Scalable Production of Bioactive Indole Dihydrofuran Derivatives Using Novel Rhodium Catalysis
The pharmaceutical industry is constantly seeking robust synthetic routes for complex heterocyclic scaffolds that serve as the backbone for next-generation therapeutics. Patent CN113999214B introduces a groundbreaking methodology for the preparation of indole dihydrofuran ring derivatives, a class of compounds renowned for their presence in bioactive natural products such as Naltrindole and Clausenaline D. These structures are not merely academic curiosities; they represent critical intermediates in the development of potent delta-opioid receptor antagonists and novel antitumor agents. The disclosed technology leverages a sophisticated metal-catalyzed strategy to construct these intricate bicyclic systems with high precision. By utilizing indole aldehydes and alkenyl diazo compounds as primary building blocks, the invention achieves a level of structural diversity and stereochemical control that was previously difficult to attain. This advancement is particularly significant for R&D teams focused on oncology and infectious disease, where the demand for new chemical entities with dual antitumor and antibacterial profiles is escalating rapidly.
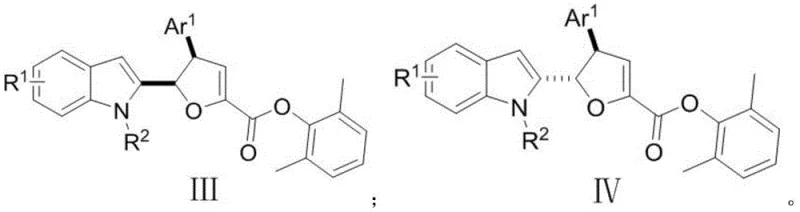
Furthermore, the versatility of this synthetic platform allows for the generation of a broad library of analogues, enabling medicinal chemists to perform extensive structure-activity relationship (SAR) studies efficiently. The ability to access both diastereomers, designated as formula (III) and formula (IV), through a controllable isomerization step provides a distinct advantage in optimizing pharmacokinetic properties. As a reliable pharmaceutical intermediate supplier, understanding the nuances of such patented methodologies is essential for ensuring a continuous supply of high-quality materials for drug discovery programs. The integration of these advanced synthetic tactics into commercial workflows promises to accelerate the timeline from lead identification to clinical candidate selection, addressing the urgent need for innovative therapies in the face of rising bacterial resistance and persistent malignancies.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of indole dihydrofuran skeletons has relied heavily on intramolecular or intermolecular [3+2] cycloaddition reactions, which, while powerful, often present significant challenges in terms of efficiency and selectivity. Traditional protocols frequently suffer from poor diastereoselectivity, resulting in complex mixtures of stereoisomers that require tedious and yield-losing chromatographic separations. Moreover, many existing methods necessitate harsh reaction conditions, expensive or toxic reagents, and multi-step sequences that degrade overall atom economy. For procurement managers, these inefficiencies translate directly into increased costs of goods sold (COGS) and extended lead times for critical research materials. The inability to consistently control the stereochemistry at the two chiral centers inherent in the dihydrofuran ring limits the biological utility of the resulting compounds, as different isomers can exhibit vastly different pharmacological profiles. Consequently, the search for a more streamlined, selective, and operationally simple synthesis has been a priority in the field of heterocyclic chemistry.
The Novel Approach
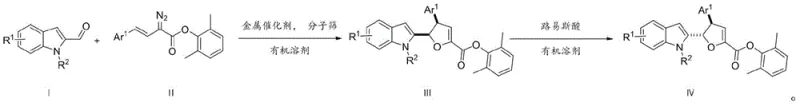
The methodology described in the patent data revolutionizes this landscape by employing a direct, one-step reaction between indole aldehydes and alkenyl diazo compounds catalyzed by dirhodium tetracarboxylates. This approach bypasses the need for pre-functionalized precursors and lengthy synthetic sequences, thereby drastically simplifying the operational workflow. The use of molecular sieves in the reaction mixture plays a crucial role in scavenging water, which ensures the stability of the sensitive diazo species and the rhodium catalyst, leading to consistently high yields. Furthermore, the subsequent treatment with a Lewis acid allows for the precise thermodynamic equilibration of the initial kinetic product (Formula III) to the more stable diastereomer (Formula IV). This level of control over the stereochemical outcome is a game-changer for process chemistry, as it eliminates the need for resolving racemic mixtures. The reaction proceeds under mild conditions, typically starting at 0°C and warming to room temperature, which enhances safety and reduces energy consumption compared to high-temperature alternatives.
