Revolutionizing Oxazole Synthesis: A Green Electrocatalytic Strategy for Commercial Pharmaceutical Intermediates
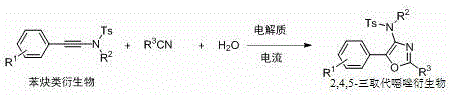
The pharmaceutical and fine chemical industries are constantly seeking more sustainable and efficient pathways to access privileged heterocyclic scaffolds, among which oxazole derivatives hold a paramount position due to their widespread biological activities. Patent CN115233243A, published in late 2022, introduces a groundbreaking preparation method for 2,4,5-trisubstituted oxazole derivatives utilizing an innovative electrocatalysis strategy. This technology represents a significant paradigm shift from traditional thermal catalysis, leveraging electrical energy to drive the cyclization of alkynamide derivatives with nitrile solvents in the presence of selenide mediators. The disclosed method not only addresses the growing demand for green chemistry principles but also offers a robust solution for the scalable production of high-value pharmaceutical intermediates used in treating neurological, infectious, and cardiovascular diseases. By replacing hazardous reagents and expensive catalysts with electricity and benign electrolytes, this invention paves the way for a more environmentally responsible manufacturing landscape.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of phenyloxazole and related trisubstituted oxazole compounds has relied heavily on transition metal catalysis, often involving palladium, copper, or other noble metals coupled with complex ligand systems. These conventional routes typically necessitate stringent reaction conditions, including high temperatures, strictly anhydrous environments, and inert atmospheres to prevent catalyst deactivation or side reactions. Furthermore, the reliance on stoichiometric or near-stoichiometric amounts of oxidants and the generation of heavy metal waste streams pose significant environmental and economic challenges. The downstream processing required to remove trace metal residues to meet pharmaceutical purity standards adds substantial complexity and cost to the manufacturing process. Additionally, many traditional methods suffer from limited substrate scope or poor atom economy, making them less attractive for large-scale industrial applications where efficiency and waste minimization are critical metrics for success.
The Novel Approach
In stark contrast to these legacy methods, the electrocatalytic protocol described in the patent offers a streamlined, metal-free alternative that operates under remarkably mild conditions. The core innovation lies in the use of constant current electrolysis to activate selenide species, which then mediate the cyclization of alkynamides with the nitrile solvent itself acting as a reactant. This approach eliminates the need for external oxidants and precious metal catalysts, drastically simplifying the reaction setup. The process proceeds efficiently at room temperature and, crucially, tolerates the presence of air and moisture, removing the burden of specialized equipment like gloveboxes or Schlenk lines. This operational simplicity translates directly into reduced capital expenditure and lower energy consumption, making it an highly attractive option for the cost reduction in pharmaceutical intermediate manufacturing.
Mechanistic Insights into Electrocatalytic Oxazole Cyclization
The mechanistic pathway of this electrocatalytic transformation is fascinating and distinct from thermal radical processes. It is postulated that the anodic oxidation of the selenide compound generates a reactive selenyl cation or radical species, which subsequently activates the alkyne moiety of the alkynamide substrate. This activation facilitates a nucleophilic attack by the nitrogen atom of the nitrile solvent, initiating the ring-closing sequence that forms the oxazole core. The use of electricity allows for precise control over the oxidation potential, minimizing over-oxidation side reactions that often plague chemical oxidant-based methods. This precise control contributes to the high selectivity observed across a broad range of substrates, including those with electron-donating or electron-withdrawing groups on the aromatic rings. The ability to tune the reaction outcome simply by adjusting the current density provides chemists with a powerful tool for optimizing yield and purity without altering the chemical composition of the reaction mixture.
From an impurity control perspective, the absence of transition metals inherently reduces the risk of metal-catalyzed decomposition pathways or metal-complex formation, which are common sources of difficult-to-remove impurities in API synthesis. The mild reaction temperature further suppresses thermal degradation of sensitive functional groups, ensuring that the final product profile is clean and易于 purification. The patent data indicates that the reaction can tolerate various substituents such as halogens, alkyl groups, and alkoxy groups without significant loss in efficiency, suggesting a robust tolerance for diverse chemical functionalities. This broad compatibility is essential for medicinal chemists who need to rapidly iterate on molecular structures during drug discovery phases, as it allows for the late-stage functionalization of complex molecules without compromising the integrity of the oxazole scaffold.
How to Synthesize 2,4,5-Trisubstituted Oxazoles Efficiently
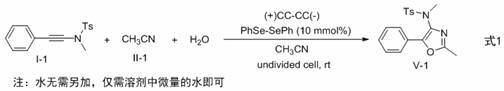
The practical implementation of this synthesis is designed for ease of execution, requiring standard electrochemical equipment rather than specialized high-pressure reactors. The procedure involves mixing the alkynamide precursor, a catalytic amount of diselenide, a supporting electrolyte like tetrabutylammonium tetrafluoroborate, and the nitrile solvent in a simple undivided cell. Carbon cloth electrodes are typically employed due to their high surface area and stability, although other electrode materials can also be effective depending on the specific substrate. The reaction is driven by applying a constant current, typically around 10 mA for small-scale preparations, and stirring is maintained at ambient temperature. Detailed standardized synthesis steps for replicating this high-efficiency route are provided in the guide below, ensuring reproducibility for process development teams looking to adopt this technology.
