Advanced Synthesis of N-Acetyl Tellurate Carbamates for Pharmaceutical Applications
The landscape of organotellurium chemistry has evolved significantly over the past three decades, driven by the unique reactivity and potential biological activity of tellurium-containing scaffolds in medicinal chemistry. Patent CN113912526B introduces a groundbreaking preparation method for N-acetyl tellurate carbamate compounds, addressing long-standing synthetic challenges in this niche field. This innovation utilizes readily available isonitriles and symmetrical ditellurides as starting materials, catalyzed by inexpensive acetate salts, to efficiently construct the N-acetyl tellurate carbamate backbone. For R&D directors and procurement specialists seeking reliable pharmaceutical intermediate suppliers, this technology represents a paradigm shift from hazardous, multi-step legacy processes to a streamlined, scalable, and economically viable manufacturing route suitable for complex drug discovery programs.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of tellurium carbamate derivatives has been plagued by severe operational hazards and restrictive reaction conditions that hinder commercial scalability. The seminal work by the Sonoda research group in 1993, for instance, relied on the reaction of secondary amines with butyllithium and tellurium powder followed by carbon monoxide insertion at cryogenic temperatures.
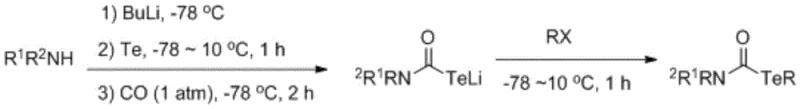
This conventional pathway necessitates the handling of highly toxic carbon monoxide gas under pressure and the use of pyrophoric butyllithium reagents at extremely low temperatures of -78 °C, creating significant safety risks and imposing heavy demands on specialized cryogenic equipment. Furthermore, subsequent variations involving butyl tellurium lithium and carbamoyl halides were limited to simple methyl and ethyl substrates, failing to accommodate the structural complexity required for modern API intermediates. Other reported methods utilizing strong bases like NaH or reducing agents like NaBH4 often suffer from cumbersome multi-step procedures and poor substrate scope, making them unsuitable for cost reduction in pharmaceutical intermediate manufacturing.
The Novel Approach
In stark contrast, the methodology disclosed in CN113912526B offers a remarkably mild and operationally simple alternative that bypasses the need for cryogenics and toxic gases. The core innovation involves the direct reaction of isonitriles with symmetrical ditellurides in the presence of cheap acetate additives, such as manganese acetate or copper acetate, under standard air atmosphere conditions.

This novel approach allows the reaction to proceed at moderate heating temperatures ranging from 50 °C to 150 °C, eliminating the energy-intensive cooling requirements of legacy methods. By leveraging the nucleophilic attack of the ditelluride species on the isonitrile carbon facilitated by the acetate catalyst, the process efficiently installs the N-acetyl tellurate moiety in a single pot. This transformation not only simplifies the workflow but also dramatically expands the accessible chemical space, enabling the synthesis of complex aryl, heterocyclic, and alkyl-substituted derivatives that were previously difficult or impossible to access via traditional routes.
Mechanistic Insights into Acetate-Catalyzed Telluration
The mechanistic pathway of this transformation is believed to involve the activation of the tellurium-tellurium bond by the acetate salt, generating a reactive tellurium species capable of inserting into the carbon-nitrogen triple bond of the isonitrile. The acetate anion serves a dual role, acting both as a base to facilitate proton transfer and as the source of the acetyl group that ultimately becomes part of the N-acetyl carbamate structure. This intramolecular rearrangement ensures high atom economy and minimizes the formation of unwanted byproducts, which is critical for maintaining high purity specifications in pharmaceutical applications. The tolerance of the catalytic system to air and moisture suggests a robust mechanism that does not rely on highly sensitive organometallic intermediates, thereby reducing the risk of batch-to-batch variability caused by trace impurities.
From an impurity control perspective, the use of symmetrical ditellurides and simple isonitriles limits the potential for complex side reactions often seen with asymmetric reagents. The reaction conditions promote the selective formation of the desired N-acetyl tellurate carbamate while suppressing the formation of tellurium oxides or other degradation products common in organotellurium chemistry. For example, the synthesis of tellurium-(4-methylphenyl)-N-acetyl-N-(2,6-dimethylphenyl) telluride carbamate proceeds with a yield of 74% under optimized conditions, demonstrating the efficiency of the catalytic cycle.
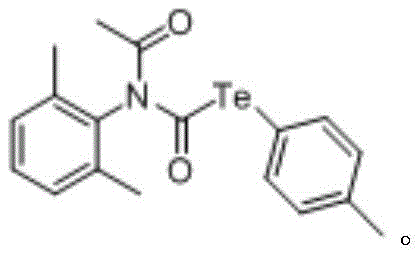
The ability to tolerate diverse functional groups, such as methoxy, chloro, and ester moieties, further underscores the versatility of this mechanism. This broad compatibility allows medicinal chemists to introduce the tellurium motif late-stage in the synthesis of complex drug candidates without protecting group strategies, significantly accelerating lead optimization timelines.
