Advanced Synthesis of C15 Acetate: A Strategic Upgrade for Carotenoid Intermediate Manufacturing
The landscape of fine chemical manufacturing is constantly evolving, driven by the relentless pursuit of efficiency and cost-effectiveness in producing complex molecular scaffolds. A pivotal advancement in this domain is detailed in patent CN103553916A, which discloses a highly efficient preparation method for 3,7,11-trimethyl-2,4,6,10-dodecatetraen-1-ol acetate, a critical intermediate often referred to as C15 acetate. This compound serves as a fundamental precursor in the synthesis of high-value carotenoids, including Lycopene, making its production methodology of paramount interest to the global pharmaceutical and nutraceutical sectors. The patent outlines a strategic shift from traditional, multi-step organometallic couplings to a streamlined Wittig-Horner condensation strategy. By leveraging a rearrangement dissociation of a C10 phosphonate followed by immediate condensation with a C5 aldehyde, this technology offers a robust pathway that addresses longstanding challenges in yield optimization and process safety. For industry leaders seeking a reliable pharma intermediate supplier, understanding the nuances of this patented route provides a competitive edge in securing supply chains for next-generation antioxidant ingredients.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of this specific terpene structure relied heavily on cumbersome and economically inefficient pathways that posed significant barriers to industrial scalability. As illustrated in the prior art, the conventional route typically commenced with phantol as a starting material, which was first converted into a C10 sulfone derivative through reaction with sodium benzenesulfinate. This intermediate then required a sophisticated coupling reaction with a pi-allylpalladium complex to extend the carbon chain, followed by a final desulfonylation step to reveal the target tetraene structure. 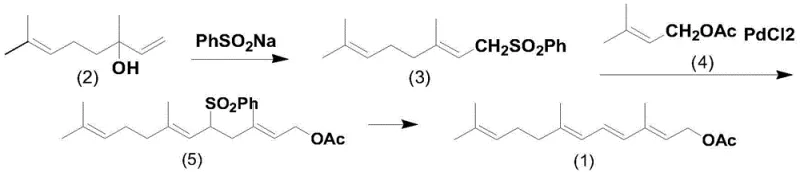 This legacy approach suffered from multiple critical drawbacks, including the necessity for expensive transition metal catalysts like Palladium, which not only inflated raw material costs but also introduced complex purification requirements to meet stringent heavy metal limits. Furthermore, the involvement of sulfone chemistry often generated substantial waste streams and required harsh reaction conditions, leading to lower overall atom economy and increased environmental compliance burdens for manufacturing facilities.
This legacy approach suffered from multiple critical drawbacks, including the necessity for expensive transition metal catalysts like Palladium, which not only inflated raw material costs but also introduced complex purification requirements to meet stringent heavy metal limits. Furthermore, the involvement of sulfone chemistry often generated substantial waste streams and required harsh reaction conditions, leading to lower overall atom economy and increased environmental compliance burdens for manufacturing facilities.
The Novel Approach
In stark contrast to the convoluted legacy methods, the innovative process described in the patent utilizes a direct and elegant convergent synthesis that dramatically simplifies the operational workflow. The core of this breakthrough lies in the utilization of 3,7-dimethyl-1,6-octadiene-diethyl phosphonate (C10 phosphonate) and 2-methyl-4-acetoxy-2-butylene aldehyde (C5 aldehyde) as the primary building blocks. 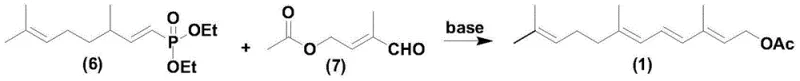 Under the protection of an inert gas, the C10 phosphonate undergoes a controlled rearrangement dissociation in the presence of a strong base to generate a reactive carbanion species. This activated intermediate is then immediately subjected to a Wittig-Horner condensation with the C5 aldehyde at mild temperatures ranging from -40°C to 30°C. This single-pot strategy effectively merges what were previously distinct synthetic operations, eliminating the need for isolation of unstable intermediates and removing the dependency on precious metal catalysts. The result is a process that is not only chemically superior in terms of selectivity but also operationally simpler, allowing for easier scale-up and reduced capital expenditure on specialized reactor equipment.
