Advanced Homocamptothecin Synthesis: Enhancing Stability and Commercial Scalability for Global Pharma
The pharmaceutical industry continuously seeks robust synthetic pathways for potent antitumor agents, particularly those targeting DNA topoisomerase I. A pivotal advancement in this domain is detailed in patent CN100408582C, which discloses a novel class of homocamptothecin compounds exhibiting superior metabolic stability compared to their natural counterparts. Unlike traditional camptothecin derivatives that suffer from rapid lactone ring hydrolysis in physiological conditions, these new analogues feature a seven-membered beta-hydroxylactone ring that resists degradation, thereby maintaining therapeutic efficacy. This technological breakthrough addresses a critical limitation in oncology drug development, offering a stable scaffold for next-generation anticancer therapeutics. For global procurement teams and R&D directors, understanding the underlying chemistry of this patent is essential for securing a reliable homocamptothecin supplier capable of delivering high-purity intermediates. The innovation lies not only in the molecular structure but also in the practical, scalable preparation method that circumvents the economic and technical bottlenecks of prior art.
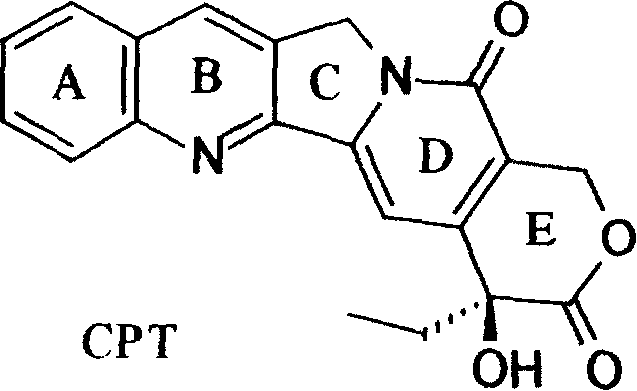
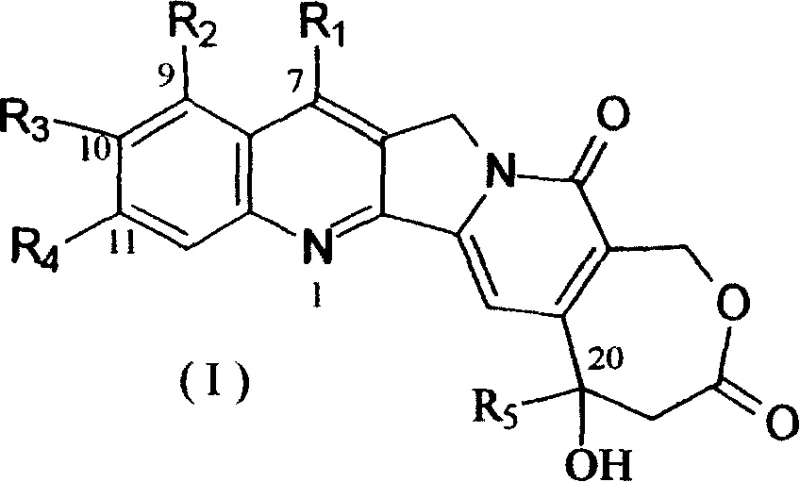
Natural camptothecin, while possessing significant antiproliferative activity, is fundamentally limited by the instability of its six-membered alpha-hydroxylactone E-ring. In the human body, this ring easily hydrolyzes into an inactive carboxylate form, drastically reducing bioavailability and therapeutic potential. To overcome this, researchers modified the core structure to create homocamptothecin (hCPT) derivatives, as illustrated in the general formula below. This structural modification expands the lactone ring from six members to seven, creating a beta-hydroxylactone system that is kinetically more stable against hydrolysis. The patent specifies various substituents at positions R1 through R5, allowing for fine-tuning of solubility, potency, and pharmacokinetic properties. This flexibility is crucial for developing a diverse portfolio of high-purity antitumor intermediates that can meet the stringent requirements of modern drug discovery pipelines.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to this invention, the total synthesis of homocamptothecin compounds was fraught with significant challenges that hindered commercial viability. Established routes, such as those described by Lavergne and Curran, relied heavily on the use of expensive and difficult-to-source reagents like palladium acetate and organotin compounds (e.g., Me3SnSnMe3). These transition metal catalysts not only inflate raw material costs but also introduce complex purification burdens, as removing trace heavy metals to meet pharmaceutical standards requires additional, costly processing steps. Furthermore, the overall chemical efficiency of these legacy methods was disappointingly low, with total yields often falling below 5%. Such poor atom economy and high waste generation make these processes unsustainable for large-scale manufacturing, creating supply chain vulnerabilities for companies seeking cost reduction in pharmaceutical intermediate manufacturing. The reliance on sensitive reagents also complicates process safety and operational control in a production environment.
