Scalable Synthesis of tert-Butyl 3-Oxazetidine-1-Carboxylate for Commercial API Production
Scalable Synthesis of tert-Butyl 3-Oxazetidine-1-Carboxylate for Commercial API Production
The pharmaceutical industry continuously seeks robust and economically viable synthetic routes for critical heterocyclic intermediates, particularly those serving as key building blocks for high-value active pharmaceutical ingredients (APIs). Patent CN111362853A introduces a groundbreaking preparation method for tert-butyl 3-oxazetidine-1-carboxylate, a vital scaffold utilized in the synthesis of prominent drugs such as Baricitinib for rheumatoid arthritis, Tebipenem pivoxil as a broad-spectrum antibacterial agent, and Cobimetinib for antitumor applications. This technical disclosure represents a significant leap forward in process chemistry, addressing long-standing challenges related to cost, safety, and scalability that have plagued previous manufacturing methodologies. By shifting away from complex multi-step sequences involving precious metal catalysts, this innovation offers a streamlined pathway that aligns perfectly with the rigorous demands of modern GMP production environments.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 3-oxazetidine derivatives has been fraught with significant operational hurdles and economic inefficiencies that hinder large-scale adoption. One prevalent conventional strategy involves the ring-opening of epichlorohydrin with benzylamine or benzhydrylamine, followed by base-mediated cyclization to form the azetidine skeleton. However, this route necessitates a subsequent hydrogenolysis step using palladium on carbon (Pd/C) catalysts to remove the protecting groups. This reliance on heterogeneous hydrogenation not only imposes stringent safety requirements due to the handling of hydrogen gas but also introduces substantial costs associated with the procurement and recovery of expensive palladium catalysts. Furthermore, alternative routes utilizing imine intermediates derived from epoxy compounds and aldehydes often suffer from poor purification profiles, where the separation of closely related byproducts becomes technically demanding and yield-limiting.
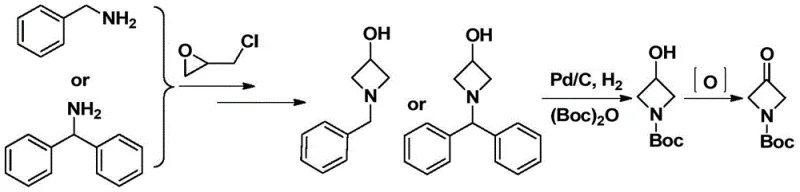
The Novel Approach
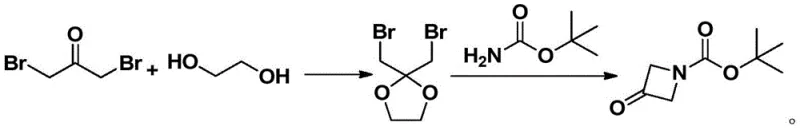
In stark contrast to these legacy methods, the methodology disclosed in CN111362853A presents a remarkably efficient two-step sequence that circumvents the need for hydrogenation entirely. The process initiates with the acid-catalyzed condensation of 1,3-dibromoacetone and ethylene glycol to form a stable ketal intermediate, 2,2-bis(bromomethyl)-1,3-dioxolane. This intermediate then undergoes a direct cyclization with tert-butyl carbamate under basic conditions to yield the target oxazetidine ring system. This strategic redesign eliminates the dependency on transition metal catalysts and simplifies the downstream processing significantly. The use of inexpensive commodity chemicals as starting materials, combined with mild reaction parameters, ensures that the process is not only chemically elegant but also commercially superior, offering a clear path toward cost reduction in pharmaceutical intermediate manufacturing.
Mechanistic Insights into Acid-Catalyzed Ketalization and Base-Mediated Cyclization
The chemical elegance of this new route lies in its precise control over reactivity and selectivity through well-understood mechanistic principles. The first step leverages a Brønsted acid catalyst, such as p-toluenesulfonic acid or sulfuric acid, to activate the carbonyl group of 1,3-dibromoacetone towards nucleophilic attack by ethylene glycol. This ketalization serves a dual purpose: it protects the ketone functionality from unwanted side reactions and simultaneously positions the bromomethyl groups for the subsequent intramolecular displacement. The reaction is typically conducted in aromatic solvents like toluene under reflux, utilizing a Dean-Stark trap to azeotropically remove water, thereby driving the equilibrium towards the formation of the 2,2-bis(bromomethyl)-1,3-dioxolane intermediate with high conversion efficiency.
Following the isolation or direct use of the ketal intermediate, the second step involves a nucleophilic substitution cascade mediated by a base. The nitrogen atom of tert-butyl carbamate acts as the nucleophile, displacing one of the bromide ions to form an open-chain amino-ketal species, which subsequently undergoes intramolecular cyclization to close the four-membered oxazetidine ring. The choice of base is critical; mild inorganic bases like sodium carbonate or potassium carbonate are sufficient to deprotonate the carbamate without promoting elimination side reactions that could degrade the sensitive bromomethyl groups. This controlled basic environment ensures that the cyclization proceeds cleanly to form the desired N-Boc protected oxazetidine, minimizing the formation of polymeric byproducts and facilitating straightforward purification via recrystallization.
