Advancing Chiral Intermediate Production: A Safer Route to (R)-2-Benzyloxypropionic Acid
The landscape of chiral pharmaceutical intermediate manufacturing is constantly evolving, driven by the dual imperatives of safety and efficiency. A pivotal advancement in this domain is detailed in patent CN113233972B, which discloses a novel synthesis method for (R)-2-benzyloxypropionic acid and its key intermediates. This technology represents a significant departure from traditional etherification protocols that rely on hazardous reagents like sodium hydride or metallic sodium. By substituting these dangerous bases with sodium tert-amylate, the process not only mitigates the risk of generating explosive hydrogen gas but also streamlines the production workflow for scalable manufacturing. For R&D directors and procurement strategists, this patent offers a compelling blueprint for producing high-value chiral building blocks with enhanced operational safety and consistent quality profiles.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of benzyloxy-substituted propionic acids has relied heavily on aggressive alkylation conditions that pose substantial safety and logistical challenges. Traditional Method I often involves the hydrolysis of esters under harsh alkaline conditions, which can lead to racemization and reduced optical purity. More critically, Method III, which utilizes sodium hydride (NaH) as the base, introduces severe safety liabilities; the reaction between NaH and alcohol substrates inevitably releases hydrogen gas, creating a highly flammable atmosphere that requires specialized explosion-proof infrastructure. Furthermore, the use of metallic sodium in Method II presents similar hazards, alongside difficulties in handling solid metal dispersions on a multi-ton scale. These conventional routes often suffer from inconsistent yields and require extensive safety mitigation measures that inflate capital expenditure and operational costs, making them less attractive for modern, compliant chemical manufacturing facilities.
The Novel Approach
The innovative methodology described in the patent circumvents these legacy issues by employing sodium tert-amylate as the base catalyst in a tetrahydrofuran (THF) solvent system. This approach fundamentally alters the safety profile of the reaction; unlike sodium hydride, sodium tert-amylate does not generate hydrogen gas upon reaction with the hydroxyl group of the R-methyl lactate substrate. The process involves mixing R-methyl lactate with the base at low temperatures (below 5°C) before the dropwise addition of benzyl bromide. This controlled environment suppresses side reactions and ensures high conversion rates. As illustrated in the reaction scheme below, the transformation proceeds smoothly to form the intermediate methyl ester, which is subsequently hydrolyzed to the target acid. This route not only simplifies the engineering controls required for the reactor but also utilizes readily available, cost-effective raw materials, positioning it as a superior alternative for industrial scale-up.
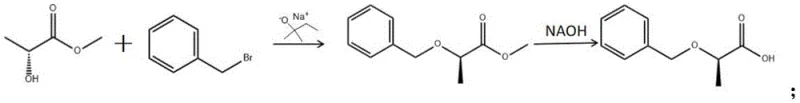
Mechanistic Insights into Sodium tert-Amylate Catalyzed Etherification
The core of this technological breakthrough lies in the specific reactivity and steric properties of sodium tert-amylate. In the initial step, the alkoxide acts as a strong, non-nucleophilic base to deprotonate the hydroxyl group of the (R)-methyl lactate, generating the corresponding alkoxide intermediate in situ. Unlike smaller alkoxides like sodium methoxide, the bulky tert-amyl group provides a degree of steric shielding that can help modulate the reactivity of the oxygen anion, favoring the desired SN2 attack on the benzyl bromide while minimizing competing elimination reactions or transesterification side pathways. The reaction is conducted in THF, a polar aprotic solvent that effectively solvates the cation (Na+) while leaving the alkoxide anion relatively "naked" and nucleophilic, thereby accelerating the substitution rate. Crucially, the maintenance of low temperatures during the addition phase is mechanistically vital; it kinetically traps the desired transition state and prevents thermal energy from overcoming the activation barrier for racemization at the chiral center, thus preserving the enantiomeric excess of the starting material throughout the synthesis.
Impurity control is another critical aspect where this mechanism excels. In conventional methods using NaH, local hot spots and rapid gas evolution can lead to uneven mixing and the formation of degradation byproducts. The homogeneous nature of the sodium tert-amylate reaction mixture ensures uniform heat distribution and reaction progression. Post-reaction workup involves a straightforward extraction protocol where the organic phase is washed and concentrated. The subsequent hydrolysis step using sodium hydroxide is robust and high-yielding, converting the intermediate ester to the free acid with minimal loss of optical purity. The patent data indicates that by optimizing the molar ratios—specifically maintaining a slight excess of benzyl bromide and base (1:1 to 1:2)—the process consistently achieves purity levels exceeding 99%, demonstrating the efficacy of this mechanistic pathway in suppressing trace impurities that are often difficult to remove in chiral pharmaceutical intermediates.