The Novel Approach
The methodology presented in CN100408582C represents a paradigm shift towards efficiency and accessibility. By redesigning the synthetic strategy, the inventors eliminated the dependency on precious metal catalysts, opting instead for more abundant and cost-effective reagents such as zinc powder and standard alpha-haloesters. This strategic substitution drastically simplifies the reaction workflow and reduces the environmental footprint associated with heavy metal waste disposal. The new route achieves a marked improvement in chemical yield, with the total yield of the final homocamptothecin compound exceeding 12%, a substantial increase over the sub-5% yields of previous methods. This enhancement in efficiency translates directly into better resource utilization and lower production costs, making the commercial scale-up of complex camptothecin analogues a realistic and economically attractive proposition for industrial partners. The robustness of the reaction conditions further ensures consistent quality across different batch sizes.
Mechanistic Insights into Zinc-Mediated Reformatsky Cyclization
The core of this innovative synthesis lies in the strategic construction of the seven-membered lactone ring via a modified Reformatsky reaction. The process begins with the reduction of a lactone carbonyl group using mild reducing agents like potassium borohydride or sodium borohydride, followed by oxidative cleavage with periodates to generate a key formyl intermediate. This aldehyde functionality then serves as the electrophile in a zinc-mediated coupling with an alpha-haloester. The use of zinc is particularly advantageous as it is less oxophilic than magnesium, allowing for better tolerance of functional groups present in the complex indolizine scaffold. The resulting beta-hydroxy ester intermediate undergoes acid-mediated hydrolysis and intramolecular cyclization to close the seven-membered ring. This sequence is meticulously optimized to minimize side reactions and epimerization at the chiral center, ensuring the formation of the desired stereochemistry which is critical for biological activity. Understanding this mechanism allows R&D teams to appreciate the precision required in controlling reaction parameters such as temperature and pH during scale-up.
Impurity control is another critical aspect where this novel pathway excels, particularly concerning the stability of the lactone ring and the purity of the final API intermediate. Traditional methods often struggle with the formation of open-ring carboxylate impurities due to the harsh conditions required for cyclization. In contrast, the new method employs specific organic acids like p-toluenesulfonic acid in non-polar solvents such as toluene to drive the condensation reaction under controlled dehydration conditions. This approach effectively suppresses the hydrolysis of the sensitive lactone moiety during synthesis, leading to a cleaner crude product profile. Additionally, the avoidance of tin and palladium residues eliminates a major class of toxic impurities that require extensive chromatographic purification. The result is a process that inherently supports the production of high-purity OLED material grade or pharmaceutical grade intermediates, reducing the burden on downstream purification units and accelerating the time to market for new drug candidates.
How to Synthesize Homocamptothecin Efficiently
The synthesis of these advanced antitumor intermediates requires a precise sequence of chemical transformations that balance reactivity with selectivity. The process initiates with the preparation of a protected indolizine precursor, which is subsequently subjected to reduction and oxidation to install the necessary aldehyde handle. Following this, the chain extension is achieved through the Reformatsky reaction, a cornerstone step that builds the carbon framework for the expanded lactone ring. The final stages involve acidic cyclization and condensation with substituted anilines to complete the pentacyclic core. Each step is optimized for yield and purity, ensuring that the final product meets the rigorous standards expected of a reliable agrochemical intermediate supplier or pharmaceutical partner. Detailed standard operating procedures for these transformations are essential for maintaining consistency across production batches.
- Reduction and Oxidation: Convert the starting lactone precursor into a formyl intermediate using borohydride reduction followed by periodate oxidation.
- Reformatsky Reaction: React the formyl intermediate with an alpha-haloester in the presence of zinc to extend the carbon chain and introduce the ester functionality.