Mechanistic Insights into Rhodium-Catalyzed [3+2] Cycloaddition
The core of this transformative synthesis lies in the generation of a reactive rhodium-carbenoid intermediate from the alkenyl diazo compound. Upon exposure to the dirhodium catalyst, such as Rh2(Oct)4, the diazo species undergoes denitrogenation to form a metal-bound carbene. This electrophilic species is then attacked by the nucleophilic C3 position of the indole ring, initiating the cascade that leads to ring closure. The transition state is highly organized, facilitated by the coordination of the carbonyl oxygen to the metal center, which directs the approach of the indole nucleophile and ensures the formation of the desired cis-relationship between the aryl group and the indole moiety in the initial product. This mechanistic pathway explains the high diastereoselectivity observed in the formation of Formula III. The reaction is essentially a formal [3+2] cycloaddition where the dipole is generated in situ, avoiding the instability associated with isolating 1,3-dipoles. Understanding this mechanism is vital for R&D directors aiming to troubleshoot potential scale-up issues or to modify the substrate scope for novel analogues.
Following the initial cyclization, the system offers a unique opportunity for stereochemical editing through Lewis acid catalysis. When the kinetic product (Formula III) is treated with a Lewis acid like scandium triflate (Sc(OTf)3) in a solvent like toluene at elevated temperatures (50°C), the dihydrofuran ring undergoes a reversible opening and closing or a concerted isomerization. This process allows the molecule to relax into the thermodynamically more stable diastereomer, Formula IV, where the stereochemistry at the benzylic position is inverted relative to the furan ring junction. This isomerization step is critical because it allows access to both stereoisomers from the same initial reaction setup simply by adjusting the post-reaction conditions. For quality control teams, this means that impurity profiles can be managed effectively, as the thermodynamic product is often the cleaner, more stable species. The ability to toggle between these forms adds a layer of strategic flexibility to the synthesis of complex pharmaceutical intermediates.
How to Synthesize Indole Dihydrofuran Derivatives Efficiently
The practical execution of this synthesis involves a carefully controlled addition of reagents to manage the exothermic nature of carbene formation and ensure optimal catalyst turnover. The protocol dictates dissolving the indole aldehyde and the rhodium catalyst in an anhydrous solvent such as ethyl acetate, along with activated molecular sieves to maintain a dry environment. The alkenyl diazo compound, dissolved separately, is then added dropwise via a syringe pump over a period of one hour at 0°C. This slow addition is paramount to preventing the dimerization of the diazo compound and minimizing side reactions. Once the addition is complete, the reaction is allowed to warm to room temperature to drive the conversion to completion. After filtration to remove the molecular sieves and catalyst residues, the crude product can be purified via column chromatography. For those requiring the alternative diastereomer, the purified intermediate is subjected to Lewis acid conditions in toluene. Detailed standardized synthesis steps follow below.
- Dissolve indolylaldehyde and rhodium catalyst in anhydrous ethyl acetate with molecular sieves to form the initial reaction mixture.
- Slowly add the alkenyl diazo compound solution via syringe pump at 0°C to control the exothermic carbene formation.
- Treat the resulting intermediate with a Lewis acid such as Sc(OTf)3 in toluene at elevated temperatures to achieve diastereomeric conversion.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this patented process offers substantial benefits that align perfectly with the goals of cost reduction and supply chain resilience in pharmaceutical manufacturing. The reliance on readily available starting materials, such as substituted indole aldehydes and diazo compounds, ensures that raw material sourcing is not a bottleneck. These precursors are commodity chemicals that can be procured from multiple global suppliers, mitigating the risk of single-source dependency. Furthermore, the reaction utilizes common organic solvents like ethyl acetate and toluene, which are inexpensive and easy to recover and recycle on an industrial scale. The elimination of complex multi-step sequences significantly reduces the overall processing time and labor costs associated with production. For procurement managers, this translates into a more predictable pricing structure and the ability to negotiate better terms due to the simplified logistics of the supply chain.