Under the protection of an inert gas, the C10 phosphonate undergoes a controlled rearrangement dissociation in the presence of a strong base to generate a reactive carbanion species. This activated intermediate is then immediately subjected to a Wittig-Horner condensation with the C5 aldehyde at mild temperatures ranging from -40°C to 30°C. This single-pot strategy effectively merges what were previously distinct synthetic operations, eliminating the need for isolation of unstable intermediates and removing the dependency on precious metal catalysts. The result is a process that is not only chemically superior in terms of selectivity but also operationally simpler, allowing for easier scale-up and reduced capital expenditure on specialized reactor equipment.
Mechanistic Insights into Wittig-Horner Condensation and Rearrangement
The success of this synthetic route hinges on the precise mechanistic execution of the rearrangement dissociation and subsequent nucleophilic attack, which dictates the stereochemical integrity and purity of the final product. The process initiates with the deprotonation of the C10 phosphonate by a strong base, such as potassium tert-butoxide, sodium ethoxide, or lithium diisopropylamide (LDA), to form a stabilized carbanion. This step is critical; the patent emphasizes that allowing the phosphonate to fully dissociate into the carbanion before introducing the electrophile ensures a higher concentration of the active nucleophile, thereby driving the equilibrium towards product formation. The reaction temperature is meticulously controlled between -40°C and 30°C to balance reaction kinetics with the stability of the polyene chain, preventing thermal degradation or isomerization that could compromise the biological activity of the downstream carotenoid. Following the formation of the carbanion, the addition of the C5 aldehyde triggers the condensation, where the nucleophilic carbon attacks the carbonyl carbon, eventually eliminating the phosphate group to form the new carbon-carbon double bond with high E/Z selectivity.
From an impurity control perspective, this mechanism offers inherent advantages over the sulfone-based alternatives. In the traditional route, side reactions involving the palladium catalyst could lead to complex organometallic byproducts that are notoriously difficult to remove without extensive chromatography. Conversely, the byproducts of the Wittig-Horner reaction in this novel method are primarily water-soluble phosphate salts, such as sodium diethyl phosphonate. These salts can be effortlessly removed through a simple aqueous workup procedure involving water and organic solvent layering, followed by washing with brine. This phase separation capability means that the crude product obtained after solvent removal is of significantly higher purity, often requiring only a final vacuum distillation to achieve specifications suitable for sensitive pharmaceutical applications. The ability to purify the product via distillation rather than column chromatography is a massive advantage for cost reduction in fine chemical manufacturing, as it drastically reduces solvent consumption and processing time.
How to Synthesize 3,7,11-Trimethyl-2,4,6,10-dodecatetraen-1-ol Acetate Efficiently
Implementing this synthesis requires strict adherence to the optimized conditions regarding base selection, solvent systems, and thermal profiles to ensure reproducibility and high yield. The patent data indicates that while various bases like sodium ethoxide and butyllithium are viable, alkali metal alkoxides such as potassium tert-butoxide in polar aprotic solvents like DMF or THF/DMSO mixtures provide excellent results. The molar ratio of the base to the aldehyde is maintained between 0.8 and 1.2 equivalents to ensure complete deprotonation without excessive waste. Operators must ensure the reaction vessel is thoroughly purged with inert gas to prevent oxidation of the sensitive polyene intermediates. The detailed standardized synthesis steps, including specific addition rates and quenching protocols, are outlined below to guide process engineers in replicating this high-efficiency route.
- Under inert gas protection, perform a rearrangement dissociation reaction on C10 phosphonate (3,7-dimethyl-1,6-octadiene-diethyl phosphonate) using an organic base at temperatures between -40°C and 30°C.
- Upon completion of dissociation, introduce C5 aldehyde (2-methyl-4-acetoxy-2-butylene aldehyde) to the reaction mixture to initiate the Wittig-Horner condensation.
