Advanced Palladium-Catalyzed Synthesis of 3-Arylquinoline-2(1H)one Derivatives for Commercial Scale-Up
Introduction to Novel Quinolin-2(1H)one Synthesis
The pharmaceutical and fine chemical industries are constantly seeking robust, scalable, and cost-effective routes to access privileged heterocyclic scaffolds. Patent CN113045489A discloses a groundbreaking preparation method for 3-arylquinoline-2(1H)one derivatives, a class of compounds renowned for their profound biological activities. These structures serve as critical cores in various therapeutic agents, functioning as antibiotics, antiplatelet drugs, antitumor agents, and receptor antagonists. The significance of this technology lies in its ability to utilize benzisoxazole simultaneously as a nitrogen source and a formyl source, streamlining the synthetic pathway significantly. By leveraging a palladium-catalyzed aminocarbonylation strategy, this method overcomes many limitations associated with classical heterocycle synthesis, offering a versatile platform for generating high-purity API intermediates.
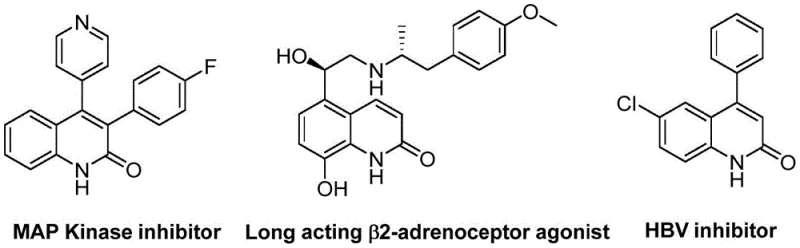
The structural diversity achievable through this method is exemplified by the broad range of biologically active molecules that share this core scaffold. As illustrated in the reference data, these derivatives are pivotal in developing MAP Kinase inhibitors, long-acting β2-adrenoceptor agonists, and HBV inhibitors. The ability to efficiently construct the quinolin-2(1H)one ring system with high regioselectivity and yield makes this patent a valuable asset for R&D teams aiming to expand their pipeline of potential drug candidates. Furthermore, the operational simplicity described in the patent suggests a high degree of translatability from laboratory discovery to commercial manufacturing, addressing a key pain point for supply chain stakeholders looking for reliable sources of complex heterocyclic building blocks.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of quinolin-2(1H)one derivatives has relied heavily on classical named reactions such as the Vilsmeier-Haack, Knorr, and Friedlander condensations. While these methods are well-established, they often suffer from significant drawbacks when applied to modern drug discovery and process chemistry. Traditional routes frequently require harsh reaction conditions, including strong acids or bases, elevated temperatures, and prolonged reaction times, which can lead to poor functional group tolerance and the formation of difficult-to-remove impurities. Moreover, many conventional strategies involve multi-step sequences to install the necessary carbonyl functionality, resulting in lower overall atom economy and increased waste generation. The reliance on volatile or toxic reagents in some older protocols also poses safety and environmental compliance challenges, complicating the scale-up process for industrial applications.
The Novel Approach
In stark contrast, the method disclosed in CN113045489A represents a paradigm shift towards greener and more efficient synthesis. By employing a transition metal-catalyzed carbonylation reaction, this novel approach utilizes benzisoxazole and benzyl chloride compounds as readily available starting materials. The key innovation is the dual role of benzisoxazole, which provides both the nitrogen atom for the ring closure and the carbonyl carbon, effectively simplifying the retrosynthetic analysis. The reaction proceeds under relatively mild conditions using a palladium catalyst system with (S)-BINAP as a chiral ligand and molybdenum hexacarbonyl as the CO source. This strategy not only reduces the number of synthetic steps but also enhances the safety profile by avoiding the direct handling of carbon monoxide gas. The result is a streamlined process that delivers high yields and exceptional purity, making it an ideal candidate for cost reduction in pharmaceutical intermediate manufacturing.
Mechanistic Insights into Pd-Catalyzed Aminocarbonylation
The core of this technological advancement lies in the sophisticated palladium-catalyzed aminocarbonylation mechanism. The reaction initiates with the oxidative addition of the benzyl chloride compound to the active palladium(0) species generated in situ from palladium acetate and the (S)-BINAP ligand. This step forms an organopalladium(II) intermediate, which is crucial for the subsequent insertion of the carbonyl group. Molybdenum hexacarbonyl serves as a solid surrogate for carbon monoxide, releasing CO under the thermal conditions of the reaction (100°C). The coordination and insertion of CO into the palladium-carbon bond generate an acyl-palladium complex. This electrophilic species is then poised for nucleophilic attack by the nitrogen atom derived from the benzisoxazole ring opening.
Following the nucleophilic attack, the intermediate undergoes a series of transformations including cyclization and elimination to restore aromaticity and form the final quinolin-2(1H)one structure. The presence of triethylamine as a base facilitates the deprotonation steps necessary for the catalytic cycle to turnover efficiently. Water is also included in the reaction mixture, playing a subtle yet vital role in hydrolyzing intermediates or stabilizing the catalytic species. The use of the chiral ligand (S)-BINAP, although the product is achiral in the examples shown, ensures high catalytic activity and stability of the palladium center, preventing premature catalyst decomposition. This mechanistic understanding allows chemists to fine-tune reaction parameters, such as temperature and stoichiometry, to maximize yield and minimize side reactions, ensuring a robust process suitable for the commercial scale-up of complex pharmaceutical intermediates.
