Advanced Rare-Earth Catalysis for Efficient Six-Membered Carbocyclic Ring Derivative Production
Advanced Rare-Earth Catalysis for Efficient Six-Membered Carbocyclic Ring Derivative Production
The landscape of organic synthesis for complex cyclic structures is undergoing a significant transformation, driven by the urgent need for more sustainable and efficient catalytic systems. Patent CN109369514B introduces a groundbreaking methodology for the synthesis of six-membered carbocyclic ring derivatives, utilizing a sophisticated organic rare-earth catalyst system coupled with a boron salt cocatalyst. This innovation addresses critical bottlenecks in the production of high-value pharmaceutical intermediates and fine chemicals, offering a robust alternative to traditional transition metal catalysis. By leveraging the unique electronic properties of rare-earth elements such as scandium, yttrium, or lutetium, this process achieves exceptional regioselectivity and stereoselectivity under remarkably mild conditions. For R&D directors and process chemists, this represents a pivotal shift towards greener chemistry that does not compromise on yield or purity, ensuring a reliable supply chain for complex molecular scaffolds essential in modern drug discovery.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of unsaturated double-bond substituted carbocyclic compounds has relied heavily on metal-catalyzed cyclization isomerization of dienes or eneynes. Pioneering work by researchers such as RajanBabu and Carboni utilized zero-valent palladium or other transition metals to facilitate these transformations. However, these conventional pathways are fraught with significant technical challenges that hinder their industrial applicability. The oxidative cyclization of dienes often leads to an increase in kinetic and thermodynamic potential energy, resulting in reduced stability of the unsaturated products. Consequently, these reactions are prone to generating substantial amounts of complex polycyclic byproducts through tandem [4+2] cyclization events. Furthermore, the reaction conditions required for these traditional methods are often harsh, necessitating high temperatures or aggressive reagents that can degrade sensitive functional groups, thereby complicating downstream purification and reducing overall process efficiency.
The Novel Approach
In stark contrast, the methodology disclosed in patent CN109369514B employs a hydrocarbon activation strategy mediated by an organic rare-earth catalyst and a boron salt cocatalyst. This novel approach fundamentally alters the reaction landscape by enabling the direct coupling of substituted aromatic compounds, conjugated dienes, and substituted ethylenes to form stable six-membered rings. The use of rare-earth metals allows for precise control over the coordination environment, minimizing unwanted side reactions and ensuring high atom economy. The reaction proceeds smoothly in common organic solvents like chlorobenzene at temperatures ranging from 25°C to 100°C, significantly lowering the energy footprint compared to legacy methods. This versatility allows for the synthesis of a wide array of derivatives simply by varying the substituents on the starting materials, making it an invaluable tool for the rapid generation of chemical libraries.
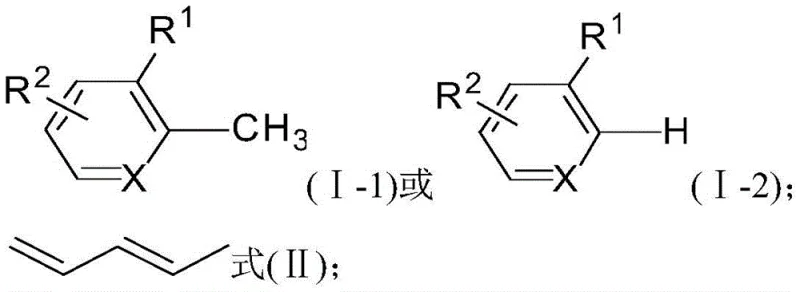
The general reaction scope is illustrated above, demonstrating the flexibility of the system to accommodate both carbon and nitrogen-containing heterocycles (where X is C or N). This adaptability is crucial for cost reduction in pharmaceutical intermediate manufacturing, as it allows a single catalytic platform to produce diverse structural motifs required for different drug candidates without the need for extensive re-optimization of reaction conditions.
Mechanistic Insights into Rare-Earth Catalyzed Hydrocarbon Activation
The core of this technological breakthrough lies in the intricate interplay between the organic rare-earth catalyst and the boron salt cocatalyst. The catalyst, typically structured as (η5-C5Me4-C5H3N-R)Ln(CH2SiMe3)2, features a tetramethylcyclopentadienyl ligand with a pyridine side chain coordinated to a central rare-earth ion (Ln). Upon activation by the boron salt cocatalyst, such as [Ph3C][B(C6F5)4], the catalyst undergoes a transformation that generates a highly active cationic species. This active species initially coordinates with the substituted aromatic compound (Formula I-1 or I-2) through a hydrocarbon activation mechanism. The steric hindrance and electronic effects imparted by the specific ligands on the rare-earth center dictate the orientation of this coordination, which is the primary determinant of the stereochemical outcome of the final product.
Following the initial coordination, the conjugated diene (Formula II) inserts into the metal-carbon bond with high stereoselectivity, forming a key intermediate. Subsequently, the substituted ethylene (Formula III) inserts into the coordination bond of this intermediate, closing the six-membered ring structure. This sequential insertion mechanism ensures that the double bonds are positioned precisely, avoiding the formation of unstable isomers common in palladium-catalyzed routes. The robustness of the rare-earth catalyst against moisture and oxygen, while still requiring inert gas protection for optimal performance, adds a layer of operational safety and reliability. This mechanistic clarity provides process engineers with the confidence to scale the reaction, knowing that the impurity profile is tightly controlled by the inherent selectivity of the catalyst system rather than relying on difficult separations post-reaction.
