Advanced Solid-Phase Synthesis of 8-Arylamino-3H-Imidazo[4,5-g]Quinazoline Derivatives for Oncology
Introduction to Next-Generation EGFR Inhibitor Scaffolds
The landscape of oncology drug discovery has been significantly transformed by the development of epidermal growth factor receptor (EGFR) tyrosine kinase inhibitors. As detailed in patent CN100522965C, a novel class of 8-arylamino-3H-imidazo[4,5-g]quinazoline derivatives has emerged as a potent scaffold for targeting these critical signaling pathways. Unlike traditional quinazolines, this invention introduces a linearly arranged tricyclic system where an imidazole ring is fused to the benzene ring of the quinazoline parent structure. This structural modification not only enhances binding affinity but also improves the physicochemical properties necessary for cellular uptake. The patent outlines a robust solid-phase synthesis methodology that facilitates the rapid generation of molecular libraries, allowing medicinal chemists to explore diverse substitution patterns at the 2, 3, and 8 positions efficiently.
![General structural formula of 8-arylamino-3H-imidazo[4,5-g]quinazoline derivatives showing variable R groups](/insights/img/8-arylamino-imidazoquinazoline-solid-phase-synthesis-supplier-20260304232321-01.png)
For R&D directors seeking high-purity pharmaceutical intermediates, this technology offers a distinct advantage by enabling the systematic variation of substituents such as alkoxy, alkyl, and halogen groups. The ability to rapidly synthesize these complex heterocycles is crucial for high-throughput screening campaigns aimed at identifying lead compounds against various cancer cell lines, including HepG2, U251, and MCF-7. By leveraging this patented approach, organizations can accelerate their pipeline development while maintaining rigorous control over impurity profiles and structural integrity.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional solution-phase synthesis of complex heterocyclic libraries often presents significant bottlenecks in both time and resource allocation. Conventional routes typically require multiple isolation and purification steps, such as column chromatography, after each synthetic transformation. This not only increases the consumption of solvents and silica gel but also leads to substantial material loss, reducing overall yields. Furthermore, handling diverse intermediates in solution can be cumbersome when attempting to introduce variability at multiple positions simultaneously. The accumulation of by-products and the difficulty in driving reactions to completion without degrading sensitive functional groups are common challenges that hinder the efficient exploration of chemical space.
The Novel Approach
In contrast, the solid-phase synthesis method described in the patent revolutionizes the production of these derivatives by anchoring the growing molecule to an insoluble polymer support. This strategy allows for the use of excess reagents to drive reactions to completion, with unreacted materials and by-products simply washed away. The process begins with the preparation of a key intermediate, 4-chloro-7-fluoro-6-nitroquinazoline, which is then coupled to a resin-bound amine. This modular approach enables the parallel synthesis of numerous analogues by varying the amine loaded onto the resin and the aldehyde used in the final cyclization step. Consequently, this method drastically simplifies the workflow, reduces solvent usage, and enhances the speed at which new candidates can be generated for biological evaluation.
Mechanistic Insights into Solid-Phase Cyclization and Substitution
The core of this synthetic strategy lies in the sequential construction of the tricyclic framework on a solid support. The process initiates with the synthesis of the quinazoline core from 2-amino-4-fluorobenzoic acid and acetamidine, followed by nitration and chlorination to activate the 4-position for nucleophilic attack. A critical mechanistic step involves the displacement of the fluorine atom at the 7-position by a primary amine (R2-NH2) attached to the resin-bound arylamine. This nucleophilic aromatic substitution is facilitated by the electron-withdrawing nature of the adjacent nitro group, ensuring high regioselectivity. The subsequent reductive cyclization is particularly noteworthy; it employs tin(II) chloride (SnCl2) in DMF alongside an aldehyde (R3-CHO) to simultaneously reduce the nitro group and condense it with the amine and aldehyde to form the imidazole ring.
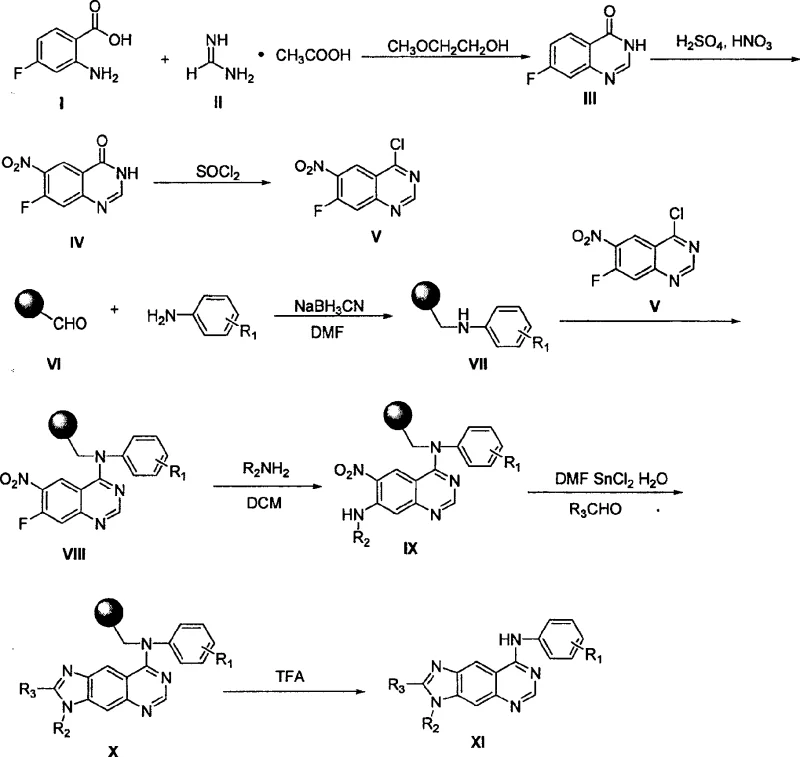
From an impurity control perspective, the use of solid-phase synthesis inherently minimizes the carryover of side products. Since the intermediate remains bound to the resin throughout the sequence, soluble impurities generated during the nitration or chlorination steps are removed during the washing phases prior to resin loading. The final cleavage step using a mixture of trifluoroacetic acid and dichloromethane releases the target molecule from the resin. This cleavage condition is mild enough to preserve the integrity of the newly formed imidazole ring while effectively breaking the linker bond. For procurement managers focused on cost reduction in API manufacturing, understanding these mechanistic details highlights the efficiency of the process, as it eliminates the need for expensive purification columns and reduces the labor hours associated with traditional workups.
