Advanced Synthesis of Scutellarin Aglycone Derivatives for High-Purity Antitumor Pharmaceutical Intermediates
Advanced Synthesis of Scutellarin Aglycone Derivatives for High-Purity Antitumor Pharmaceutical Intermediates
The escalating demand for novel antitumor agents has driven significant innovation in the synthesis of flavonoid-inspired scaffolds. Patent CN107501222B introduces a groundbreaking class of scutellarin aglycone derivatives, characterized by a distinct chromone backbone substituted with various aryl and heteroaryl groups. These compounds are not merely theoretical constructs but represent tangible advancements in medicinal chemistry, offering potent inhibition against a spectrum of malignancies including leukemia, lung cancer, and hepatocellular carcinoma. For R&D directors and procurement specialists in the pharmaceutical sector, this patent signifies a shift from reliance on variable natural extraction to robust, reproducible chemical synthesis. The disclosed methodologies provide a clear pathway for generating high-purity pharmaceutical intermediates that can serve as critical building blocks for next-generation oncology therapeutics, addressing both the efficacy and supply chain stability concerns prevalent in the industry today.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the sourcing of scutellarin aglycone and its analogs has been heavily dependent on extraction from botanical sources such as Erigeron breviscapus or other flavonoid-rich plants. This biological sourcing model presents inherent vulnerabilities for industrial supply chains, including seasonal variability, geographical constraints, and the co-extraction of complex impurity profiles that complicate downstream purification. Furthermore, semi-synthetic modifications of natural extracts often suffer from low regioselectivity, requiring extensive chromatographic separation which is economically unfeasible at a commercial scale. The structural rigidity of natural scaffolds also limits the ability to perform Structure-Activity Relationship (SAR) studies efficiently, as introducing specific substituents to optimize pharmacokinetic properties is chemically challenging without a total synthesis approach. These bottlenecks result in inconsistent batch-to-batch quality and inflated costs, hindering the rapid development of derivative libraries needed for modern drug discovery pipelines.
The Novel Approach
In stark contrast, the methodology detailed in CN107501222B employs a fully synthetic strategy that decouples production from agricultural variables. The patent discloses two primary convergent routes: one initiating with an esterification-rearrangement sequence and another utilizing a Claisen-Schmidt condensation followed by oxidative cyclization. 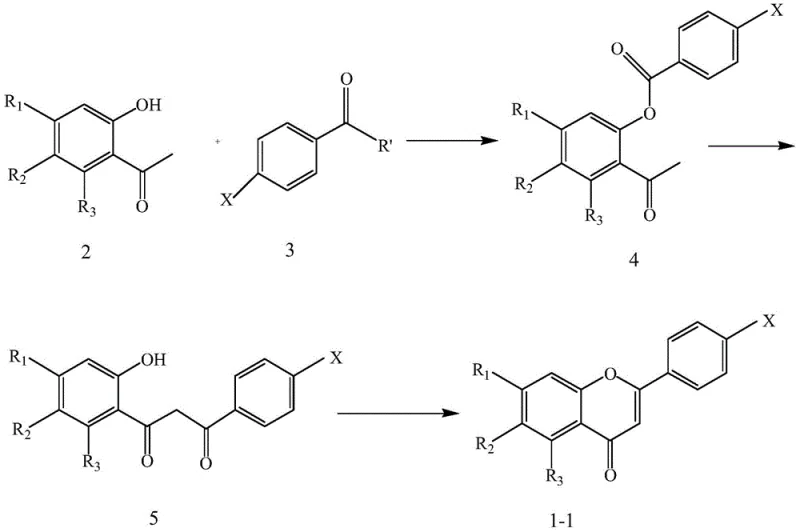
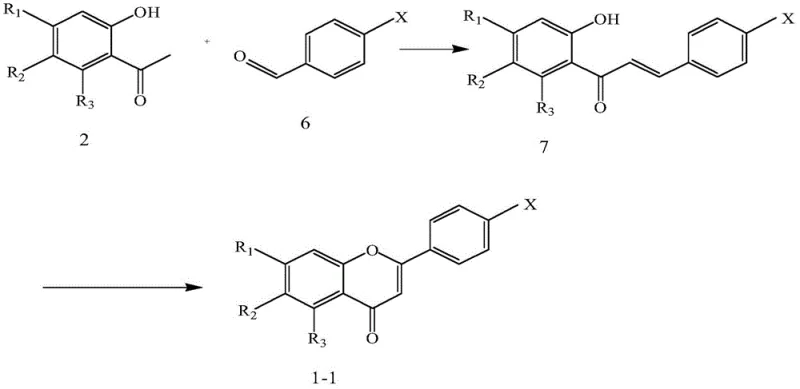 These approaches utilize commodity chemicals such as substituted acetophenones and benzoyl chlorides or aldehydes, which are globally available and cost-effective. The synthetic design allows for precise control over the substitution pattern on the A and B rings of the chromone core, enabling the systematic exploration of chemical space. By establishing a linear, step-wise progression from simple precursors to complex targets, this novel approach ensures reproducibility and scalability, effectively transforming the production of these valuable intermediates from an artisanal extraction process into a streamlined industrial chemical operation.
