Advanced Lewis Acid Catalysis for High-Purity Indolizine-3-Carboxamide Derivatives Manufacturing
Introduction to Next-Generation Indolizine Synthesis
The pharmaceutical and materials science sectors are constantly seeking more efficient pathways to access complex N-heterocyclic scaffolds, particularly those with proven biological relevance. Patent CN112979645B introduces a groundbreaking methodology for the preparation of indolizine-3-formamidated derivatives, a class of compounds known for their potential as NF-κB inhibitors and fluorescent probes. Unlike traditional multi-step syntheses that often suffer from poor atom economy and harsh conditions, this invention leverages a direct C-H functionalization strategy. By utilizing indolizine derivatives and 1,4,2-dioxazol-5-one derivatives under Lewis acid catalysis, the process achieves a one-step conversion with high regioselectivity. This technological leap not only simplifies the synthetic route but also aligns perfectly with modern green chemistry mandates by producing only carbon dioxide as a byproduct. For R&D teams focused on rapid library generation and process chemists aiming for streamlined manufacturing, this patent represents a significant optimization in accessing these valuable pharmacophores.

The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the indolizine core functionalized at the 3-position has been a laborious endeavor requiring multiple discrete chemical transformations. Prior art, such as the work by Yong Fu et al. and Eunha Kim et al., typically relies on the cycloaddition of alkyl pyridinium salts with alkenes to first generate an indolizine-3-carboxylate ester intermediate. This ester must then undergo a subsequent aminolysis reaction to yield the desired carboxamide. This two-step sequence inherently suffers from cumulative yield losses, increased solvent consumption, and the generation of stoichiometric salt waste from the initial pyridinium salt formation. Furthermore, the requirement for pre-functionalized starting materials limits the structural diversity that can be rapidly explored during the lead optimization phase. The reliance on ester intermediates also introduces purification challenges, as separating unreacted esters from the final amide products can be difficult without extensive chromatographic efforts, driving up the cost of goods for potential API intermediates.
The Novel Approach
The methodology disclosed in CN112979645B fundamentally disrupts this status quo by enabling a direct, single-step transformation. By reacting readily available indolizine substrates with 1,4,2-dioxazol-5-one derivatives in the presence of a Lewis acid, the synthesis bypasses the need for ester intermediates entirely. This approach utilizes the inherent reactivity of the indolizine C3-H bond, which is activated towards electrophilic attack. The use of dioxazolones as amidating reagents is particularly advantageous because they serve as efficient nitrene precursors that release only CO2 upon reaction, ensuring high atom economy. This direct functionalization strategy drastically reduces the number of unit operations required, minimizes solvent usage, and simplifies the overall workflow. For procurement managers, this translates to a reduction in raw material complexity and a more robust supply chain, as fewer specialized intermediates need to be sourced or manufactured in-house.
Mechanistic Insights into Lewis Acid-Catalyzed C-H Amidation
Understanding the mechanistic underpinnings of this transformation is crucial for R&D directors evaluating its feasibility for complex molecule synthesis. The reaction is initiated by the coordination of the Lewis acid catalyst, such as BF3·OEt2, to the carbonyl oxygen of the 1,4,2-dioxazol-5-one substrate. This coordination significantly enhances the electrophilicity of the carbonyl carbon, facilitating a nucleophilic attack or ring-opening event. As detailed in the patent, this interaction leads to the formation of a reactive acyl nitrenoid or similar active intermediate species. This highly reactive intermediate then engages in a selective C-H insertion at the 3-position of the indolizine ring. The electronic properties of the indolizine nucleus, specifically the electron-rich nature of the five-membered ring fused to the pyridine, direct this functionalization with exceptional regioselectivity, avoiding substitution at other potential sites on the aromatic system.
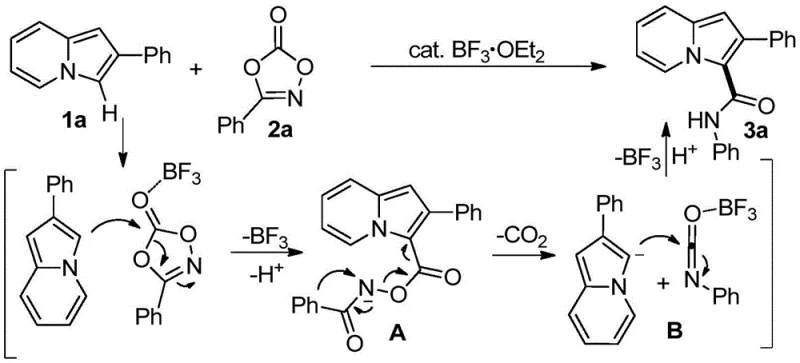
Furthermore, the mechanism ensures excellent impurity control, a critical factor for pharmaceutical applications. Because the reaction proceeds through a well-defined catalytic cycle involving the activation of the dioxazolone, side reactions such as over-alkylation or polymerization are minimized. The release of carbon dioxide gas acts as a thermodynamic driving force, pushing the equilibrium towards the product and preventing reversible side reactions. The choice of Lewis acid is also pivotal; while BF3·OEt2 is highlighted, the patent notes that protic acids can also facilitate the transformation, offering flexibility in catalyst selection based on substrate sensitivity. This mechanistic robustness means that the process can tolerate a wide range of substituents on both the indolizine and the dioxazolone components, allowing for the synthesis of diverse derivatives without compromising purity profiles.
