Revolutionizing Ester Production: Mild NHC Organocatalysis for Commercial Scale-up
Revolutionizing Ester Production: Mild NHC Organocatalysis for Commercial Scale-up
The landscape of fine chemical synthesis is undergoing a paradigm shift towards greener, more sustainable methodologies, and patent CN102260129A stands at the forefront of this evolution. This groundbreaking intellectual property details a novel application of N-heterocyclic carbenes (NHCs) in the direct esterification of aldehyde compounds with halogenated hydrocarbons. Unlike traditional pathways that rely heavily on scarce transition metals or harsh oxidative conditions, this organocatalytic approach leverages the unique electronic properties of nitrogen-containing heterocycles to facilitate bond formation under remarkably mild conditions. For R&D directors and process chemists, this represents a significant opportunity to redesign synthetic routes for aromatic carboxylic acid esters, which are critical scaffolds in drug discovery and agrochemical development. The technology promises to streamline manufacturing workflows by removing the necessity for expensive metal scavengers and complex inert atmosphere setups.
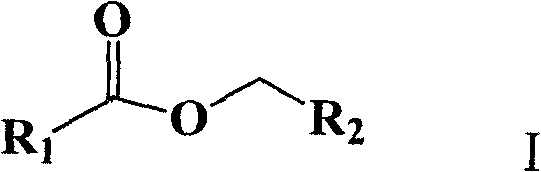
At the heart of this innovation is the ability to construct the ester linkage directly from readily available aldehydes and alkyl halides, bypassing the need for pre-oxidized carboxylic acids or activated acyl chlorides. The general structure of the target esters, as depicted in the patent, allows for significant structural diversity through the variation of R1 and R2 groups. This flexibility is crucial for medicinal chemists who require rapid access to diverse libraries of analogues for structure-activity relationship (SAR) studies. By enabling the use of simple starting materials under ambient pressure and near-room temperature conditions, this method addresses several pain points simultaneously: cost efficiency, operational safety, and environmental compliance. As we delve deeper into the technical specifics, it becomes clear why this methodology is poised to become a standard tool in the arsenal of reliable pharmaceutical intermediate supplier networks globally.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of aromatic carboxylic acid esters has been plagued by significant technical and economic hurdles that hinder efficient commercial production. Conventional methods often necessitate the use of precious metal catalysts such as palladium, rhodium, or ruthenium, which not only drive up raw material costs but also introduce severe supply chain vulnerabilities due to the geopolitical scarcity of these elements. Furthermore, these metal-catalyzed processes typically demand rigorous reaction conditions, including strictly anhydrous and oxygen-free environments, high pressures, and elevated temperatures. Maintaining such conditions on an industrial scale requires specialized equipment, extensive energy input, and complex safety protocols to prevent runaway reactions or catalyst deactivation. Additionally, the removal of trace metal residues from the final product is a costly and time-consuming purification step, often requiring specialized scavenging resins or repeated recrystallization, which inevitably lowers the overall yield and increases the environmental footprint through solvent waste generation.
The Novel Approach
In stark contrast, the methodology disclosed in CN102260129A offers a transformative solution by utilizing organic small molecule catalysis to drive the esterification reaction. This novel approach employs N-heterocyclic carbene precursors, specifically thiazolium salts, which generate the active catalytic species in situ under basic conditions. The reaction proceeds efficiently in the presence of oxygen, utilizing molecular oxygen from the air as a benign oxidant, which completely eliminates the need for expensive stoichiometric oxidants or inert gas blanketing. The reaction conditions are exceptionally mild, operating effectively at temperatures between 30°C and 40°C, which significantly reduces thermal stress on sensitive functional groups and minimizes energy consumption. This shift from metal-based to metal-free catalysis not only simplifies the workup procedure but also ensures that the final product is free from heavy metal contamination, a critical quality attribute for pharmaceutical intermediates intended for human therapeutic use.