How to Synthesize tert-Butyl 3-Oxazetidine-1-Carboxylate Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires careful attention to stoichiometry and thermal management to maximize yield and purity. The process begins by dissolving 1,3-dibromoacetone in toluene and adding ethylene glycol along with a catalytic amount of p-toluenesulfonic acid. The mixture is heated to reflux, typically around 110°C, with continuous water removal until TLC analysis indicates complete consumption of the starting ketone. Upon cooling, the crude ketal intermediate can be extracted and used directly in the next step without further purification, which is a significant advantage for throughput. For the cyclization, the crude intermediate is dissolved in 1,4-dioxane, and tert-butyl carbamate is added along with sodium carbonate. The reaction is maintained at a moderate temperature, such as 50°C to 70°C, for approximately 16 hours to ensure complete ring closure. The detailed standardized synthesis steps are provided below.
- React 1,3-dibromoacetone with ethylene glycol using an acid catalyst like p-toluenesulfonic acid in toluene under reflux to form 2,2-bis(bromomethyl)-1,3-dioxolane.
- Cyclize the intermediate with tert-butyl carbamate using a base such as sodium carbonate in 1,4-dioxane at moderate temperatures.
- Purify the final crude product via recrystallization using petroleum ether to obtain high-purity tert-butyl 3-oxazetidine-1-carboxylate.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition to this novel synthetic route offers compelling strategic benefits that extend far beyond simple chemical curiosity. The elimination of palladium catalysts removes a major variable cost driver and mitigates the risk of supply chain disruptions associated with precious metal availability. Additionally, the avoidance of high-pressure hydrogenation equipment reduces capital expenditure requirements for manufacturing facilities, allowing for production in standard glass-lined or stainless steel reactors that are more readily available. The simplicity of the workup procedures, which rely on standard extraction and recrystallization techniques rather than complex chromatography or distillation, translates directly into reduced processing times and lower utility consumption per kilogram of product produced.
- Cost Reduction in Manufacturing: The economic impact of this process is profound due to the substitution of expensive reagents with commodity chemicals. By removing the need for palladium on carbon and hydrogen gas, manufacturers can achieve substantial cost savings on raw material procurement and waste disposal. The high yields observed in the exemplary data, reaching up to 92% in the final cyclization step, further enhance the overall material efficiency, ensuring that less starting material is wasted. This efficiency gain allows for a more competitive pricing structure for the final API, providing a distinct market advantage in price-sensitive therapeutic areas.
- Enhanced Supply Chain Reliability: The reliance on 1,3-dibromoacetone and ethylene glycol as primary feedstocks ensures a robust and stable supply chain, as these are widely produced industrial chemicals with multiple global suppliers. Unlike specialized chiral amines or complex epoxy derivatives that may have limited sources, these bulk chemicals are less susceptible to shortage-induced price volatility. Furthermore, the mild reaction conditions reduce the likelihood of batch failures due to equipment malfunction or operator error, thereby improving the consistency of supply and reducing lead times for high-purity pharmaceutical intermediates required by downstream customers.
- Scalability and Environmental Compliance: From an environmental and regulatory perspective, this route is inherently greener and easier to scale. The absence of heavy metal residues simplifies the regulatory filing process, as there is no need for extensive validation of metal clearance steps which can be a bottleneck in API registration. The process generates fewer hazardous byproducts compared to oxidative or hydrogenolytic methods, aligning with increasingly stringent environmental regulations regarding solvent use and waste generation. This compliance ease facilitates faster technology transfer from R&D to commercial scale-up of complex heterocycles, enabling quicker time-to-market for new drug formulations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis method. These answers are derived directly from the experimental data and technical specifications outlined in the patent documentation to provide clarity for potential partners and licensees. Understanding these nuances is essential for evaluating the feasibility of adopting this technology within existing manufacturing frameworks.
Q: Why is this new synthesis route superior to traditional methods for tert-butyl 3-oxazetidine-1-carboxylate?
A: Traditional methods often rely on expensive palladium/carbon catalytic hydrogenation for deprotection or involve difficult-to-purify imine intermediates. This novel route eliminates the need for precious metal catalysts and utilizes cheap, readily available raw materials like 1,3-dibromoacetone, significantly simplifying the purification process and reducing equipment requirements.
Q: What are the key reaction conditions for the cyclization step?
A: The cyclization step involves reacting 2,2-bis(bromomethyl)-1,3-dioxolane with tert-butyl carbamate in the presence of a base. Optimal conditions identified include using sodium carbonate as the base in solvents like 1,4-dioxane or toluene at temperatures ranging from 50°C to 70°C, ensuring high conversion rates without harsh conditions.
Q: Is this process suitable for large-scale industrial manufacturing?
A: Yes, the process is specifically designed for industrial scalability. It operates under mild reaction conditions, uses common organic solvents, and avoids hazardous hydrogenation steps. The high yields reported in examples (up to 92% in the final step) and the simplicity of the workup procedure make it highly viable for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable tert-Butyl 3-Oxazetidine-1-Carboxylate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the success of modern drug development programs. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the volumetric demands of global pharmaceutical clients without compromising on quality. We maintain stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee that every batch of tert-butyl 3-oxazetidine-1-carboxylate meets the highest industry standards. Our commitment to technical excellence allows us to navigate the complexities of process optimization effectively.
We invite you to collaborate with us to leverage this advanced synthetic technology for your upcoming projects. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. Please contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise can accelerate your supply chain and reduce your overall manufacturing costs.