How to Synthesize 3-Arylquinoline-2(1H)one Efficiently
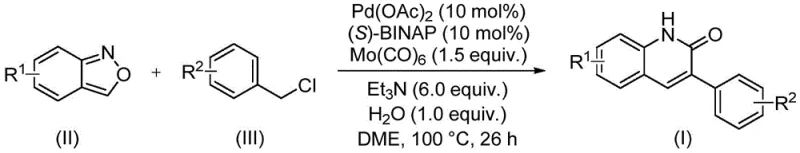
Implementing this synthesis requires precise control over reaction parameters to achieve the reported high efficiencies. The protocol involves charging a sealed vessel with the specific molar ratios of catalysts and reagents: typically 10 mol% Pd(OAc)2, 10 mol% (S)-BINAP, and 1.5 equivalents of Mo(CO)6 relative to the limiting substrate. The reaction is conducted in ethylene glycol dimethyl ether (DME) at 100°C for approximately 26 hours. This extended time ensures complete consumption of the starting materials, particularly for substrates with electron-withdrawing groups that might slow down the oxidative addition step. The detailed standardized synthesis steps, including specific workup procedures like filtration and silica gel treatment followed by column chromatography, are outlined below to guide process development teams.
- Charge a sealed tube with palladium acetate, (S)-BINAP, molybdenum hexacarbonyl, triethylamine, water, benzisoxazole, and the specific benzyl chloride compound in DME solvent.
- Heat the reaction mixture to 100°C and maintain stirring for 26 hours to ensure complete conversion via the aminocarbonylation pathway.
- Upon completion, filter the mixture, mix with silica gel, and purify the crude product using column chromatography to isolate the target 3-arylquinoline-2(1H)one derivative.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic route offers tangible strategic benefits beyond mere chemical elegance. The primary advantage stems from the availability and cost of raw materials. Benzisoxazole and substituted benzyl chlorides are commodity chemicals that are widely produced and easily sourced from the global market, reducing the risk of supply bottlenecks. Unlike specialized reagents that require custom synthesis, these starting materials benefit from established supply chains, ensuring consistent quality and competitive pricing. Furthermore, the elimination of hazardous gaseous carbon monoxide from the process inventory simplifies facility requirements, potentially lowering capital expenditure on safety infrastructure and reducing insurance costs associated with handling toxic gases.
- Cost Reduction in Manufacturing: The economic viability of this process is driven by its high atom economy and simplified operational workflow. By combining the nitrogen and carbonyl sources into a single efficient catalytic cycle, the method eliminates the need for multiple isolation and purification steps that are typical in classical syntheses. This reduction in unit operations translates directly to lower labor costs, reduced solvent consumption, and decreased energy usage per kilogram of product. Additionally, the high yields reported (often exceeding 90%) mean that less raw material is wasted, further driving down the cost of goods sold (COGS) for the final API intermediate.
- Enhanced Supply Chain Reliability: The robustness of the catalytic system contributes significantly to supply chain stability. The tolerance of the reaction to various functional groups means that a single set of reaction conditions can be applied to a wide library of derivatives without extensive re-optimization. This flexibility allows manufacturers to respond quickly to changing demand for different analogues without retooling production lines. Moreover, the use of stable, solid reagents like Mo(CO)6 instead of pressurized gas cylinders mitigates logistics risks, ensuring that production schedules are not disrupted by transportation regulations or storage limitations associated with hazardous gases.
- Scalability and Environmental Compliance: From an environmental perspective, this method aligns well with green chemistry principles. The use of DME as a solvent and the avoidance of stoichiometric amounts of toxic heavy metals or corrosive reagents simplify waste treatment protocols. The post-treatment procedure involving simple filtration and standard chromatography is easily adaptable to large-scale preparative HPLC or crystallization techniques. This scalability ensures that the transition from gram-scale R&D to ton-scale commercial production is seamless, maintaining high purity specifications while minimizing the environmental footprint of the manufacturing process.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of this synthesis is critical for stakeholders evaluating its potential for their specific projects. The following questions address common inquiries regarding the reaction scope, safety, and scalability. These answers are derived directly from the experimental data and technical disclosures within the patent, providing a factual basis for decision-making. Whether you are concerned about the compatibility of sensitive functional groups or the specifics of the catalyst loading, the information below clarifies the operational boundaries of this technology.
Q: What acts as the carbonyl source in this novel synthesis method?
A: Unlike traditional methods that may use toxic carbon monoxide gas directly, this process utilizes Molybdenum Hexacarbonyl (Mo(CO)6) as a safe and solid carbonyl source, which releases CO in situ under the reaction conditions.
Q: Does this method tolerate diverse functional groups on the substrate?
A: Yes, the patent data demonstrates excellent functional group tolerance. Substituents such as methoxy, chloro, cyano, trifluoromethyl, and tert-butyl groups on both the benzisoxazole and benzyl chloride rings are well-tolerated, yielding products with high purity.
Q: What are the typical yields achieved with this catalytic system?
A: The process exhibits high reaction efficiency. Experimental data in the patent shows isolated yields ranging significantly high, with specific examples achieving yields up to 97% for derivatives like 3-(4-cyanophenyl)quinolin-2(1H)one.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Arylquinoline-2(1H)one Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the success of drug development programs. Our team of expert chemists has thoroughly analyzed the technology disclosed in CN113045489A and is fully prepared to leverage this advanced palladium-catalyzed methodology for your projects. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with consistency and precision. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of 3-arylquinoline-2(1H)one derivative delivered meets the highest industry standards.
We invite you to collaborate with us to optimize this synthesis for your specific target molecules. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your volume requirements. We encourage you to reach out today to discuss your project needs,索取 specific COA data for our catalog compounds, or request route feasibility assessments for novel derivatives. Let us help you accelerate your timeline to market with our reliable supply of high-purity pharmaceutical intermediates.