How to Synthesize Indolizine-3-Carboxamide Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires adherence to specific operational parameters to maximize yield and safety. The protocol involves combining the indolizine substrate and the dioxazolone coupling partner in a suitable solvent such as 1,2-dichloroethane (DCE). The reaction mixture is then treated with a catalytic amount of BF3·OEt2 and heated to temperatures ranging from 90°C to 130°C, with 110°C being optimal for most substrates. Detailed standard operating procedures regarding stoichiometry, addition rates, and workup protocols are essential for reproducibility.
- Mix indolizine derivative (1 eq) and 1,4,2-dioxazol-5-one derivative (0.5-1.5 eq) in a reaction vessel with a stir bar.
- Add Lewis acid catalyst BF3·OEt2 (0.1-1.5 eq) and solvent DCE to achieve a concentration of 0.05-0.5 mol/L.
- Heat the mixture to 110°C and stir for 12 hours, then purify via silica gel chromatography to isolate the target product.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this Lewis acid-catalyzed route offers substantial strategic benefits for supply chain stability and cost management. The elimination of transition metal catalysts, which are often expensive and subject to strict regulatory limits in final drug substances, removes the need for costly metal scavenging steps. This simplification of the downstream processing directly contributes to cost reduction in API manufacturing by reducing the number of purification columns and the volume of solvents required for metal removal. Additionally, the reagents used, specifically the indolizine cores and dioxazolones, are becoming increasingly accessible through established chemical supply chains, ensuring reliable sourcing for large-scale campaigns.
- Cost Reduction in Manufacturing: The one-step nature of this reaction significantly lowers the operational expenditure associated with production. By consolidating what was previously a multi-step sequence into a single pot, manufacturers save on labor, energy, and equipment time. The high atom economy, characterized by the release of only CO2 as a byproduct, means that a larger proportion of the input mass is converted into valuable product rather than waste. This efficiency reduces the burden on waste treatment facilities and lowers the overall environmental compliance costs associated with hazardous waste disposal, providing a clear economic advantage over traditional ester-based routes.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions enhances supply chain resilience. The process does not rely on air-sensitive organometallic reagents or cryogenic conditions, which can be logistical bottlenecks in global manufacturing networks. Instead, it utilizes stable Lewis acids and common organic solvents that are readily available in bulk quantities worldwide. This accessibility reduces the risk of production delays caused by raw material shortages. Furthermore, the broad substrate scope implies that the same manufacturing infrastructure can be used to produce a wide variety of analogues, allowing for flexible capacity allocation depending on market demand for specific derivatives.
- Scalability and Environmental Compliance: Scalability is a key strength of this methodology, making it ideal for the commercial scale-up of complex heterocycles. The reaction generates minimal waste, adhering to the principles of green chemistry which are increasingly mandated by regulatory bodies and corporate sustainability goals. The absence of heavy metal residues simplifies the regulatory filing process for new drug applications, as extensive data on metal clearance is not required. This environmental friendliness not only mitigates regulatory risk but also enhances the brand value of the manufacturer as a sustainable partner in the pharmaceutical value chain.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented technology. These insights are derived directly from the experimental data and claims within CN112979645B, providing a factual basis for decision-making.
Q: What are the primary advantages of this new synthesis method over traditional cycloaddition routes?
A: The new method utilizes a direct C-H functionalization strategy catalyzed by Lewis acids, eliminating the need for multi-step sequences involving alkyl pyridinium salts and subsequent aminolysis. This results in higher atom economy, simpler post-processing, and significantly reduced waste generation, aligning with green chemistry principles.
Q: Is the catalyst system compatible with large-scale industrial production?
A: Yes, the process uses commercially available and safe Lewis acids like BF3·OEt2 or protic acids. The reaction conditions (110°C in DCE) are robust and scalable, avoiding expensive transition metal catalysts that require complex removal steps, thereby facilitating easier commercial scale-up.
Q: What is the regioselectivity of the formamidation reaction?
A: The reaction exhibits excellent regioselectivity for the 3-position of the indolizine ring. The mechanism involves the activation of the 1,4,2-dioxazol-5-one by the Lewis acid, leading to a specific nitrene-like intermediate that selectively functionalizes the C3-H bond without affecting other positions on the heterocyclic core.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Indolizine-3-Carboxamide Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the synthetic route described in CN112979645B for the production of high-value pharmaceutical intermediates. Our team of process chemists has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from bench-scale discovery to industrial manufacturing is seamless. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of indolizine derivative meets the exacting standards required for clinical and commercial applications. Our commitment to quality assurance ensures that the impurity profiles of our products are fully characterized and controlled.
We invite potential partners to leverage our technical expertise to optimize this pathway for their specific pipeline candidates. By collaborating with us, you can access a Customized Cost-Saving Analysis tailored to your project's unique volume and purity requirements. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments. Together, we can accelerate the development of next-generation therapeutics while maintaining the highest standards of efficiency and sustainability in chemical manufacturing.