- Cyclization and Coupling: Perform acid-mediated hydrolysis and cyclization to form the seven-membered lactone ring, followed by condensation with substituted o-amino aromatic ketones.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route offers tangible strategic benefits beyond mere chemical elegance. The primary advantage lies in the significant cost savings driven by the elimination of precious metal catalysts. By replacing palladium and tin reagents with zinc and common organic acids, the raw material cost structure is fundamentally improved, allowing for more competitive pricing models without sacrificing quality. This shift also mitigates the risk associated with the supply volatility of rare earth and precious metals, ensuring greater supply chain reliability for long-term contracts. Furthermore, the simplified purification process reduces solvent consumption and waste treatment costs, contributing to a more sustainable and economically efficient manufacturing operation. These factors collectively enhance the overall value proposition for partners seeking cost reduction in electronic chemical manufacturing or pharmaceutical sectors.
- Cost Reduction in Manufacturing: The replacement of expensive transition metal catalysts with abundant zinc powder removes the need for costly metal scavenging steps and reduces the overall bill of materials. This qualitative shift in reagent selection leads to substantial cost savings in the production budget, allowing for more flexible pricing strategies in a competitive market. Additionally, the higher overall yield means less starting material is wasted per kilogram of final product, further driving down the unit cost of goods sold. The process efficiency also translates to reduced energy consumption and shorter cycle times, optimizing the utilization of manufacturing assets.
- Enhanced Supply Chain Reliability: By relying on commodity chemicals rather than specialized organometallic reagents, the risk of supply disruption is significantly minimized. Zinc and standard haloesters are widely available from multiple global vendors, ensuring that production schedules are not held hostage by the availability of a single niche supplier. This diversification of the supply base enhances the resilience of the manufacturing pipeline, guaranteeing consistent delivery timelines for clients. The robustness of the chemistry also means that technology transfer between sites is smoother, facilitating regional production capabilities to reduce lead time for high-purity pharmaceutical intermediates.
- Scalability and Environmental Compliance: The synthetic route is designed with inherent scalability, utilizing reaction conditions that are safe and manageable in large-scale reactors. The absence of toxic tin byproducts simplifies waste stream management and ensures easier compliance with increasingly stringent environmental regulations. This eco-friendly profile aligns with the sustainability goals of modern multinational corporations, making the manufacturing process more attractive for green chemistry initiatives. The ability to scale from laboratory to commercial production without fundamental changes to the chemistry ensures a seamless transition from clinical trial material to market supply.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these homocamptothecin derivatives. They are derived from the specific technical advantages and structural features detailed in the patent documentation. Understanding these aspects helps stakeholders make informed decisions regarding sourcing and development partnerships. The answers reflect the consensus on the stability, efficiency, and applicability of the described technology in real-world scenarios.
Q: How does the seven-membered lactone ring improve drug stability compared to natural camptothecin?
A: Natural camptothecin contains a six-membered alpha-hydroxylactone ring that is prone to hydrolysis in vivo, converting into an inactive carboxylate form. The homocamptothecin structure features a seven-membered beta-hydroxylactone ring, which significantly enhances metabolic stability and maintains antitumor activity for longer durations within the biological system.
Q: What are the primary cost advantages of this new synthetic route over conventional methods?
A: Conventional total synthesis routes often rely on expensive and scarce reagents such as palladium acetate and organotin compounds, resulting in low overall yields below 5%. The novel method utilizes more accessible reagents like zinc powder and standard haloesters, eliminating the need for precious metal catalysts and complex removal steps, thereby drastically simplifying the purification process and reducing raw material costs.
Q: Is this synthesis method suitable for large-scale commercial production?
A: Yes, the process is designed with scalability in mind. By avoiding sensitive transition metal catalysts and utilizing robust reactions like the Reformatsky reaction and acid-mediated cyclization, the method offers better control over exotherms and impurity profiles. This makes it highly adaptable for commercial scale-up of complex pharmaceutical intermediates while ensuring consistent quality and supply continuity.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Homocamptothecin Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the homocamptothecin platform in modern oncology therapy. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from benchtop discovery to full-scale market supply. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of intermediate meets the highest international standards. We understand the critical nature of supply continuity in the pharmaceutical industry and have built our operations to prioritize reliability and quality above all else.
We invite you to collaborate with us to leverage this advanced synthetic technology for your next-generation drug candidates. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. Please contact us to request specific COA data and route feasibility assessments for your projects. By partnering with us, you gain access to a supply chain that is not only cost-effective but also technically robust, ensuring that your development timelines are met with precision and confidence.