These approaches utilize commodity chemicals such as substituted acetophenones and benzoyl chlorides or aldehydes, which are globally available and cost-effective. The synthetic design allows for precise control over the substitution pattern on the A and B rings of the chromone core, enabling the systematic exploration of chemical space. By establishing a linear, step-wise progression from simple precursors to complex targets, this novel approach ensures reproducibility and scalability, effectively transforming the production of these valuable intermediates from an artisanal extraction process into a streamlined industrial chemical operation.
Mechanistic Insights into Iodine-Catalyzed Oxidative Cyclization
A pivotal mechanistic feature of this technology is the iodine-catalyzed oxidative cyclization step, particularly in the second synthetic route. This transformation converts a chalcone-like intermediate (compound 7 or 8) into the rigid chromone system (compound 1-1 or 1-3) under relatively mild thermal conditions (130°C in DMSO). The mechanism likely involves the activation of the alkene moiety by molecular iodine, facilitating an intramolecular nucleophilic attack by the phenolic hydroxyl group. This is followed by an oxidation step that restores aromaticity and establishes the conjugated carbonyl system essential for the molecule's biological activity. The use of iodine as a catalyst is particularly advantageous from a process chemistry perspective; it is inexpensive, readily available, and easier to remove from the final product compared to heavy metal catalysts. This step effectively locks the conformational flexibility of the precursor, creating the planar structure required for intercalation or binding within biological targets, thereby enhancing the compound's potency against tumor cells.
Furthermore, the patent highlights the versatility of the scaffold through post-synthetic modification via palladium-catalyzed cross-coupling. 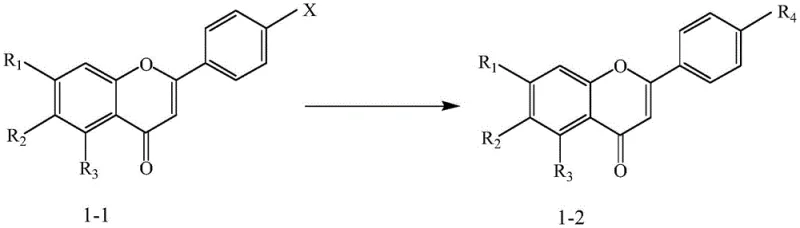 This allows for the introduction of diverse aryl groups at the 2-position of the chromone ring, as seen in the conversion of the bromo-intermediate (1-1) to various fluoro- or chloro-substituted analogs (1-2). From an impurity control standpoint, the modularity of this approach means that side reactions are largely confined to specific steps, making the impurity profile predictable and manageable. The use of standard bases like potassium carbonate or cesium carbonate in the coupling steps ensures that the reaction environment remains compatible with sensitive functional groups, minimizing degradation products. This level of mechanistic control is crucial for R&D teams aiming to meet stringent regulatory requirements for impurity identification and quantification in new drug applications.
This allows for the introduction of diverse aryl groups at the 2-position of the chromone ring, as seen in the conversion of the bromo-intermediate (1-1) to various fluoro- or chloro-substituted analogs (1-2). From an impurity control standpoint, the modularity of this approach means that side reactions are largely confined to specific steps, making the impurity profile predictable and manageable. The use of standard bases like potassium carbonate or cesium carbonate in the coupling steps ensures that the reaction environment remains compatible with sensitive functional groups, minimizing degradation products. This level of mechanistic control is crucial for R&D teams aiming to meet stringent regulatory requirements for impurity identification and quantification in new drug applications.
How to Synthesize Scutellarin Aglycone Derivatives Efficiently
The synthesis of these high-value intermediates is designed to be operationally simple, utilizing standard laboratory equipment that translates well to pilot and production scales. The process generally begins with the activation of a phenolic acetophenone, either through acylation or aldol condensation, depending on the desired substitution pattern. The subsequent cyclization steps are thermally driven and do not require exotic reagents or high-pressure equipment, reducing the capital expenditure required for implementation. Detailed standard operating procedures for each transformation, including specific solvent systems like dichloromethane for esterification and DMSO for cyclization, are provided to ensure consistency. For a comprehensive breakdown of the specific reaction conditions, stoichiometry, and workup procedures required to achieve the reported yields, please refer to the standardized synthesis guide below.
- Perform esterification of acetophenone derivatives with benzoyl chlorides using DMAP in DCM, followed by base-catalyzed rearrangement at 110°C to form the 1,3-diketone intermediate.
- Alternatively, conduct a Claisen-Schmidt condensation between acetophenones and aldehydes using t-BuOK, followed by iodine-catalyzed oxidative cyclization in DMSO at 130°C.