- Cost Reduction in Manufacturing: The streamlined nature of this one-pot synthesis drastically cuts down on the number of unit operations required, which directly lowers utility consumption and waste disposal costs. By avoiding the use of exotic or prohibitively expensive reagents and instead utilizing efficient rhodium catalysts at low loadings, the overall material cost is optimized. The high atom economy of the [3+2] cycloaddition means that a greater proportion of the reactant mass ends up in the final product, reducing the burden of waste treatment. Additionally, the ability to produce high-purity intermediates without extensive purification steps minimizes solvent usage and chromatography media costs, leading to significant savings in the cost of goods sold.
- Enhanced Supply Chain Reliability: The robustness of this chemical process ensures consistent batch-to-batch quality, which is critical for maintaining uninterrupted supply to downstream drug manufacturers. The mild reaction conditions reduce the risk of thermal runaways or safety incidents, thereby enhancing operational continuity. Since the raw materials are stable and widely traded, the supply chain is less susceptible to geopolitical disruptions or market volatility compared to processes relying on niche specialty chemicals. This reliability allows supply chain heads to plan inventory levels more accurately and reduce the need for excessive safety stock, freeing up working capital.
- Scalability and Environmental Compliance: The process is inherently scalable, having been demonstrated to work efficiently from milligram to gram scales in the patent examples, with clear pathways to kilogram and tonne production. The use of molecular sieves and standard filtration techniques simplifies the workup, making it amenable to continuous flow processing or large batch reactors. From an environmental standpoint, the reduction in waste generation and the use of less hazardous solvents align with green chemistry principles, facilitating easier regulatory approval and compliance with increasingly stringent environmental regulations. This sustainability profile is a key selling point for partnerships with major pharmaceutical companies committed to reducing their carbon footprint.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these indole dihydrofuran derivatives. These insights are derived directly from the technical specifications and experimental data provided in the patent documentation, ensuring accuracy and relevance for industry professionals. Understanding these details is crucial for evaluating the feasibility of integrating these intermediates into your own drug development pipelines.
Q: What are the primary biological activities of these indole dihydrofuran derivatives?
A: According to patent CN113999214B, these derivatives exhibit significant inhibitory effects against human colon cancer (HCT-116), breast cancer (MCF-7), non-small cell lung cancer (A549), and osteosarcoma cells, as well as antibacterial activity against Staphylococcus aureus and Acinetobacter baumannii.
Q: How does the new synthesis method improve upon conventional [3+2] cycloaddition techniques?
A: The novel method utilizes a one-step rhodium-catalyzed reaction between indole aldehydes and alkenyl diazo compounds, offering superior diastereoselectivity control and higher atom economy compared to traditional multi-step approaches that often struggle with stereochemical purity.
Q: Is this process suitable for large-scale commercial manufacturing?
A: Yes, the process employs commercially available raw materials, operates under mild conditions (0°C to 50°C), and uses standard organic solvents like ethyl acetate and toluene, making it highly scalable for industrial production of complex pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Indole Dihydrofuran Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the synthetic route disclosed in CN113999214B for accelerating the discovery of novel antitumor and antibacterial agents. As a premier CDMO partner, we possess the technical expertise and infrastructure to translate this laboratory-scale innovation into commercial reality. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project can move seamlessly from preclinical evaluation to clinical trials and beyond. We adhere to stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee that every batch of indole dihydrofuran derivatives meets the highest international standards.
We invite you to collaborate with us to leverage this advanced chemistry for your next breakthrough therapy. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and timeline. Please contact us today to request specific COA data and comprehensive route feasibility assessments. Let us help you secure a stable, high-quality supply of these critical pharmaceutical intermediates, enabling you to focus on what matters most: delivering life-saving medicines to patients worldwide.
