Advanced Synthesis of Coumarin Laser Dyes for High-Performance Optical Applications
Introduction to Next-Generation Optical Materials
The rapidly evolving landscape of optoelectronics and photonics demands materials with precise spectral properties, particularly in the realm of laser dyes and fluorescence sensors. Patent CN102321091A introduces a groundbreaking methodology for synthesizing a novel class of coumarin-based laser dyes, specifically designated as Coumarin Q, which overcomes the historical limitations of emission wavelengths in this chemical family. Traditionally, coumarin fluorophores have been restricted to the blue-green spectrum, typically emitting between 400nm and 500nm, which constrains their utility in advanced red-shifted applications. This patented technology achieves a significant breakthrough by engineering a molecular structure that pushes fluorescence emission beyond 500nm while maintaining a substantial Stokes shift of up to 110nm. For R&D directors and procurement specialists in the electronic chemical sector, this represents a critical opportunity to access high-purity intermediates that enable next-generation electroluminescent devices and high-sensitivity optical sensors without the prohibitive costs often associated with custom organic synthesis.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of long-wavelength coumarin derivatives has been plagued by complex multi-step routes that rely on expensive, specialized starting materials or harsh reaction conditions that degrade product quality. Conventional approaches often struggle to introduce the necessary electron-donating groups at the 7-position of the coumarin ring without compromising the integrity of the lactone bond or introducing difficult-to-remove impurities. Furthermore, many existing methods fail to achieve the large Stokes displacement required for effective signal-to-noise ratios in fluorescence sensing, leading to self-absorption issues that diminish device efficiency. The reliance on non-scalable catalysts or unstable intermediates in older protocols frequently results in batch-to-batch variability, creating significant supply chain risks for manufacturers who require consistent optical performance. These technical bottlenecks have historically limited the commercial adoption of red-shifted coumarins in mass-market laser and display applications.
The Novel Approach
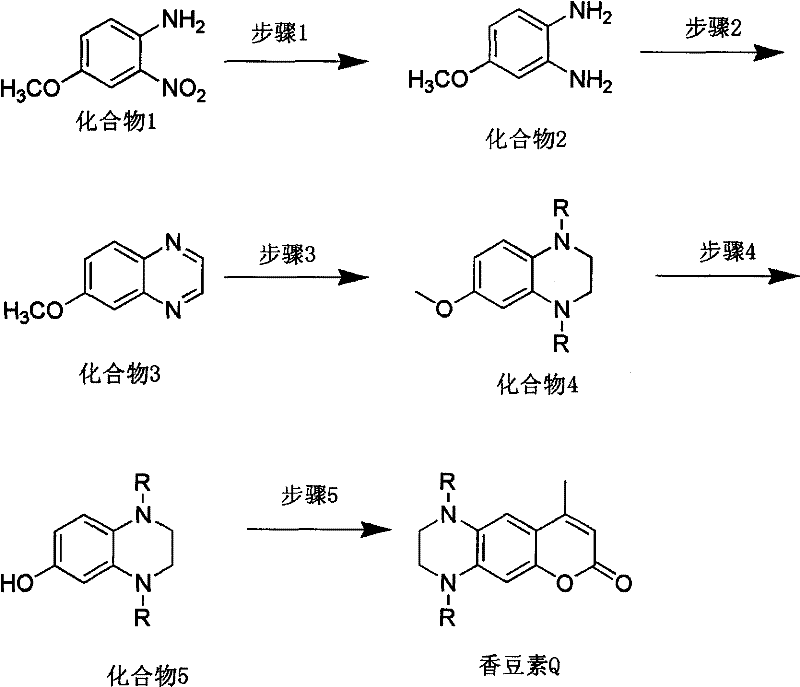
The methodology outlined in CN102321091A presents a streamlined, five-step synthetic pathway that utilizes readily available commodity chemicals to construct the complex heterocyclic framework of Coumarin Q. By starting with a substituted nitro-anisole derivative (Compound 1) and employing a sequence of catalytic hydrogenation, glyoxal condensation, and selective reduction, the process efficiently builds the requisite piperazine-fused scaffold. This route is distinct in its ability to modulate the electronic properties of the final dye through the introduction of variable alkyl groups (R = methyl, ethyl, or propyl) at a specific stage, allowing for fine-tuning of solubility and spectral characteristics. The use of standard reagents such as sodium borohydride and ethyl acetoacetate ensures that the process is not only chemically robust but also economically viable for large-scale production. This strategic design eliminates the need for exotic precursors, thereby drastically simplifying the procurement landscape and enhancing the overall reliability of the manufacturing supply chain.