- After the condensation is complete, perform an aqueous workup to separate the sodium salt byproduct, followed by solvent removal and vacuum distillation to isolate the pure C15 acetate.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented methodology translates into tangible strategic benefits that extend far beyond simple chemical yield improvements. The elimination of the Palladium catalyst represents a direct and substantial reduction in raw material costs, as precious metals constitute a significant portion of the expense in traditional organometallic syntheses. Furthermore, the simplified workup procedure, which relies on aqueous extraction rather than complex metal scavenging or chromatographic purification, significantly lowers the operational expenditure associated with solvent usage and waste disposal. This streamlining of the process flow enhances the overall throughput of the manufacturing facility, allowing for faster batch turnover and improved responsiveness to market demand fluctuations. By adopting this route, companies can achieve significant cost savings in production while simultaneously mitigating the supply risk associated with sourcing expensive and potentially volatile catalytic reagents.
- Cost Reduction in Manufacturing: The removal of expensive transition metal catalysts and the simplification of the purification train lead to a drastic decrease in the cost of goods sold (COGS). The process replaces costly sulfone reagents and palladium complexes with readily available phosphonates and aldehydes, which are generally more stable and cheaper to source in bulk quantities. Additionally, the ability to isolate the product via distillation rather than chromatography reduces solvent consumption and labor hours, contributing to a leaner and more profitable manufacturing model.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals like C10 phosphonate and C5 aldehyde, which are produced by established suppliers, ensures a stable and continuous supply of raw materials. Unlike specialized catalysts that may have long lead times or single-source dependencies, the key inputs for this process are widely available in the global chemical market. This diversification of the supply base reduces the risk of production stoppages due to raw material shortages, ensuring consistent delivery schedules for downstream customers.
- Scalability and Environmental Compliance: The process is inherently designed for scale, utilizing standard unit operations such as stirred tank reactors and distillation columns that are common in existing fine chemical plants. The generation of water-soluble phosphate byproducts simplifies effluent treatment compared to the heavy metal waste streams of the old method, facilitating easier compliance with increasingly stringent environmental regulations. This green chemistry profile not only reduces regulatory risk but also aligns with the sustainability goals of modern multinational corporations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and benefits of this advanced synthesis route. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on how this technology outperforms legacy methods in terms of purity, yield, and operational simplicity. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this process into their existing manufacturing portfolios.
Q: What are the primary advantages of the Wittig-Horner route over the traditional Palladium-catalyzed method?
A: The novel Wittig-Horner approach eliminates the need for expensive Palladium catalysts and toxic sulfone reagents, significantly reducing raw material costs and simplifying the purification process by avoiding heavy metal removal steps.
Q: What represents the critical control parameters for ensuring high yield in this synthesis?
A: Strict temperature control during the rearrangement dissociation phase (maintaining between -40°C and 30°C) and the precise stoichiometric ratio of the base (0.8 to 1.2 equivalents relative to the aldehyde) are essential for maximizing conversion and minimizing side reactions.
Q: Can this intermediate be directly converted into Lycopene precursors?
A: Yes, the resulting 3,7,11-trimethyl-2,4,6,10-dodecatetraen-1-ol acetate can be efficiently hydrolyzed under basic conditions to yield the corresponding alcohol, which serves as a vital building block for Lycopene and other carotenoid syntheses.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3,7,11-Trimethyl-2,4,6,10-dodecatetraen-1-ol Acetate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-purity intermediates in the successful development of life-saving drugs and premium nutraceuticals. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. We are committed to maintaining stringent purity specifications and operating rigorous QC labs to guarantee that every batch of 3,7,11-trimethyl-2,4,6,10-dodecatetraen-1-ol acetate meets the exacting standards required for carotenoid synthesis. Our expertise in optimizing Wittig-Horner condensations allows us to deliver this complex terpene intermediate with superior consistency and reliability.
We invite potential partners to engage with our technical procurement team to discuss how this innovative synthesis route can be tailored to your specific volume requirements and quality targets. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic benefits of switching to this streamlined process. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, ensuring that your supply chain is built on a foundation of technological excellence and commercial viability.