How to Synthesize N-Acetyl Tellurate Carbamates Efficiently
The practical implementation of this synthesis route is designed for ease of execution in standard laboratory and pilot plant settings. The process begins by charging a reaction vessel with the isonitrile substrate, the symmetrical ditelluride, and the acetate catalyst in a molar ratio of approximately 1:(1.0-3.0):(1.0-4.0). A polar aprotic solvent such as acetonitrile, DMF, or DMSO is added, and the mixture is heated to between 60 °C and 100 °C for a duration of 2 to 12 hours. Upon completion, indicated by the disappearance of the starting isonitrile, the reaction mixture is cooled, filtered, and subjected to a standard aqueous workup involving extraction with dichloromethane and washing with brine. The detailed standardized synthesis steps are provided in the guide below.
- Combine isonitrile, symmetrical ditelluride, and an acetate salt (such as manganese acetate) in a reaction vessel under air atmosphere with a suitable solvent.
- Heat the reaction mixture to a temperature range of 50-150 °C and maintain stirring for 1 to 24 hours until the starting isonitrile is consumed.
- Cool the mixture to room temperature, filter through celite, extract with dichloromethane, wash with brine, dry, and purify via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this acetate-catalyzed protocol offers substantial strategic benefits regarding cost structure and supply continuity. The elimination of hazardous reagents like carbon monoxide and butyllithium removes the need for specialized containment infrastructure and expensive safety protocols, leading to significant overhead reductions. Additionally, the reliance on commodity chemicals such as acetates and commercially available isonitriles ensures a stable and resilient supply chain, mitigating the risks associated with sourcing exotic or highly regulated precursors.
- Cost Reduction in Manufacturing: The replacement of cryogenic operations with moderate heating drastically lowers energy consumption and capital expenditure on refrigeration equipment. Furthermore, the use of inexpensive manganese or copper acetates instead of stoichiometric amounts of organolithium reagents results in substantial raw material cost savings. The simplified one-pot nature of the reaction reduces labor hours and solvent usage, contributing to a leaner overall manufacturing cost profile for high-purity pharmaceutical intermediates.
- Enhanced Supply Chain Reliability: By avoiding reagents that require strict inert atmosphere handling or have short shelf-lives, the process becomes more robust against logistical disruptions. The starting materials, including symmetrical ditellurides and various isonitriles, are either commercially available or can be synthesized via established, scalable routes, ensuring consistent availability for commercial scale-up of complex pharmaceutical intermediates. This reliability is crucial for maintaining uninterrupted production schedules in a GMP environment.
- Scalability and Environmental Compliance: The mild reaction conditions and absence of toxic gas inputs simplify the scale-up process from gram to ton quantities. The reduced hazard profile aligns well with increasingly stringent environmental, health, and safety (EHS) regulations, minimizing waste treatment costs and regulatory burdens. The high yields and selectivity observed in the patent examples suggest that the process can be scaled with minimal loss of efficiency, supporting the rapid deployment of new tellurium-based therapeutics.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel synthesis technology. These insights are derived directly from the experimental data and comparative analysis presented in the patent documentation, providing clarity for technical teams evaluating this route for adoption.
Q: What are the primary advantages of this acetate-catalyzed method over traditional Sonoda protocols?
A: Unlike the traditional Sonoda methods which require cryogenic conditions (-78 °C), toxic carbon monoxide gas, and pyrophoric butyllithium reagents, this novel protocol operates under mild heating (50-150 °C) in air atmosphere using inexpensive acetate salts, significantly improving safety and operational simplicity.
Q: Is the reaction sensitive to moisture or oxygen?
A: No, the process is remarkably robust and is explicitly designed to be conducted under air atmosphere, eliminating the need for rigorous inert gas protection or anhydrous conditions typically required for organometallic tellurium chemistry.
Q: What types of substrates are compatible with this synthesis route?
A: The method exhibits excellent functional group tolerance, successfully accommodating various isonitriles including aryl, heterocyclic, and alkyl variants, as well as symmetrical ditellurides, allowing for the introduction of the N-acetyl tellurate motif into complex molecular frameworks.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-Acetyl Tellurate Carbamate Supplier
As the demand for specialized organotellurium compounds grows in the pharmaceutical sector, partnering with an experienced CDMO is essential for successful commercialization. NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from lab-scale discovery to industrial manufacturing is seamless. Our facility is equipped with state-of-the-art rigorous QC labs capable of meeting stringent purity specifications required for clinical and commercial API intermediates, guaranteeing the quality and consistency of every batch produced.
We invite you to collaborate with our technical procurement team to explore how this advanced acetate-catalyzed technology can optimize your supply chain. Contact us today to request a Customized Cost-Saving Analysis tailored to your specific project needs. Our experts are ready to provide specific COA data and comprehensive route feasibility assessments to help you accelerate your development timeline and achieve your commercial goals efficiently.