Mechanistic Insights into Catalytic Reduction and Cyclization
A deep dive into the reaction mechanism reveals the sophistication of the catalytic systems employed to ensure high selectivity and yield. The initial transformation involves the reduction of the nitro group in Compound 1 to an amine, which can be achieved via multiple catalytic systems including Pt/C with hydrogen, Raney nickel with hydrazine hydrate, or iron powder with hydrochloric acid. This flexibility allows manufacturers to choose the most cost-effective reduction method based on their existing infrastructure, with the hydrazine/Raney nickel system offering a particularly attractive balance of activity and safety at moderate temperatures of 60°C. Subsequent condensation with glyoxal forms a quinoxaline ring (Compound 3), a critical structural motif that serves as the precursor for the nitrogen-containing heterocycle. The precision required in this step is paramount, as the stoichiometry of glyoxal (20-40 molar equivalents) drives the equilibrium towards the desired cyclic product while minimizing oligomerization side reactions.
The subsequent reduction of the quinoxaline ring to the piperazine derivative (Compound 4) using sodium borohydride in anhydrous toluene is a masterclass in chemoselectivity. By maintaining low temperatures (5°C) during the addition of the reducing agent and carefully controlling the addition of organic acid, the process prevents the violent decomposition of borohydride while ensuring the complete reduction of the imine bonds without affecting the methoxy ether linkage. This intermediate is then subjected to haloid acid demethylation, a crucial step that unveils the phenolic hydroxyl group necessary for the final Pechmann condensation. The final cyclization with ethyl acetoacetate under acid catalysis (using p-toluenesulfonic acid or concentrated sulfuric acid) closes the coumarin lactone ring. The rigorous control of reaction parameters throughout this sequence ensures that the final product exhibits the desired fluorescence properties with minimal contamination from isomeric byproducts, a key requirement for high-performance optical materials.
How to Synthesize Coumarin Q Efficiently
Implementing this synthesis requires strict adherence to the thermal and stoichiometric parameters defined in the patent to maximize yield and purity. The process is designed to be telescoped where possible, particularly with unstable intermediates like Compound 2 and Compound 4, which are used directly in subsequent steps to prevent oxidation or degradation. Operators must pay close attention to the exothermic nature of the borohydride reduction and the demethylation steps, utilizing appropriate cooling and reflux setups to maintain safety and reaction fidelity. The final purification via column chromatography using dichloromethane and ethyl acetate ensures the removal of any residual starting materials or polymeric tars, yielding a yellow solid with the characteristic long-wavelength emission. For detailed operational protocols, safety data sheets, and specific equipment recommendations tailored to your facility's capabilities, please refer to the standardized synthesis guide below.
- Perform catalytic hydrogenation of Compound 1 (nitro-anisole derivative) using Raney Nickel or Pt/C at 60°C to obtain the diamine intermediate (Compound 2).
- Condense Compound 2 with glyoxal in acetonitrile at 60°C to form the quinoxaline derivative (Compound 3), followed by purification.
- Reduce Compound 3 using sodium borohydride in anhydrous toluene with organic acid to generate the piperazine derivative (Compound 4).
- Execute demethylation of Compound 4 using hydrohalic acid in glacial acetic acid under reflux to yield the phenolic intermediate (Compound 5).
- Complete the synthesis via Pechmann condensation of Compound 5 with ethyl acetoacetate and an acid catalyst at 100°C to form the final Coumarin Q product.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this synthetic route offers profound advantages for procurement managers and supply chain heads looking to optimize the cost structure of electronic chemical manufacturing. The elimination of rare earth metals or precious metal catalysts in favor of base metal systems (like iron or nickel) or reusable heterogeneous catalysts (Pt/C) significantly reduces the raw material cost basis. Furthermore, the reliance on bulk commodity solvents such as methanol, acetonitrile, and toluene means that the process is not vulnerable to the price volatility often seen with specialized fluorinated or chlorinated solvents. The robustness of the chemistry allows for potential scale-up from kilogram to multi-ton quantities without requiring fundamental changes to the reactor configuration, thereby reducing the capital expenditure required for technology transfer. This stability translates directly into more predictable lead times and a lower total cost of ownership for downstream users in the laser and sensor industries.
