Revolutionizing 1,2-Diarylethane Synthesis with Air-Stable Nickel Catalysis for Commercial Scale-Up
Revolutionizing 1,2-Diarylethane Synthesis with Air-Stable Nickel Catalysis for Commercial Scale-Up
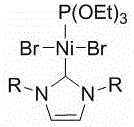
The landscape of organic synthesis for pharmaceutical intermediates is constantly evolving, driven by the urgent need for safer, more cost-effective, and scalable methodologies. A significant breakthrough in this domain is documented in Chinese Patent CN108002966B, which discloses a novel method for synthesizing 1,2-diarylethane compounds. This technology addresses critical bottlenecks in the production of these vital building blocks, which are ubiquitous in drug molecules and biologically active agents. The core innovation lies in the deployment of a specifically designed, air-stable divalent nickel(II) complex, formulated as Ni[P(OEt)3][(RNCHCHNR)C]Br2, where R represents a bulky 2,6-bis(diphenylmethyl)-4-methoxyphenyl group. By leveraging this robust catalyst in the presence of metallic magnesium, the process achieves efficient cross-coupling between benzoheteroarenes and styrene derivatives under mild conditions. This approach not only circumvents the inherent dangers associated with traditional zero-valent nickel catalysts but also eliminates the requirement for hazardous organoaluminum reagents, marking a substantial leap forward in process safety and operational simplicity for industrial applications.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 1,2-diarylethanes via transition metal-catalyzed cross-coupling has been plagued by significant operational hazards and economic inefficiencies. Traditional protocols often rely on noble metal catalysts like iridium, which are prohibitively expensive for large-scale manufacturing, or they utilize zero-valent nickel complexes such as Ni(COD)2. The latter, while effective, is notoriously sensitive to oxygen and moisture, necessitating rigorous exclusion of air and specialized handling equipment that drives up capital expenditure. Furthermore, these legacy methods frequently require the use of trimethylaluminum as a co-catalyst or activator. Trimethylaluminum is a pyrophoric liquid that ignites spontaneously upon contact with air and reacts explosively with water, posing severe safety risks to personnel and facilities. The combination of sensitive catalysts and dangerous reagents creates a high-barrier entry for commercial scale-up, limiting the availability of these key intermediates and inflating supply chain costs due to the need for specialized containment and waste disposal procedures.
The Novel Approach
The methodology presented in the patent data offers a transformative solution by replacing these hazardous components with a stable, single-component nickel(II) system. The use of the air-stable Ni(II) complex allows for handling in ambient conditions prior to the reaction, significantly reducing the complexity of raw material storage and dosing. Instead of pyrophoric aluminum reagents, the process employs metallic magnesium, a benign and inexpensive reducing agent that is widely available and easy to manage. This substitution fundamentally alters the risk profile of the synthesis, making it amenable to standard reactor setups found in most fine chemical manufacturing plants. Moreover, the catalyst exhibits excellent universality, accommodating a wide range of substrates including benzothiazole, benzoxazole, and N-methylbenzimidazole, thereby providing a versatile platform for generating diverse chemical libraries without the need for extensive method redevelopment for each new analog.

Mechanistic Insights into Ni(II)-Catalyzed Cross-Coupling
Understanding the mechanistic underpinnings of this transformation is crucial for R&D directors aiming to optimize process parameters and ensure reproducibility. The catalytic cycle initiates with the in-situ reduction of the precatalyst. Upon introduction of metallic magnesium into the reaction mixture containing the divalent nickel(II) complex, a single-electron transfer or two-electron reduction occurs, generating the active zero-valent nickel(0) species directly within the reaction medium. This active species is coordinatively unsaturated and highly reactive towards oxidative addition. It inserts into the carbon-heteroatom bond of the benzoheteroarene substrate, forming a nickel-heteroaryl intermediate. Subsequently, the styrene derivative coordinates to the metal center and undergoes migratory insertion, extending the carbon chain. The cycle concludes with a reductive elimination step that releases the desired 1,2-diarylethane product and regenerates the nickel(0) catalyst, allowing the turnover to continue. This mechanism ensures high atom economy and minimizes the formation of homocoupling byproducts, which are common impurities in less selective systems.
Impurity control is another critical aspect where this mechanism excels. The steric bulk of the ligand system, particularly the large R groups on the N-heterocyclic carbene moiety, creates a defined coordination sphere around the nickel center. This steric environment kinetically favors the cross-coupling pathway over competing side reactions such as beta-hydride elimination or unproductive oligomerization of the styrene. By maintaining a precise molar ratio of catalyst to magnesium and substrates, typically around 0.1:1:1, the reaction kinetics can be tuned to maximize yield while suppressing the formation of difficult-to-remove impurities. The use of tetrahydrofuran as a solvent further stabilizes the organometallic intermediates through coordination, ensuring a smooth progression of the catalytic cycle and facilitating the isolation of high-purity products after simple aqueous workup and chromatographic purification.
How to Synthesize 1,2-Diarylethane Efficiently
The practical implementation of this synthesis route is straightforward and designed for robustness in a production environment. The protocol involves charging a reactor with the specific Ni(II) catalyst, magnesium turnings or chips, the benzoheteroarene starting material, and the styrene derivative under an inert atmosphere. Tetrahydrofuran is added as the reaction medium, and the mixture is heated to temperatures between 60°C and 90°C. The reaction proceeds over a period of 36 to 72 hours, during which the magnesium activates the catalyst and drives the coupling to completion. Detailed standardized operating procedures regarding exact dosing sequences, agitation rates, and quenching protocols are essential for consistent results. For a comprehensive breakdown of the experimental steps and optimization parameters, please refer to the technical guide below.