- Purify the resulting chromone scaffolds and optionally functionalize the aryl ring via palladium-catalyzed Suzuki coupling to introduce diverse substituents.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to the synthetic routes described in this patent offers profound strategic benefits beyond mere technical feasibility. The primary advantage lies in the decoupling of raw material supply from agricultural cycles. By relying on petrochemical-derived starting materials such as acetophenones and benzaldehydes, manufacturers can secure long-term contracts with stable pricing, insulating the production of these critical intermediates from the volatility of crop yields and climate change impacts. This stability is essential for maintaining continuous supply lines for downstream API manufacturing, preventing costly production stoppages due to raw material shortages. Additionally, the synthetic nature of the process allows for inventory management of key intermediates, enabling a "make-to-stock" strategy that significantly reduces lead times for customer orders compared to the seasonal "make-to-order" constraints of botanical extraction.
- Cost Reduction in Manufacturing: The elimination of expensive extraction solvents and the reduction of purification steps associated with natural product isolation lead to substantial cost savings. The synthetic routes utilize commodity reagents and catalysts, such as iodine and palladium complexes, which are used in catalytic amounts and can often be recovered or sourced at low cost. Furthermore, the high selectivity of the reactions minimizes the formation of difficult-to-remove byproducts, reducing the burden on downstream purification processes like preparative HPLC. This efficiency translates directly into a lower cost of goods sold (COGS), allowing for more competitive pricing in the global market for pharmaceutical intermediates while maintaining healthy margins.
- Enhanced Supply Chain Reliability: The modular design of the synthesis, particularly the ability to diversify the final product via Suzuki coupling from a common bromo-intermediate, creates a resilient supply chain architecture. Instead of maintaining separate inventory lines for dozens of different analogs, a manufacturer can stockpile the common precursor and rapidly convert it to the specific derivative required by a client. This agility drastically reduces the risk of obsolescence and allows for rapid response to shifting market demands. It also simplifies the qualification of suppliers, as the core process remains constant regardless of the final substituent, streamlining the audit and validation processes required by regulatory bodies.
- Scalability and Environmental Compliance: The reactions described operate at atmospheric pressure and moderate temperatures, removing the need for specialized high-pressure reactors and enhancing safety profiles for large-scale operations. The solvent systems employed, such as ethyl acetate and ethanol, are greener alternatives to chlorinated solvents often used in older methodologies, aligning with increasingly strict environmental regulations. The simplicity of the workup procedures, involving standard aqueous extractions and crystallizations, facilitates waste stream management and solvent recovery. This environmental compatibility not only reduces disposal costs but also future-proofs the manufacturing process against tightening global emissions standards, ensuring long-term operational viability.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these scutellarin aglycone derivatives. The answers are derived directly from the experimental data and process descriptions within the patent documentation, providing a transparent view of the technology's capabilities. Understanding these details is vital for stakeholders evaluating the feasibility of integrating these intermediates into their own development pipelines or supply networks.
Q: What are the primary advantages of the synthetic routes described in CN107501222B?
A: The patent outlines two complementary routes: one via esterification-rearrangement and another via condensation-cyclization. Both utilize readily available starting materials like acetophenones and benzaldehydes, avoiding the complexity and variability of natural extraction, thereby ensuring consistent quality and supply continuity for pharmaceutical manufacturing.
Q: How does the Suzuki coupling step enhance the versatility of these intermediates?
A: The inclusion of a palladium-catalyzed Suzuki coupling step allows for the late-stage introduction of diverse aryl groups (e.g., fluorophenyl, chlorophenyl) onto the core chromone scaffold. This modularity enables rapid generation of analog libraries for SAR studies without redesigning the entire synthetic pathway, significantly accelerating drug discovery timelines.
Q: What is the biological potential of these scutellarin aglycone derivatives?
A: Experimental data indicates significant antitumor activity against various cell lines including leukemia (HL-60), lung cancer (A-549), and liver cancer (SMMC-7721). Specific compounds demonstrated inhibition rates comparable to or exceeding positive controls like cisplatin, highlighting their potential as lead compounds for oncology drug development.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Scutellarin Aglycone Derivative Supplier
The technological breakthroughs encapsulated in CN107501222B represent a significant opportunity for the pharmaceutical industry, yet realizing this potential requires a manufacturing partner with deep technical expertise and robust infrastructure. NINGBO INNO PHARMCHEM stands at the forefront of this domain, possessing extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our state-of-the-art facilities are equipped to handle the specific thermal and catalytic requirements of these synthetic routes, ensuring that every batch meets stringent purity specifications. With our rigorous QC labs and commitment to process optimization, we guarantee the delivery of high-purity pharmaceutical intermediates that adhere to the highest international standards, providing a solid foundation for your drug development programs.
We invite you to collaborate with us to unlock the full commercial potential of these antitumor candidates. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how our optimized processes can reduce your overall procurement costs. We encourage you to contact our technical procurement team today to request specific COA data for our catalog compounds and to discuss route feasibility assessments for your proprietary analogs. Together, we can accelerate the journey from bench-scale discovery to life-saving therapies.
