Advanced Synthesis of Tetrahydropyrazolopiperazine Derivatives for Commercial Pharmaceutical Applications
The pharmaceutical industry continuously seeks novel small molecule compounds that can enhance therapeutic efficacy while minimizing systemic toxicity, particularly in the realm of oncology. Patent CN110156816B introduces a significant advancement in the synthesis of tetrahydropyrazolopiperazine derivatives, specifically targeting human breast cancer treatment protocols. This intellectual property details a robust preparation method for (R)-6-methyl-3-(4-(isopropylsulfonyl)phenyl)-1H-pyrrolo[2,3-b]pyridine)-4,5,6,7-tetrahydropyrazolo[1,5-a]piperazine, a complex heterocyclic structure with promising biological activity. The disclosed technology addresses critical bottlenecks in prior art, such as harsh reaction conditions and suboptimal yields, by implementing a streamlined multi-step synthetic route. For R&D directors and procurement specialists, this patent represents a viable pathway for developing next-generation anticancer agents that can be synergistically combined with established drugs like doxorubicin. The strategic value lies not only in the biological potential but also in the chemical feasibility of manufacturing this high-purity pharmaceutical intermediate at a commercial scale.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of pyrazolopiperazine scaffolds has been plagued by significant chemical challenges that hinder efficient large-scale production. Traditional routes often require extreme reaction conditions, including high temperatures and pressures, which increase energy consumption and operational risks in a manufacturing setting. Furthermore, conventional methods frequently suffer from low overall yields due to the formation of complex byproduct profiles and the instability of intermediate species during the cyclization steps. The reliance on expensive or difficult-to-remove catalysts in older methodologies also complicates the purification process, leading to higher costs associated with downstream processing and waste management. These inefficiencies create substantial barriers for supply chain heads who need reliable, cost-effective sources of high-purity intermediates for clinical and commercial applications. The inability to consistently produce these compounds with stringent purity specifications often delays drug development timelines and increases the financial burden on pharmaceutical companies seeking to bring new therapies to market.
The Novel Approach
The methodology outlined in patent CN110156816B offers a transformative solution by optimizing the synthetic pathway to overcome these historical limitations. This novel approach utilizes a sequence of well-controlled reactions, including reductive amination and palladium-catalyzed coupling, which proceed under milder conditions compared to traditional methods. By carefully selecting reagents such as sodium triacetoxyborohydride and specific palladium catalysts, the process achieves higher conversion rates and minimizes the formation of unwanted impurities. The strategic design of the synthesis route allows for easier purification using standard silica gel column chromatography, significantly reducing the complexity of isolating the final product. This improvement in process chemistry translates directly to enhanced manufacturability, making it a highly attractive option for cost reduction in pharmaceutical intermediate manufacturing. The ability to produce the target tetrahydropyrazolopiperazine compound with improved yield and purity establishes a stronger foundation for scaling up production to meet global demand.
Mechanistic Insights into Suzuki Coupling and Cyclization
A deep understanding of the reaction mechanism is crucial for R&D teams evaluating the feasibility of this synthesis for commercial adoption. The core of this synthetic strategy involves a sophisticated palladium-catalyzed Suzuki coupling reaction, which connects the tetrahydropyrazolo piperazine core with the functionalized phenylboronic acid moiety. This step is critical for introducing the isopropylsulfonyl group, which plays a vital role in the biological activity of the final molecule. The use of PdCl2 as a catalyst in a 1,4-dioxane and potassium carbonate solvent system ensures efficient cross-coupling while maintaining the stereochemical integrity of the chiral centers within the molecule. The reaction conditions, specifically the reflux at 110°C under nitrogen, are optimized to maximize the turnover number of the catalyst and ensure complete consumption of the starting materials. This mechanistic precision is essential for achieving the high purity required for pharmaceutical applications, as it minimizes the presence of residual metals and unreacted halides that could compromise safety profiles.
Furthermore, the impurity control mechanism is inherently built into the stepwise progression of the synthesis, particularly during the cyclization and deprotection phases. The intramolecular cyclization step, facilitated by thionyl chloride and sodium hydride, is carefully controlled at 0°C to prevent side reactions that could lead to structural isomers or degradation products. Subsequent deprotection steps using ammonium formate and palladium on carbon are designed to selectively remove protecting groups without affecting the sensitive heterocyclic rings. This level of control over the chemical transformation ensures that the final product, Compound I, meets stringent quality standards with minimal impurity burdens. For quality assurance teams, this predictable impurity profile simplifies the validation process and reduces the risk of batch failures during commercial production. The robust nature of this chemistry provides confidence in the consistency of the supply, which is a key factor for long-term procurement planning.
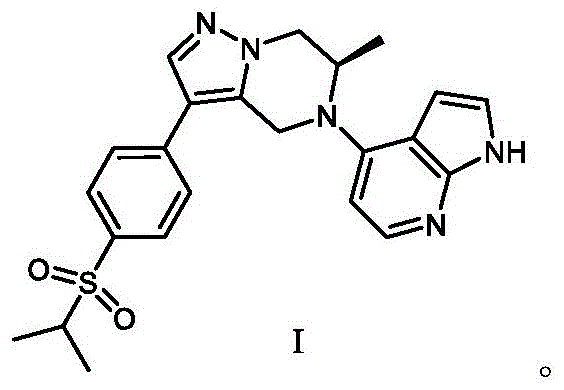
How to Synthesize (R)-6-methyl-3-(4-(isopropylsulfonyl)phenyl) Derivatives Efficiently
Implementing this synthesis route requires a systematic approach to ensure reproducibility and safety at every stage of the manufacturing process. The patent provides a detailed blueprint that begins with the reductive amination of 3-aldehyde-4-bromopyrazole, followed by cyclization to form the core piperazine structure. Subsequent steps involve precise stoichiometric control during the Suzuki coupling and final deprotection to yield the target molecule. Operators must adhere to strict temperature controls and inert atmosphere conditions, particularly during the palladium-catalyzed steps, to maintain reaction efficiency. The detailed standardized synthesis steps see the guide below for specific operational parameters and safety precautions required for industrial execution.