- Mix alkynamide derivatives, selenide compounds (e.g., diphenyl diselenide), electrolyte (NBu4BF4), and nitrile solvent (e.g., acetonitrile) in a reaction vessel equipped with carbon cloth electrodes.
- Apply a constant current (e.g., 10 mA) at room temperature under an air atmosphere for approximately 1 hour to drive the cyclization reaction.
- Upon completion, concentrate the mixture, extract with ethyl acetate, wash with brine, dry over sodium sulfate, and purify via silica gel column chromatography to isolate the target oxazole.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this electrocatalytic technology presents a compelling value proposition centered on cost efficiency and operational resilience. By eliminating the dependency on volatile and expensive precious metal markets, companies can stabilize their raw material costs and mitigate supply risks associated with geopolitical fluctuations in metal availability. The simplified operational requirements, such as the ability to run reactions in air without rigorous drying of solvents, significantly reduce the utility costs associated with HVAC and nitrogen generation in production facilities. Furthermore, the reduction in hazardous waste generation aligns with increasingly strict environmental regulations, potentially lowering waste disposal fees and improving the overall sustainability profile of the manufacturing site. These factors collectively contribute to a more agile and cost-effective supply chain capable of responding quickly to market demands.
- Cost Reduction in Manufacturing: The most immediate financial benefit stems from the complete removal of expensive transition metal catalysts like palladium or rhodium, which often constitute a significant portion of the bill of materials for complex heterocycle synthesis. Additionally, the elimination of specialized ligands and the reduced need for extensive purification steps to remove metal residues lead to substantial savings in both consumables and labor. The use of electricity as a traceless reagent further enhances the atom economy of the process, ensuring that a higher percentage of input materials are converted into valuable product rather than waste. This holistic reduction in input costs allows for a more competitive pricing structure for the final pharmaceutical intermediates.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions, specifically its tolerance to air and moisture, greatly simplifies the logistics of raw material handling and storage. There is no longer a critical dependency on high-purity anhydrous solvents or inert gas supplies, which can sometimes face bottlenecks during global supply disruptions. The use of commercially available and stable selenide mediators ensures a consistent supply of key reagents, while the modular nature of electrochemical reactors allows for flexible production scaling. This reliability ensures that production schedules can be maintained with minimal interruption, securing the continuity of supply for downstream API manufacturers who depend on timely delivery of these critical building blocks.
- Scalability and Environmental Compliance: The patent demonstrates successful gram-scale synthesis, indicating a clear pathway for commercial scale-up of complex pharmaceutical intermediates. Electrochemical processes are inherently scalable through the numbering up of cells or the use of continuous flow electrochemical reactors, which offer superior heat and mass transfer characteristics compared to batch thermal processes. From an environmental standpoint, the avoidance of stoichiometric chemical oxidants reduces the generation of toxic byproducts, simplifying wastewater treatment and helping facilities meet stringent discharge limits. This alignment with green chemistry principles not only reduces regulatory risk but also enhances the brand reputation of the manufacturer as a leader in sustainable chemical production.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this electrocatalytic method, derived directly from the patent specifications and experimental data. Understanding these nuances is vital for technical teams evaluating the feasibility of integrating this process into existing manufacturing workflows. The answers reflect the specific advantages and operational parameters defined in the intellectual property, providing a clear framework for decision-making.
Q: Does this electrocatalytic method require strict anhydrous or anaerobic conditions?
A: No, one of the significant advantages of this patented process is that it operates efficiently under an air atmosphere at room temperature, eliminating the need for rigorous inert gas protection or anhydrous solvents typically required in transition metal catalysis.
Q: Are expensive transition metal catalysts like Palladium or Copper needed?
A: No, this method completely avoids the use of precious transition metal catalysts. Instead, it utilizes inexpensive selenide compounds as mediators and electricity as the primary driving force, significantly reducing raw material costs and downstream purification burdens.
Q: What is the role of the nitrile solvent in this reaction?
A: In this unique synthetic strategy, the nitrile solvent serves a dual function: it acts as the reaction medium and simultaneously participates as a reactant, providing the carbon and nitrogen atoms necessary to construct the oxazole ring structure.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2,4,5-Trisubstituted Oxazole Derivatives Supplier
As the global demand for high-quality pharmaceutical intermediates continues to rise, partnering with a technically proficient CDMO is essential for bringing novel therapies to market efficiently. NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that promising laboratory discoveries like this electrocatalytic method can be successfully translated into industrial reality. Our state-of-the-art facilities are equipped with advanced electrochemical synthesis capabilities and rigorous QC labs to guarantee stringent purity specifications for every batch. We understand the critical nature of supply chain stability and are committed to delivering consistent quality that meets the exacting standards of the international pharmaceutical industry.
We invite you to collaborate with us to explore the full potential of this green synthesis technology for your specific project needs. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your target molecule, demonstrating how this method can optimize your production economics. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us help you accelerate your development timeline with reliable, high-performance chemical solutions.
