Scalable Synthesis of Lycopene Intermediates via Optimized Wittig Condensation Technology
The global demand for high-purity carotenoids, particularly lycopene, continues to surge across the pharmaceutical, nutraceutical, and food additive sectors, driving an urgent need for robust and scalable synthetic pathways. In this context, Chinese Patent CN102311320A presents a transformative approach to synthesizing 2,6,10-trimethyl-1,1-dialkoxy-3,5,9-undecatriene, a critical C14 acetal intermediate. This technology addresses long-standing bottlenecks in carotenoid manufacturing by replacing complex, multi-step phosphonate routes with a direct and highly efficient Wittig condensation. By leveraging readily available C10 triphenylphosphonium salts and C4 acetals, this method offers a compelling value proposition for reliable lycopene intermediate suppliers seeking to optimize their production portfolios. The innovation lies not just in the reagents, but in the precise kinetic control of the ylide formation, ensuring high selectivity and yield while drastically simplifying the downstream purification landscape.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of lycopene has been plagued by reliance on cumbersome Wittig-Horner reactions that utilize sensitive phosphonate esters. For instance, prior art described in WO 0031086 necessitates the use of 3,7,11-trimethyl-2,4,6,10-tetraenyl dodecyl phosphonate, which requires the handling of diethyl chlorophosphate (ClP(OEt)2). This reagent is notoriously sensitive to moisture and air, posing significant safety and storage challenges in large-scale agrochemical intermediate and pharmaceutical facilities. Furthermore, these traditional routes often mandate expensive hydrogenation steps using precious metal catalysts like Pd/C and reducing agents such as NaBH4 to manage double bond selectivity. The competitive nature of reducing conjugated double bonds versus isolated ones creates a nightmare for quality control, often resulting in complex impurity profiles that are difficult and costly to remove. Additionally, alternative routes utilizing C14 acetals have struggled with the availability of key precursors like 4-methyl-5,5-dialkoxy-1-pentene-1-phosphonic acid dialkyl esters, which are synthetically difficult to access and hinder commercial scale-up of complex carotenoid intermediates.
The Novel Approach
In stark contrast, the methodology disclosed in CN102311320A circumvents these hurdles by employing a direct coupling between a C10 triphenylphosphonium salt and a C4 acetal. This strategic shift eliminates the need for unstable chlorophosphonates and the subsequent reduction steps entirely. The process operates under mild conditions, typically between -40°C and 30°C, allowing for precise thermal management that preserves the integrity of the polyene chain. By decoupling the ylide formation from the aldehyde addition, the reaction achieves superior control over the stereochemistry, favoring the desired all-trans configuration essential for biological activity. This streamlined pathway not only reduces the number of unit operations but also utilizes commodity chemicals that are abundant in the global supply chain, thereby facilitating cost reduction in carotenoid manufacturing. The result is a process that is inherently safer, more economical, and significantly more amenable to multi-ton production scales compared to its predecessors.
Mechanistic Insights into the Optimized Wittig Condensation
The core of this technological breakthrough resides in the sequential addition protocol designed to maximize the concentration of the reactive ylide species before introducing the electrophile. Initially, the C10 triphenylphosphonium salt undergoes a dissociation reaction in the presence of a strong base, such as potassium tert-butoxide or sodium methylate, within an aprotic solvent system like THF or DMSO. This step is critical; by allowing the salt to fully dissociate into the corresponding carbanion at temperatures ranging from -20°C to 10°C, the system ensures a high local concentration of the nucleophile. This pre-formation minimizes side reactions that typically occur when base and aldehyde are present simultaneously, such as aldol condensations or polymerization of the sensitive aldehyde. Once the carbanion population is stabilized, the C4 acetal (2-methyl-3,3-dialkoxy-1-propanal) is introduced dropwise. This controlled addition maintains the reaction kinetics within an optimal window, preventing exothermic runaways that could degrade the delicate conjugated system.
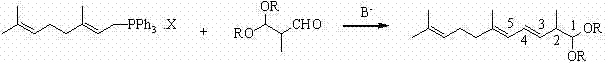
Following the condensation, the reaction mixture contains the target C14 acetal alongside triphenylphosphine oxide (TPPO) as the primary byproduct. The genius of this process is evident in the workup strategy, which exploits the differential solubility of these components. Upon quenching with water and extracting with organic solvents, the TPPO is effectively partitioned into a methanol-water phase, while the lipophilic C14 acetal remains in the n-hexane layer. This physical separation is far more efficient than chromatographic methods, enabling the production of high-purity material with a trans-to-cis ratio of approximately 3:1 when starting with the trans-phosphonium salt. Such mechanistic precision ensures that the final product meets the stringent purity specifications required for high-purity pharmaceutical intermediates, reducing the burden on downstream crystallization and purification units.