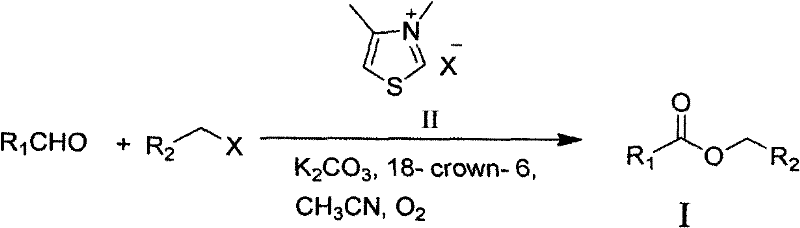
Mechanistic Insights into NHC-Catalyzed Oxidative Esterification
The mechanistic pathway of this transformation is a fascinating example of polarity inversion catalysis, often referred to as Umpolung chemistry. The N-heterocyclic carbene, generated from the thiazolium salt precursor upon deprotonation by a base like potassium carbonate, acts as a nucleophile that attacks the carbonyl carbon of the aldehyde substrate. This interaction forms a Breslow-like intermediate, effectively reversing the natural polarity of the carbonyl carbon from electrophilic to nucleophilic. In the presence of oxygen, this intermediate undergoes oxidation to form an acyl azolium species, which is a highly reactive acylating agent. This activated species is then susceptible to nucleophilic attack by the alcohol generated in situ or directly by the halide displacement mechanism depending on the specific pathway variant, ultimately yielding the ester product and regenerating the carbene catalyst. Understanding this cycle is vital for process optimization, as it highlights the dual role of the catalyst in both activating the aldehyde and facilitating the oxidative step without external metal oxidants.
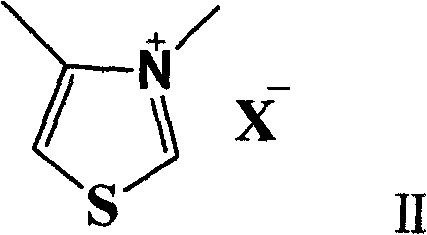
From an impurity control perspective, this organocatalytic mechanism offers distinct advantages over traditional radical or metal-mediated pathways. Since the reaction does not involve free radical chains that can lead to polymerization or non-selective oxidation side products, the impurity profile is generally cleaner and more predictable. The use of mild bases like potassium carbonate ensures that acid-sensitive protecting groups on the substrate remain intact, preserving the integrity of complex molecular architectures. Furthermore, the absence of transition metals means there is no risk of metal-induced decomposition or coloration of the product, which is a common issue in large-scale batch processing. The selectivity of the NHC catalyst towards the aldehyde functionality allows for high chemoselectivity even in the presence of other reactive groups such as nitro, cyano, or halo substituents, as evidenced by the broad substrate scope demonstrated in the patent examples. This robustness makes the process highly reliable for the synthesis of high-purity OLED material precursors and other specialty chemicals where trace impurities can be detrimental.
How to Synthesize Aromatic Carboxylic Acid Esters Efficiently
Implementing this synthesis route in a laboratory or pilot plant setting requires careful attention to reagent quality and atmospheric conditions, although the tolerance for oxygen is a key feature. The process begins with the preparation of a dry, oxygen-saturated acetonitrile solvent system, which serves as both the reaction medium and the source of the oxidant. The precise addition of the phase transfer catalyst, 18-crown-6, is critical for solubilizing the inorganic base and enhancing the nucleophilicity of the carbene species. While the general procedure is straightforward, scaling up requires monitoring the exotherm during the initial mixing of the base and the thiazolium salt to ensure thermal stability. Detailed standardized synthetic steps see the guide below for specific molar ratios and timing optimizations that have been proven to maximize yield across diverse substrate classes.
- Prepare the reaction system by mixing the aldehyde substrate, thiazole salt precursor, base (potassium carbonate), and phase transfer catalyst (18-crown-6) in dry acetonitrile under an oxygen atmosphere.
- Slowly add the halogenated hydrocarbon reactant to the mixture while maintaining the temperature between 30°C and 40°C to ensure controlled reaction kinetics.
- Stir the reaction for 20 to 30 hours, then remove the solvent via rotary evaporation and purify the crude product using column chromatography to obtain the high-purity ester.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this NHC-catalyzed technology translates into tangible strategic benefits that extend beyond simple reaction yields. The primary driver for cost reduction in ester manufacturing lies in the substitution of expensive noble metal catalysts with inexpensive, commercially available organic salts. Thiazolium salts are easy to synthesize in bulk and do not suffer from the price volatility associated with platinum group metals. Moreover, the elimination of metal scavenging steps reduces the consumption of auxiliary materials and shortens the overall production cycle time, leading to significant operational expenditure savings. The ability to run the reaction under aerobic conditions removes the capital expense of maintaining large-scale inert gas systems and reduces the risk of production stoppages due to nitrogen supply interruptions. These factors combined create a more resilient and cost-effective supply chain for critical intermediates.