- Prepare the reaction vessel under an inert argon atmosphere and sequentially add the air-stable Ni(II) catalyst, magnesium chips, benzoheteroarene substrate, styrene derivative, and anhydrous tetrahydrofuran solvent.
- Heat the reaction mixture to a temperature range of 60-90°C and maintain stirring for a duration of 36 to 72 hours to allow for the in-situ generation of the active zero-valent nickel species and subsequent cross-coupling.
- Quench the reaction with water upon completion, extract the organic product using ethyl acetate, and purify the crude mixture via column chromatography to isolate the high-purity 1,2-diarylethane target molecule.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this nickel-catalyzed methodology represents a strategic opportunity to de-risk the supply of critical pharmaceutical intermediates. The elimination of pyrophoric reagents like trimethylaluminum removes a major logistical hurdle, as it negates the need for specialized transport and storage infrastructure dedicated to hazardous materials. This simplification translates directly into reduced overhead costs and lower insurance premiums for manufacturing sites. Furthermore, the reliance on base metals like nickel and magnesium, rather than precious metals like iridium or palladium, insulates the production cost from the volatile fluctuations of the precious metals market. The air stability of the catalyst precursor also extends its shelf life, reducing waste associated with catalyst degradation and allowing for more flexible inventory management strategies.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the substitution of expensive and hazardous reagents with commodity chemicals. By avoiding the use of noble metal catalysts and dangerous organoaluminum compounds, the raw material cost profile is significantly improved. Additionally, the simplified safety requirements reduce the capital intensity of the manufacturing process, as standard stainless steel reactors can be utilized without the need for exotic linings or complex gas-handling systems required for pyrophoric materials. The high yields reported in the patent examples, often exceeding 85%, further contribute to cost efficiency by maximizing the output per batch and minimizing the loss of valuable starting materials.
- Enhanced Supply Chain Reliability: Supply continuity is bolstered by the use of widely available and stable reagents. Magnesium metal and tetrahydrofuran are commodity chemicals with robust global supply chains, ensuring that production is not held hostage by the scarcity of niche reagents. The air-stable nature of the nickel catalyst means it can be sourced from multiple suppliers and stored for extended periods without degradation, providing a buffer against supply disruptions. This reliability is crucial for maintaining consistent production schedules for downstream API manufacturing, preventing costly delays that can ripple through the entire pharmaceutical value chain.
- Scalability and Environmental Compliance: From an environmental and scalability perspective, this method offers distinct advantages. The absence of toxic aluminum waste streams simplifies effluent treatment and reduces the environmental footprint of the process. The reaction conditions are mild, operating at moderate temperatures that are energy-efficient and easily manageable on a multi-ton scale. The robustness of the catalyst system allows for direct scale-up from laboratory to pilot and commercial production without the extensive re-optimization often required for sensitive catalytic systems. This ease of scale-up accelerates time-to-market for new drug candidates relying on this scaffold.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this nickel-catalyzed cross-coupling technology. These insights are derived directly from the patent specifications and are intended to clarify the operational benefits and scope of the method for potential partners and licensees. Understanding these details is key to evaluating the feasibility of integrating this process into existing manufacturing portfolios.
Q: What represents the primary safety advantage of this Ni(II) catalytic method over traditional protocols?
A: Unlike conventional methods requiring pyrophoric trimethylaluminum or highly oxygen-sensitive Ni(0) complexes like Ni(COD)2, this patent utilizes an air-stable Ni(II) precursor and benign magnesium metal, drastically reducing fire hazards and simplifying operational safety protocols.
Q: Which specific heterocyclic substrates are compatible with this cross-coupling methodology?
A: The catalytic system demonstrates broad substrate tolerance, effectively coupling styrenes with various benzoheteroarenes including benzothiazole, benzoxazole, and N-methylbenzimidazole derivatives to form diverse 1,2-diarylethane scaffolds.
Q: How does the use of magnesium metal facilitate the catalytic cycle in this transformation?
A: Magnesium acts as a mild reducing agent that converts the stable divalent nickel(II) precatalyst into the active zero-valent nickel(0) species in situ, thereby initiating the oxidative addition step required for the cross-coupling reaction without needing external hazardous reductants.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1,2-Diarylethane Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the air-stable nickel catalysis described in CN108002966B for the production of high-value pharmaceutical intermediates. As a leading CDMO partner, we possess the technical expertise and infrastructure to rapidly adopt and optimize this advanced synthetic route. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from bench-scale discovery to industrial manufacturing is seamless. We are committed to delivering products that meet stringent purity specifications, utilizing our rigorous QC labs to verify that every batch of 1,2-diarylethane intermediate conforms to the highest quality standards required by global regulatory bodies.
We invite pharmaceutical companies and fine chemical manufacturers to collaborate with us to leverage this cost-effective and safe technology. By partnering with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality needs. We encourage you to contact our technical procurement team today to request specific COA data for our catalog items or to discuss route feasibility assessments for your proprietary molecules. Let us help you secure a sustainable and economical supply chain for your critical 1,2-diarylethane building blocks.
