proton transfer steps required to regenerate the active catalyst species. This mechanistic elegance ensures that the reaction proceeds with high atom economy, minimizing the formation of by-products and simplifying the downstream purification process. Understanding these mechanistic nuances is vital for process optimization, allowing manufacturers to fine-tune reaction parameters such as temperature, catalyst loading, and stoichiometry to maximize yield and purity for commercial production.
How to Synthesize 1-Trifluoroethoxy Indan-Succinimide Efficiently
The practical implementation of this synthesis route is designed for ease of execution in both laboratory and pilot plant settings. The procedure typically involves charging a pressure-resistant reaction vessel with the aromatic aldehyde, the selected maleimide derivative, and the requisite catalytic system comprising the rhodium dimer, silver salt, and cobalt acetate. Trifluoroethanol is added as the reaction medium, and the mixture is heated to the optimal temperature range of 100°C to 110°C. The reaction is allowed to stir for a period of 16 to 24 hours under ambient air pressure, removing the logistical burden of maintaining an inert atmosphere. Upon completion, the workup is straightforward: the reaction mixture is cooled to room temperature and passed through a silica gel column to adsorb the metal catalysts and insoluble salts. The crude product is then subjected to standard purification techniques such as flash column chromatography or recrystallization to afford the pure 1-trifluoroethoxy indan-succinimide compound. Detailed standardized synthesis steps are provided in the guide below.
- Combine aromatic aldehyde, maleimide compound, silver bistrifluoromethanesulfonimide, cobalt acetate, and the rhodium catalyst in trifluoroethanol solvent.
- Stir the reaction mixture under an air atmosphere at temperatures between 90°C and 120°C for 16 to 24 hours to facilitate the tandem cyclization.
- Upon completion, cool to room temperature, filter through silica gel to remove catalyst and salts, and purify the product via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this Rhodium-catalyzed methodology offers compelling economic and logistical benefits that extend beyond mere chemical efficiency. The primary driver for cost reduction lies in the simplification of the manufacturing process. By consolidating what was traditionally a multi-step sequence into a single pot operation, manufacturers can significantly reduce labor costs, solvent consumption, and waste generation. The elimination of intermediate isolation and purification steps not only shortens the production cycle but also minimizes material loss, leading to improved overall yields. Furthermore, the catalyst loading is relatively low, typically ranging from 1.25% to 7.5% molar equivalent, which helps mitigate the cost impact of using precious metal catalysts. The use of commercially available and inexpensive starting materials, such as substituted benzaldehydes and maleimides, ensures a stable and cost-effective supply chain, reducing vulnerability to raw material price fluctuations.
- Cost Reduction in Manufacturing: The streamlined one-pot nature of this reaction eliminates the need for multiple reactor setups and extensive intermediate handling, which are major cost drivers in fine chemical manufacturing. By avoiding the use of hazardous reagents or extreme conditions that require specialized equipment, capital investment is minimized. The ability to run the reaction under air atmosphere further reduces operational costs associated with nitrogen or argon consumption and the maintenance of leak-free systems. Additionally, the high selectivity of the tandem reaction reduces the formation of difficult-to-remove impurities, lowering the cost of goods sold (COGS) associated with downstream purification and quality control testing.
- Enhanced Supply Chain Reliability: The robustness of this synthetic route contributes significantly to supply chain resilience. The starting materials are commodity chemicals with established global supply networks, ensuring consistent availability and reducing the risk of production delays due to raw material shortages. The tolerance of the reaction to various functional groups means that a single platform technology can be used to produce a wide library of analogues, allowing for flexible inventory management and rapid response to changing market demands. The simplified workup procedure also accelerates the turnaround time from batch completion to finished product release, enabling faster fulfillment of customer orders and improving overall service levels.
- Scalability and Environmental Compliance: Scaling this process from gram to kilogram or ton scale is facilitated by the mild reaction conditions and the absence of sensitive reagents. The use of trifluoroethanol, while requiring appropriate handling, is manageable within standard pharmaceutical manufacturing infrastructure. The high atom economy of the tandem reaction aligns with green chemistry principles by maximizing the incorporation of starting materials into the final product and minimizing waste. This environmental compatibility simplifies regulatory compliance and waste disposal procedures, reducing the environmental footprint of the manufacturing operation. The potential for catalyst recovery and recycling further enhances the sustainability profile of the process, making it an attractive option for companies committed to responsible manufacturing practices.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and technical specifications outlined in the patent documentation, providing clarity on reaction scope, optimization, and practical application. Understanding these details is essential for technical teams evaluating the feasibility of integrating this route into their existing production workflows.
Q: What are the key advantages of the Rhodium(III) catalytic system described in CN111217737B?
A: The primary advantages include the use of readily available raw materials, a simple one-pot operation under air atmosphere, and high reaction efficiency without the need for inert gas protection, which significantly simplifies industrial scale-up.
Q: What is the substrate scope for this synthesis method?
A: The method demonstrates broad substrate tolerance, accommodating various aromatic aldehydes with electron-donating or withdrawing groups (such as alkyl, halogen) and diverse maleimides including N-alkyl, N-benzyl, and N-aryl substituted variants.
Q: How does this method improve upon conventional indane synthesis routes?
A: Unlike traditional methods that often require harsh conditions, multiple steps, or narrow substrate applicability, this tandem reaction constructs the tricyclic skeleton efficiently in a single step with high atom economy and operational simplicity.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1-Trifluoroethoxy Indan-Succinimide Supplier
As the demand for complex pharmaceutical intermediates continues to grow, partnering with an experienced CDMO is crucial for translating innovative synthetic methodologies into commercial reality. NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from the laboratory bench to full-scale manufacturing. Our state-of-the-art facilities are equipped to handle the specific requirements of Rhodium-catalyzed reactions, including precise temperature control and advanced filtration systems for catalyst removal. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of 1-trifluoroethoxy indan-succinimide meets the highest quality standards required for drug substance manufacturing. Our commitment to technical excellence ensures that the theoretical benefits of this patent are fully realized in the final product delivered to your facility.
We invite you to engage with our technical procurement team to discuss how this advanced synthesis route can optimize your supply chain and reduce costs. By requesting a Customized Cost-Saving Analysis, you can gain detailed insights into the economic advantages of switching to this methodology for your specific project needs. We encourage potential partners to contact us to obtain specific COA data and route feasibility assessments tailored to your target molecules. Let us leverage our expertise in C-H activation chemistry to support your drug development goals and ensure a reliable supply of high-quality intermediates for your critical pipelines.