- Cost Reduction in Manufacturing: The economic impact of switching to this organocatalytic method is profound, primarily driven by the removal of precious metal catalysts and the associated purification costs. Traditional methods often require palladium or rhodium complexes that can account for a substantial portion of the raw material bill, along with the additional cost of activated carbon or silica treatments to remove metal residues to ppm levels. By utilizing a metal-free system, manufacturers can eliminate these line items entirely. Additionally, the mild reaction temperatures (30°C to 40°C) significantly lower energy consumption compared to high-temperature reflux processes, contributing to reduced utility costs. The simplified workup procedure, which often involves simple solvent evaporation and chromatography, further reduces labor and solvent usage, resulting in substantial cost savings without compromising product quality.
- Enhanced Supply Chain Reliability: Supply chain continuity is greatly improved by the reliance on commodity chemicals rather than specialized reagents. The starting materials, aromatic aldehydes and benzyl halides, are produced on a massive global scale for various industries, ensuring stable availability and competitive pricing. Unlike custom-synthesized metal ligands which may have long lead times and single-source risks, the catalyst precursors for this reaction are robust and shelf-stable. The tolerance for oxygen and moisture variations also makes the process more forgiving in terms of logistics and storage, reducing the likelihood of batch failures due to minor deviations in raw material specifications. This robustness ensures a steady flow of high-purity pharmaceutical intermediates to downstream customers, minimizing the risk of stockouts.
- Scalability and Environmental Compliance: Scaling this process from gram to tonnage levels is facilitated by the inherent safety of the reaction conditions. The absence of high pressure and the use of non-toxic organic catalysts align perfectly with modern green chemistry principles and increasingly stringent environmental regulations. Waste treatment is simplified as the effluent does not contain heavy metals that require specialized hazardous waste disposal protocols. The atom economy of the reaction is favorable, and the use of acetonitrile, a common solvent with established recovery infrastructure, further supports sustainable manufacturing practices. This environmental compatibility not only reduces regulatory compliance costs but also enhances the corporate sustainability profile, which is increasingly important for securing contracts with major multinational corporations committed to green supply chains.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this NHC-catalyzed esterification technology. These answers are derived directly from the experimental data and claims presented in the patent documentation, providing a factual basis for decision-making. Understanding these nuances is essential for R&D teams evaluating the feasibility of adopting this route for their specific pipeline compounds. The clarity provided here aims to bridge the gap between academic innovation and industrial application, ensuring that stakeholders have a comprehensive understanding of the process capabilities and limitations.
Q: What are the primary advantages of this NHC catalyzed method over traditional metal-catalyzed esterification?
A: The primary advantage is the elimination of expensive and toxic transition metal catalysts. This method operates under mild, aerobic conditions at low temperatures (30-40°C), avoiding the need for rigorous anhydrous or oxygen-free environments required by conventional methods, thereby simplifying process safety and waste treatment.
Q: Is this process suitable for large-scale commercial production of pharmaceutical intermediates?
A: Yes, the process is highly scalable. It utilizes cheap, commercially available raw materials like aromatic aldehydes and benzyl halides. The mild reaction conditions reduce energy consumption and equipment corrosion risks, making it ideal for commercial scale-up of complex pharmaceutical intermediates.
Q: How does this technology impact the purity profile of the final ester product?
A: By avoiding heavy metal catalysts, the risk of metal residue contamination is completely removed. This results in a cleaner impurity profile, reducing the burden on downstream purification steps and ensuring the final product meets stringent quality specifications required for pharmaceutical applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Aromatic Carboxylic Acid Esters Supplier
As the global demand for high-quality pharmaceutical intermediates continues to rise, partnering with a manufacturer that possesses deep technical expertise in advanced catalytic methodologies is essential. NINGBO INNO PHARMCHEM stands ready to leverage this cutting-edge NHC-catalyzed technology to deliver superior aromatic carboxylic acid esters tailored to your specific project needs. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial manufacturing is seamless and efficient. We maintain stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instruments to guarantee that every batch meets the highest industry standards for identity, potency, and impurity profiles.
We invite you to engage with our technical procurement team to discuss how this innovative synthesis route can optimize your supply chain and reduce your overall manufacturing costs. By requesting a Customized Cost-Saving Analysis, you can gain detailed insights into the potential economic benefits specific to your target molecules. We encourage you to contact us today to索取 specific COA data and route feasibility assessments for your upcoming projects. Let us collaborate to bring your next generation of therapeutics to market faster and more economically through the power of advanced organocatalysis.