How to Synthesize 2,6,10-Trimethyl-1,1-dialkoxy-3,5,9-undecatriene Efficiently
Implementing this synthesis requires strict adherence to inert atmosphere protocols and temperature control to replicate the high yields reported in the patent data. The process begins with the preparation of the ylide solution, followed by the careful dosing of the acetal component. Operators must monitor the reaction progress via gas chromatography to determine the exact endpoint before proceeding to the biphasic extraction. This level of procedural discipline is essential for maintaining batch-to-batch consistency, especially when scaling from laboratory glassware to industrial reactors. For detailed operational parameters, stoichiometry, and safety guidelines, please refer to the standardized synthesis protocol outlined below.
- Dissociate C10 triphenylphosphonium salt with a strong base (e.g., potassium tert-butoxide) in an organic solvent at -40 to 30°C under inert gas.
- Add C4 acetal (2-methyl-3,3-dialkoxy-1-propanal) to the reaction mixture and maintain temperature for Wittig condensation.
- Perform aqueous workup using n-hexane and methanol-water to separate triphenylphosphine oxide byproducts and isolate the target C14 acetal.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented route offers tangible strategic benefits that extend beyond simple chemistry. By shifting away from custom-synthesized phosphonates to commodity phosphonium salts, companies can drastically simplify their raw material sourcing strategies. The precursors, such as geranyl chloride or bromide and triphenylphosphine, are produced globally in massive volumes, ensuring a stable supply base that is resistant to market fluctuations. This stability is crucial for reducing lead time for high-purity carotenoid intermediates, as it eliminates the long lead times associated with specialty reagent manufacturing. Furthermore, the elimination of precious metal catalysts like palladium removes a significant cost driver and supply chain risk, as the prices of these metals are notoriously volatile. The simplified workup also translates to reduced solvent consumption and waste generation, aligning with modern environmental compliance standards and lowering the total cost of ownership for the manufacturing facility.
- Cost Reduction in Manufacturing: The elimination of expensive hydrogenation catalysts and sensitive chlorophosphonates results in a substantial decrease in direct material costs. Additionally, the simplified purification process reduces energy consumption associated with distillation and chromatography, leading to significant operational expenditure savings. The high atom economy of the Wittig reaction further contributes to overall process efficiency, maximizing the output per unit of input.
- Enhanced Supply Chain Reliability: Sourcing relies on widely available bulk chemicals rather than niche intermediates, mitigating the risk of supply disruptions. The robustness of the reaction conditions allows for flexible manufacturing scheduling, as the process is less susceptible to minor variations in ambient conditions compared to moisture-sensitive alternatives. This reliability ensures consistent delivery schedules to downstream customers, strengthening long-term contractual relationships.
- Scalability and Environmental Compliance: The process generates triphenylphosphine oxide as the main waste stream, which is easier to handle and dispose of compared to heavy metal waste from hydrogenation steps. The use of standard organic solvents like hexane and THF fits well within existing solvent recovery infrastructure, minimizing environmental impact. The straightforward scale-up potential allows manufacturers to rapidly increase capacity to meet surging market demand without extensive capital investment in new reactor types.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. These answers are derived directly from the experimental data and claims within the patent documentation, providing a factual basis for decision-making. Understanding these nuances is vital for R&D teams evaluating the feasibility of technology transfer and for procurement teams assessing vendor capabilities.
Q: What is the stereochemical outcome of this Wittig reaction?
A: When using the trans-structured C10 triphenylphosphonium salt, the resulting C14 acetal product is predominantly all-trans, with a trans-to-cis ratio of approximately 3:1, minimizing downstream isomer separation costs.
Q: How does this method improve upon previous phosphonate-based routes?
A: Unlike prior art requiring sensitive chlorophosphonates and expensive hydrogenation catalysts, this route utilizes stable, commercially available triphenylphosphonium salts and avoids competitive reduction steps, significantly enhancing industrial feasibility.
Q: What is the purification strategy for removing triphenylphosphine oxide?
A: The process employs a specialized extraction where the byproduct triphenylphosphine oxide dissolves in a 1:1 methanol-water phase, while the target C14 acetal remains in the n-hexane layer, allowing for high-purity isolation without chromatography.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2,6,10-Trimethyl-1,1-dialkoxy-3,5,9-undecatriene Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory innovation to industrial reality requires a partner with deep technical expertise and proven manufacturing capacity. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical advantages of this Wittig-based route are fully realized in practice. We operate state-of-the-art facilities equipped with rigorous QC labs capable of verifying stringent purity specifications, including precise isomer ratios and residual solvent levels. Our commitment to quality assurance means that every batch of C14 acetal we produce is backed by comprehensive analytical data, giving our partners the confidence they need to integrate our materials into their own complex synthesis chains.
We invite you to engage with our technical procurement team to discuss how this advanced synthesis method can be tailored to your specific volume requirements. By requesting a Customized Cost-Saving Analysis, you can gain a clearer understanding of the economic benefits specific to your operation. We encourage potential partners to contact us directly to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions about securing a stable, high-quality supply of this critical lycopene intermediate for your global markets.