- Perform reductive amination of 3-aldehyde-4-bromopyrazole with (R)-2-(benzylamino)propan-1-ol using sodium triacetoxyborohydride.
- Execute intramolecular cyclization using thionyl chloride and sodium hydride to form the tetrahydropyrazolo piperazine core.
- Conduct Suzuki coupling with 4-(isopropylsulfonyl)phenylboronic acid followed by deprotection and final amination to yield the target compound.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this patented synthesis route offers substantial benefits for procurement managers and supply chain directors looking to optimize their sourcing strategies. The improved yield and simplified purification process directly contribute to significant cost savings in pharmaceutical intermediate manufacturing by reducing raw material waste and processing time. The elimination of extremely harsh reaction conditions also lowers the energy requirements and safety risks associated with production, further enhancing the economic viability of the process. For supply chain heads, the robustness of this chemistry ensures a more reliable tetrahydropyrazolopiperazine supplier capability, reducing the risk of disruptions due to batch failures or quality issues. The ability to scale this process from laboratory to commercial quantities without significant re-engineering provides a clear pathway for meeting increasing market demand for oncology intermediates.
- Cost Reduction in Manufacturing: The streamlined synthetic route eliminates the need for expensive and difficult-to-remove catalysts often found in conventional methods, leading to substantial cost savings. By optimizing the reaction conditions to achieve higher yields, the process reduces the amount of starting materials required per unit of final product, effectively lowering the cost of goods sold. Additionally, the use of standard purification techniques minimizes the need for specialized equipment or complex downstream processing, further driving down operational expenses. These efficiencies allow for a more competitive pricing structure without compromising on the quality or purity of the pharmaceutical intermediate supplied to clients.
- Enhanced Supply Chain Reliability: The robustness of the chemical process ensures consistent production output, which is critical for maintaining a stable supply of high-purity pharmaceutical intermediates. By avoiding unstable intermediates and harsh conditions that often lead to batch variability, this method enhances the predictability of delivery schedules. This reliability is essential for pharmaceutical companies that depend on timely access to key building blocks for their drug development pipelines. A stable supply chain reduces the need for excessive safety stock and allows for more efficient inventory management, ultimately strengthening the partnership between the manufacturer and the end-user.
- Scalability and Environmental Compliance: The synthesis route is designed with scalability in mind, utilizing reagents and solvents that are compatible with large-scale industrial equipment. The milder reaction conditions reduce the environmental footprint of the manufacturing process by lowering energy consumption and minimizing the generation of hazardous waste. This alignment with green chemistry principles supports environmental compliance and sustainability goals, which are increasingly important for corporate social responsibility initiatives. The ease of scaling up complex pharmaceutical intermediates ensures that production can be ramped up quickly to meet market demands without significant capital investment in new infrastructure.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of this tetrahydropyrazolopiperazine compound. These answers are derived directly from the technical specifications and beneficial effects described in the patent documentation to provide clarity for potential partners. Understanding these details is crucial for evaluating the feasibility of integrating this intermediate into your specific drug development or manufacturing workflow. We encourage reviewing these insights to fully appreciate the strategic value this technology brings to your supply chain.
Q: What are the primary advantages of this synthesis route over conventional methods?
A: The patented method overcomes difficult synthesis conditions and low yields associated with traditional pyrazolopiperazine production, utilizing optimized palladium-catalyzed steps for better scalability.
Q: How does this compound contribute to breast cancer treatment protocols?
A: The compound exhibits synergistic effects with doxorubicin, potentially allowing for reduced dosages of the chemotherapeutic agent while maintaining efficacy against MCF-7 cells.
Q: Is this intermediate suitable for large-scale commercial manufacturing?
A: Yes, the route avoids extremely harsh conditions and utilizes standard purification techniques like silica gel column chromatography, facilitating easier scale-up for industrial production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Tetrahydropyrazolopiperazine Supplier
NINGBO INNO PHARMCHEM stands at the forefront of custom synthesis and manufacturing, offering extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our technical team is well-versed in the complexities of heterocyclic chemistry and is fully equipped to replicate and optimize the synthesis route described in patent CN110156816B. We maintain stringent purity specifications and operate rigorous QC labs to ensure that every batch of tetrahydropyrazolopiperazine intermediate meets the highest industry standards. Our commitment to quality and consistency makes us an ideal partner for pharmaceutical companies seeking a reliable source for this critical oncology building block.
We invite you to engage with our technical procurement team to discuss how we can support your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into how our manufacturing capabilities can optimize your supply chain economics. We are ready to provide specific COA data and route feasibility assessments to demonstrate our capacity to deliver high-quality intermediates that align with your development timelines. Let us help you accelerate your drug discovery process with our proven expertise in complex chemical synthesis.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