- Cost Reduction in Manufacturing: The process achieves significant cost optimization by utilizing inexpensive, globally sourced reagents like glyoxal and ethyl acetoacetate, which are produced at massive scales for other industries. By avoiding the use of proprietary ligands or complex organometallic catalysts, the method removes the premium pricing typically associated with fine chemical intermediates. Additionally, the ability to reuse catalysts such as Raney nickel further drives down the variable cost per kilogram of the final product. The high atom economy of the condensation steps minimizes waste generation, which indirectly lowers disposal costs and environmental compliance burdens, contributing to a leaner and more profitable manufacturing operation.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of non-restricted, dual-use chemical precursors that are readily available from multiple global suppliers. Unlike pathways that depend on single-source specialty building blocks, this route allows procurement teams to diversify their vendor base for key inputs like sodium borohydride and hydrohalic acids. The moderate reaction conditions (temperatures ranging from 5°C to 100°C and atmospheric pressure) reduce the risk of unplanned shutdowns due to equipment failure or safety incidents. This operational stability ensures consistent delivery schedules, enabling downstream manufacturers to maintain just-in-time inventory levels without the need for excessive safety stock buffers.
- Scalability and Environmental Compliance: The synthetic pathway is inherently scalable, having been demonstrated effectively in batch reactors with standard agitation and heating capabilities. The waste streams generated are primarily aqueous acidic or organic solvent wastes that can be managed through standard industrial wastewater treatment protocols, avoiding the creation of persistent organic pollutants. The absence of heavy metal residues in the final product simplifies the regulatory approval process for use in consumer electronics and medical sensors. This environmental compatibility aligns with increasingly stringent global regulations on hazardous substances, future-proofing the supply chain against potential legislative changes and ensuring long-term market access for the final dye products.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this coumarin synthesis technology. These answers are derived directly from the experimental data and claims within the patent documentation to provide accurate guidance for technical teams. Understanding these nuances is essential for evaluating the feasibility of integrating this material into your current product lines or R&D pipeline. We encourage stakeholders to review these points carefully to assess the alignment with their specific quality and performance requirements.
Q: What distinguishes this Coumarin Q dye from conventional coumarin derivatives?
A: Unlike traditional coumarin dyes which typically emit in the 400-500nm range, this novel synthesis produces dyes with fluorescence emission wavelengths greater than 500nm and a significantly larger Stokes shift of approximately 110nm, making them superior for laser and sensor applications.
Q: Are the raw materials for this synthesis commercially scalable?
A: Yes, the process utilizes widely available commodity chemicals such as glyoxal, sodium borohydride, ethyl acetoacetate, and standard solvents like methanol and toluene, ensuring robust supply chain continuity and cost-effective manufacturing.
Q: How does the process handle impurity control during the reduction steps?
A: The method employs specific temperature controls (e.g., cooling to 5°C during borohydride addition) and solvent choices (anhydrous toluene) to manage exothermic reactions and minimize side products, ensuring high purity of the intermediate piperazine derivatives before the final cyclization.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Coumarin Laser Dye Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-performance optical materials play in the advancement of laser technology and electroluminescent displays. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from laboratory bench to full-scale manufacturing is seamless and efficient. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, including detailed analysis of fluorescence quantum yield and Stokes shift parameters. We are committed to delivering Coumarin Q intermediates that meet the exacting standards required for high-end electronic applications, providing you with a competitive edge in the global market through superior material consistency and reliability.
We invite you to engage with our technical procurement team to discuss how this patented synthesis route can be adapted to your specific volume and purity needs. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into how optimizing this supply chain can reduce your overall manufacturing expenses. We are prepared to provide specific COA data and comprehensive route feasibility assessments to support your validation processes. Let us collaborate to accelerate the deployment of next-generation optical solutions, leveraging our expertise in fine chemical synthesis to drive innovation and efficiency in your operations.