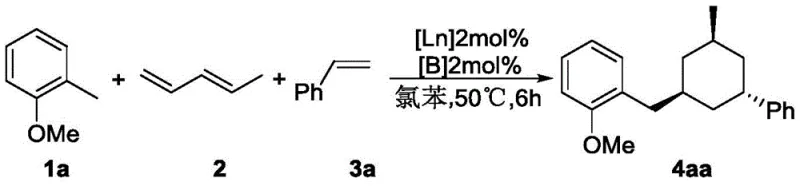
A concrete example of this mechanism in action is shown in the synthesis of compound 4aa, where 2-methylanisole reacts with 1,4-pentadiene and styrene to yield the target six-membered ring derivative with a 90% isolated yield. This high efficiency underscores the potential of this technology for commercial scale-up of complex pharmaceutical intermediates.
How to Synthesize Six-Membered Carbocyclic Derivatives Efficiently
Implementing this synthesis route requires careful attention to the preparation of the catalyst system and the maintenance of an inert atmosphere. The process begins with the activation of the rare-earth precursor by the boron salt in an aromatic solvent, followed by the sequential addition of substrates. The reaction temperature and time can be tuned based on the specific reactivity of the substrates, with typical conditions ranging from 50°C to 70°C over 4 to 10 hours. Detailed standard operating procedures regarding stoichiometry, solvent drying, and workup protocols are essential for reproducibility.
- Prepare the catalyst system by dissolving the boron salt cocatalyst in chlorobenzene and adding it dropwise to the stirred organic rare-earth catalyst solution under nitrogen protection.
- Sequentially add the substituted aromatic compound (Formula I), conjugated diene (Formula II), and substituted ethylene (Formula III) to the reaction mixture.
- Stir the reaction at mild temperatures (25-100°C) for 2-24 hours, monitor progress via TLC, and purify the final product using column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this rare-earth catalytic technology offers substantial strategic benefits beyond mere technical feasibility. The shift away from precious metals like palladium to more abundant rare-earth elements significantly mitigates the risk associated with volatile metal prices and supply constraints. Furthermore, the mild reaction conditions translate directly into lower energy consumption and reduced wear on reactor equipment, contributing to a more sustainable and cost-effective manufacturing process. The simplicity of the workup, which primarily involves solvent removal and standard chromatography, eliminates the need for complex quenching steps or specialized scavengers often required to remove toxic heavy metal residues, thereby streamlining the production timeline.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts and the reduction in energy requirements due to mild reaction temperatures lead to significant operational cost savings. Additionally, the high atom economy and minimal formation of byproducts mean that raw material utilization is maximized, reducing waste disposal costs and improving the overall yield per batch. This efficiency is critical for maintaining competitive pricing in the global market for fine chemical intermediates.
- Enhanced Supply Chain Reliability: The raw materials required for this synthesis, including substituted anisoles, pyridines, and common dienes, are commercially available in bulk quantities from multiple suppliers. This diversity in sourcing options reduces dependency on single-source vendors and enhances the resilience of the supply chain against disruptions. The robustness of the catalyst system also ensures consistent batch-to-batch quality, which is vital for maintaining long-term contracts with pharmaceutical clients.
- Scalability and Environmental Compliance: The process aligns well with green chemistry principles by avoiding toxic heavy metals and minimizing waste generation. The use of standard organic solvents and the absence of hazardous byproducts simplify regulatory compliance and environmental permitting for new production facilities. This ease of scalability ensures that production volumes can be rapidly increased from pilot scale to multi-ton annual capacity to meet surging market demand without compromising on safety or quality standards.
Frequently Asked Questions (FAQ)
The following questions address common inquiries regarding the technical specifications and commercial viability of this synthesis method. These insights are derived directly from the experimental data and technical disclosures within the patent documentation, providing a clear understanding of the process capabilities.
Q: What are the primary advantages of this rare-earth catalytic method over traditional palladium catalysis?
A: Unlike traditional palladium-catalyzed cyclization which often suffers from product instability and complex polycyclic byproducts, this rare-earth method offers superior reaction stability, milder conditions (25-100°C), and excellent atom economy with minimal side reactions.
Q: Can this synthesis method be scaled for industrial production of pharmaceutical intermediates?
A: Yes, the method utilizes readily available raw materials and standard organic solvents like chlorobenzene. The simple workup procedure involving solvent removal and column chromatography makes it highly suitable for commercial scale-up of complex pharmaceutical intermediates.
Q: What types of substituents are tolerated in this hydrocarbon activation reaction?
A: The process demonstrates broad substrate scope, tolerating various groups including alkyl, alkoxy, amino, halogen, and phenyl substituents on the aromatic rings, allowing for the synthesis of diverse six-membered carbocyclic derivatives.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Six-Membered Carbocyclic Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced catalytic technologies like the one described in patent CN109369514B for the future of pharmaceutical manufacturing. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive high-purity intermediates that meet stringent purity specifications. Our state-of-the-art rigorous QC labs are equipped to handle the complex analysis required for these novel structures, guaranteeing that every batch delivered adheres to the highest international standards for safety and efficacy.
We invite you to collaborate with us to leverage this cutting-edge synthesis method for your next project. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and timeline. Please contact our technical procurement team today to request specific COA data and comprehensive route feasibility assessments, and let us help you accelerate your path to market with confidence and efficiency.