How to Synthesize (R)-2-Benzyloxypropionic Acid Efficiently
Implementing this synthesis route requires precise adherence to temperature profiles and stoichiometric ratios to maximize the benefits of the sodium tert-amylate system. The process begins with the dissolution of the chiral lactate in THF, followed by the careful addition of the base under inert atmosphere. The subsequent addition of the benzylating agent must be strictly controlled to manage the exotherm, even though it is less violent than NaH reactions. Once the intermediate is formed, the hydrolysis step is performed under reflux conditions to ensure complete conversion. For detailed operational parameters, including specific solvent volumes, stirring rates, and quenching procedures, please refer to the standardized synthesis guide provided below.
- Prepare the reaction mixture by dissolving R-methyl lactate in tetrahydrofuran (THF) and adding sodium tert-amylate under controlled temperature conditions below 5°C.
- Dropwise add benzyl bromide to the cooled mixture while maintaining the temperature below 5°C to prevent side reactions and ensure stereochemical integrity.
- Allow the reaction to proceed at room temperature (20-30°C) until completion, followed by hydrolysis with sodium hydroxide and acidification to isolate the final acid product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the shift to this sodium tert-amylate mediated process translates into tangible strategic advantages beyond mere chemical yield. The elimination of pyrophoric reagents like sodium hydride removes the need for specialized storage and handling protocols, drastically simplifying the logistics of raw material intake and warehouse management. This change directly contributes to cost reduction in pharmaceutical intermediate manufacturing by lowering insurance premiums, reducing safety equipment maintenance costs, and minimizing the downtime associated with rigorous safety inspections required for hydrogen-generating processes. Furthermore, the use of commodity chemicals like benzyl bromide and R-methyl lactate ensures a stable supply chain, insulating production schedules from the volatility often seen with specialty metal reagents.
- Cost Reduction in Manufacturing: The replacement of expensive and hazardous sodium hydride with sodium tert-amylate results in significant raw material cost savings. Additionally, the absence of hydrogen gas evolution means that reactors do not require complex venting systems or inert gas purging to the same extent as traditional methods, leading to lower utility consumption and reduced capital investment in safety infrastructure. The high yield and purity reported in the patent examples imply less waste generation and lower downstream purification costs, further enhancing the overall economic efficiency of the production line.
- Enhanced Supply Chain Reliability: By utilizing widely available organic bases and solvents, the process reduces dependency on single-source suppliers of hazardous metals. This diversification of the supply base enhances resilience against market disruptions. The robustness of the reaction conditions, which tolerate standard industrial equipment without requiring exotic metallurgy to resist corrosion from aggressive metal dispersions, ensures that production can be easily transferred between different manufacturing sites or CDMO partners without significant requalification efforts, thereby securing long-term supply continuity for downstream API manufacturers.
- Scalability and Environmental Compliance: The inherent safety of the non-gassing reaction profile makes this method ideally suited for commercial scale-up of complex pharmaceutical intermediates. Scaling from kilogram to tonnage quantities does not introduce exponential safety risks, allowing for larger batch sizes and improved throughput. From an environmental perspective, the process generates fewer hazardous byproducts and avoids the release of toxic gases, aligning with increasingly stringent global environmental regulations. This compliance facilitates smoother permitting processes and reduces the burden on wastewater treatment facilities, supporting sustainable manufacturing practices.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and comparative examples provided in the patent documentation, offering clarity on process optimization and quality control. Understanding these nuances is essential for technical teams evaluating the feasibility of adopting this route for their specific production needs.
Q: Why is sodium tert-amylate preferred over sodium hydride for this synthesis?
A: Sodium tert-amylate eliminates the generation of flammable hydrogen gas, significantly reducing explosion risks associated with sodium hydride usage in large-scale etherification processes.
Q: What are the typical yield and purity levels achievable with this method?
A: Optimized conditions using a molar ratio of 1:1.2 for reactants can achieve yields exceeding 90% and purity levels greater than 99.5%, suitable for high-grade pharmaceutical applications.
Q: How does temperature control impact the stereochemistry of the product?
A: Maintaining the addition temperature below 5°C is critical to minimizing racemization and suppressing the formation of isomeric impurities, ensuring the optical purity of the (R)-enantiomer.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (R)-2-Benzyloxypropionic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting safer, more efficient synthetic routes for high-value chiral intermediates. Our technical team has extensively analyzed the methodology described in CN113233972B and possesses the expertise to implement this sodium tert-amylate catalyzed process at an industrial scale. We boast extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab-scale optimization to full-scale manufacturing is seamless. Our facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of (R)-2-benzyloxypropionic acid meets the exacting standards required for pharmaceutical applications.
We invite potential partners to engage with our technical sales team to discuss how this advanced synthesis route can optimize your supply chain. By leveraging our manufacturing capabilities, you can secure a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact our technical procurement team today to request specific COA data and comprehensive route feasibility assessments, ensuring that your project moves forward with the most reliable and economically viable chemical solution available.