How to Synthesize 8-Arylamino-3H-Imidazo[4,5-g]Quinazoline Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for producing these valuable intermediates with high fidelity. The procedure involves preparing the quinazoline precursor separately and then engaging it with a functionalized resin. Key steps include the reductive amination of an aldehyde resin with a substituted aniline, followed by coupling with the chloro-quinazoline intermediate. The final cyclization is achieved under mild thermal conditions (50°C) using SnCl2 as the reducing agent. This standardized approach ensures reproducibility and scalability, making it ideal for both laboratory screening and pilot plant operations. Detailed standard operating procedures for each reaction stage, including specific molar ratios and solvent volumes, are essential for maintaining batch-to-batch consistency.
- Preparation of the quinazoline core via cyclization of 2-amino-4-fluorobenzoic acid followed by nitration and chlorination.
- Loading of substituted anilines onto aldehyde resin via reductive amination to form the resin-bound amine.
- Sequential nucleophilic substitution with the chloro-quinazoline, followed by amine displacement and reductive cyclization with aldehyde/SnCl2.
Commercial Advantages for Procurement and Supply Chain Teams
Adopting this solid-phase synthesis route offers profound benefits for supply chain stability and operational expenditure. By shifting from a linear solution-phase process to a convergent solid-phase methodology, manufacturers can significantly streamline their production workflows. The elimination of intermediate isolation steps reduces the total processing time and minimizes the risk of material degradation during storage or transfer. Furthermore, the use of commercially available starting materials, such as 2-amino-4-fluorobenzoic acid and various substituted anilines, ensures a reliable supply chain that is not dependent on obscure or hard-to-source reagents. This accessibility is critical for maintaining continuous production schedules and mitigating the risks associated with raw material shortages.
- Cost Reduction in Manufacturing: The primary driver for cost optimization in this process is the drastic reduction in purification costs. Traditional methods often require extensive chromatography, which consumes large volumes of solvents and stationary phases. In this solid-phase approach, purification is achieved through simple filtration and washing, leading to substantial savings in consumables and waste disposal fees. Additionally, the ability to use excess reagents without complicating the purification process improves reaction yields, thereby maximizing the output per unit of raw material input. This efficiency translates directly into a lower cost of goods sold (COGS) for the final active pharmaceutical ingredient.
- Enhanced Supply Chain Reliability: The robustness of the synthetic route contributes to a more resilient supply chain. The reaction conditions are relatively mild, avoiding the need for extreme temperatures or pressures that could pose safety hazards or require specialized equipment. The reagents used, such as thionyl chloride and tin(II) chloride, are standard industrial chemicals with stable global supply networks. This reduces the likelihood of production delays caused by logistical bottlenecks. Moreover, the modular nature of the synthesis allows for flexible manufacturing, where different analogues can be produced on the same equipment line with minimal changeover time, enhancing overall asset utilization.
- Scalability and Environmental Compliance: Scaling up solid-phase synthesis is inherently more straightforward than optimizing complex solution-phase cascades. The process generates less liquid waste per kilogram of product due to the reduced need for extraction and chromatography solvents. This aligns well with modern environmental, health, and safety (EHS) standards, reducing the burden on wastewater treatment facilities. The high purity of the crude product obtained after cleavage often means that only a final recrystallization or simple column chromatography is needed, further minimizing the environmental footprint. For supply chain heads, this means easier regulatory compliance and a smoother path to commercial validation.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis and application of these quinazoline derivatives. Understanding these aspects is vital for stakeholders evaluating the feasibility of integrating this technology into their drug discovery pipelines. The answers are derived directly from the experimental data and technical specifications provided in the patent documentation, ensuring accuracy and relevance for decision-makers.
Q: What is the primary advantage of the solid-phase method for these derivatives?
A: The solid-phase approach allows for rapid combinatorial synthesis and simplified purification through filtration and washing, eliminating the need for complex chromatography at every intermediate step.
Q: What are the key reaction conditions for the final cyclization step?
A: The final imidazole ring formation utilizes tin(II) chloride dihydrate in DMF with an aldehyde at 50°C for 1 hour, ensuring mild conditions compatible with diverse functional groups.
Q: Is this synthesis route suitable for large-scale manufacturing?
A: Yes, the route uses readily available starting materials and avoids hazardous reagents, making it highly adaptable for commercial scale-up from kilogram to metric ton quantities.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 8-Arylamino-3H-Imidazo[4,5-g]Quinazoline Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-quality intermediates in the development of next-generation anticancer therapies. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from lead optimization to clinical supply is seamless. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of 8-arylamino-3H-imidazo[4,5-g]quinazoline derivatives meets the highest industry standards. Our commitment to quality assurance means that you can rely on us for consistent material that supports reproducible biological data.
We invite you to collaborate with us to leverage this advanced solid-phase synthesis technology for your specific project needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements. Please contact us to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise can accelerate your oncology drug development program while optimizing your manufacturing budget.
